Optimizing Nebivolol Production: A Strategic Analysis of Diastereomeric Cyanohydrin Coupling
Introduction to Advanced Nebivolol Synthesis
The pharmaceutical landscape for beta-blockers has evolved significantly with the introduction of third-generation agents like nebivolol, known for its unique nitric oxide-mediated vasodilatory effects. The patent CN101243062A details a groundbreaking preparation method for racemic nebivolol that addresses critical bottlenecks in traditional synthesis routes. Unlike previous methodologies that relied heavily on complex chromatographic separations of enantiomers, this invention leverages the strategic formation and separation of diastereomeric cyanohydrins. This approach not only simplifies the stereochemical control but also establishes a robust foundation for industrial scalability. By focusing on the relative configuration of chiral centers rather than absolute enantiomeric purity at early stages, the process achieves high yields of the active R*R*R*S* diastereomer through a clever coupling strategy.
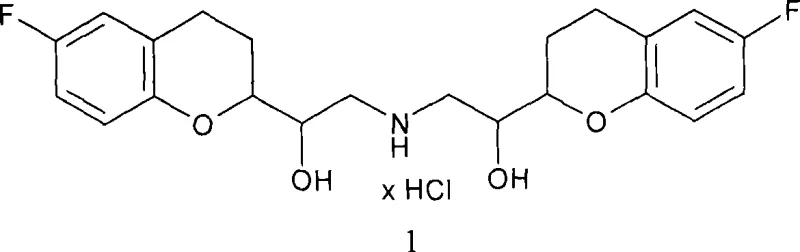
This technical breakthrough is particularly relevant for stakeholders seeking a reliable pharmaceutical intermediate supplier capable of delivering high-quality precursors for cardiovascular medications. The core innovation lies in the manipulation of cyanohydrin intermediates, which serve as versatile building blocks for constructing the complex bis-chromane skeleton of nebivolol. The method described ensures that the final product meets stringent pharmaceutical standards while optimizing the overall economic efficiency of the manufacturing process. For R&D directors and procurement managers, understanding this pathway offers valuable insights into cost reduction in API manufacturing and supply chain resilience.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of nebivolol has been plagued by significant technical and economic challenges, primarily stemming from the molecule's four chiral centers and its centrosymmetric structure. Traditional approaches often involved the direct synthesis of pure enantiomers or the separation of all possible stereoisomers using preparative chromatography. These chromatographic steps are notoriously difficult to scale, requiring specialized equipment, large volumes of solvents, and resulting in low throughput. Furthermore, literature indicates that converting synthetic methods designed for pure enantiomers into racemic active ingredient production often necessitates separate preparation and subsequent 1:1 mixing, which is inherently inefficient and costly from a synthetic design perspective. The reliance on such labor-intensive purification techniques creates a bottleneck that limits the ability to meet global demand efficiently.
The Novel Approach
The methodology presented in CN101243062A introduces a paradigm shift by utilizing diastereomeric cyanohydrins as key intermediates, specifically distinguishing between configurations A and B based on their relative stereochemistry. Instead of attempting to separate enantiomers directly, the process synthesizes a mixture of diastereomers which are then separated based on their distinct physical properties—specifically, their crystallization behavior. One diastereomer (A) is induced to crystallize while the other (B) remains in an oily state, facilitated by the use of silyl protecting groups. This physical differentiation allows for a technically simple and highly efficient separation via digestion in organic solvents like hexane, completely bypassing the need for chromatography. This novel approach streamlines the workflow, making the production of the racemic active ingredient more economically viable than even the synthesis of pure enantiomers in some contexts.
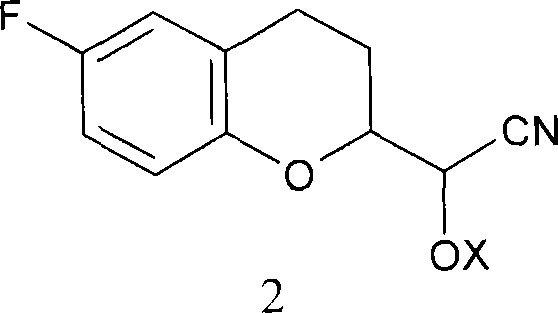
Mechanistic Insights into Diastereomeric Cyanohydrin Coupling
The heart of this synthesis lies in the precise manipulation of the cyanohydrin functionality and the strategic use of protecting groups to enable selective transformations. The process begins with the formation of racemic diastereomeric cyanohydrins from the corresponding aldehyde, preferably using tert-butyldimethylsilyl cyanide to introduce the silyl protecting group in a single step. This protection is crucial as it stabilizes the cyanohydrin and imparts the necessary lipophilicity and steric bulk to differentiate the diastereomers during the separation phase. Once separated, the two distinct molecular fragments—one serving as the amine precursor and the other as the aldehyde precursor—are processed independently. The cyanohydrin group proves exceptionally advantageous here, as it can be selectively reduced to an amine or transformed into an aldehyde depending on the specific reaction conditions employed, such as partial reduction with DIBAL or Pinner saponification.
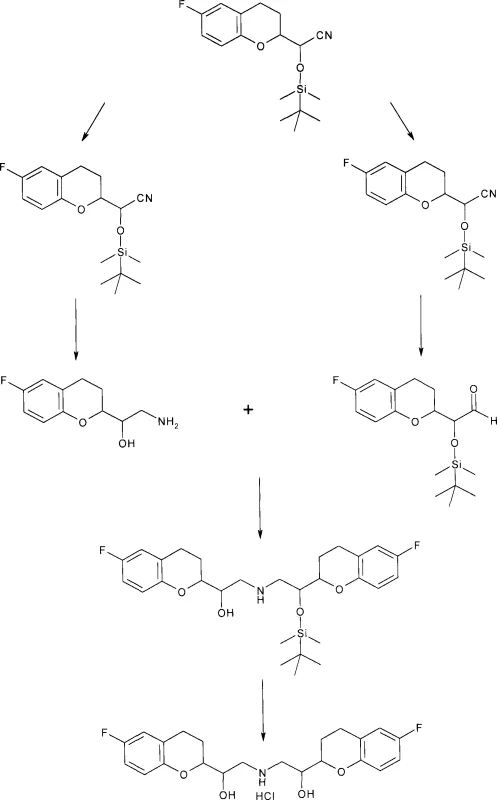
The coupling reaction itself is a masterpiece of chemoselectivity, typically achieved through reductive amination. By reacting the oxygen-protected aldehyde derived from one diastereomer with the oxygen-unprotected amine derived from the other, the process forms a Schiff base intermediate which is subsequently reduced using mild hydride sources like sodium cyanoborohydride. This one-pot transformation links the two chromane units to form the central secondary amine bridge characteristic of nebivolol. Importantly, the use of orthogonal protection strategies (one part protected, one part free) enhances the purification potential, allowing for the efficient removal of minor diastereomeric impurities formed during the coupling. The final deprotection step yields the target molecule, which can be further purified by recrystallization of its hydrochloride salt, leveraging the significant solubility differences between the desired R*R*R*S* isomer and the unwanted R*R*S*R* byproduct.
How to Synthesize Nebivolol Intermediates Efficiently
Implementing this synthesis requires careful control over reaction parameters to maximize the yield of the desired diastereomers and ensure effective separation. The initial cyanohydrin formation must be conducted under anhydrous conditions to prevent hydrolysis, followed by a controlled crystallization process where temperature and solvent choice (e.g., petroleum ether or hexane) are critical for isolating the crystalline diastereomer A. The remaining mother liquor, enriched in the oily diastereomer B, is then processed separately. Detailed standard operating procedures for these transformations, including specific molar ratios, temperature profiles, and workup protocols, are essential for reproducibility. For a comprehensive guide on executing these steps in a GMP environment, please refer to the standardized protocol below.
- Preparation of racemic diastereomeric cyanohydrins from 6-fluoro-chromene-2-carbaldehyde using silyl cyanide.
- Separation of diastereomers A (crystalline) and B (oily) via digestion in non-polar solvents like hexane.
- Conversion of separated intermediates into amine and aldehyde fragments, followed by reductive amination and deprotection.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented process offers substantial benefits that directly address the pain points of modern pharmaceutical supply chains. By eliminating the dependency on preparative chromatography, the method drastically reduces the consumption of expensive stationary phases and solvents, leading to significant operational cost savings. The shift towards crystallization-based purification not only lowers the cost of goods sold (COGS) but also enhances the environmental profile of the manufacturing process by reducing hazardous waste generation. For procurement managers, this translates into a more stable pricing structure for high-purity pharmaceutical intermediates, as the process is less susceptible to the volatility associated with specialized chromatographic resins and consumables.
- Cost Reduction in Manufacturing: The elimination of chromatographic steps is the primary driver for cost efficiency in this route. Chromatography is inherently batch-limited and resource-intensive, whereas crystallization and filtration are continuous-friendly and scalable unit operations. By relying on the differential crystallization of silylated diastereomers, manufacturers can achieve high purity without the capital expenditure and operational overhead of large-scale chromatography columns. This structural simplification of the process flow allows for a leaner manufacturing footprint, directly contributing to lower production costs and improved margin potential for the final API.
- Enhanced Supply Chain Reliability: The robustness of this synthesis pathway significantly mitigates supply chain risks. The reagents required, such as silyl cyanides and standard reducing agents, are commodity chemicals with established global supply networks, reducing the risk of raw material shortages. Furthermore, the ability to separate intermediates based on physical state (solid vs. oil) provides a built-in quality control checkpoint that ensures consistent batch-to-batch performance. This reliability is crucial for maintaining uninterrupted production schedules and meeting the rigorous delivery timelines demanded by downstream pharmaceutical partners.
- Scalability and Environmental Compliance: The process is inherently designed for commercial scale-up of complex pharmaceutical intermediates. The use of common organic solvents like toluene, MTBE, and hexane aligns with standard industrial safety and waste management protocols. Additionally, the high atom economy of the coupling steps and the ability to recycle mother liquors from the crystallization steps contribute to a greener manufacturing profile. This alignment with environmental, social, and governance (ESG) goals is increasingly important for multinational corporations seeking sustainable sourcing options for their active pharmaceutical ingredients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of nebivolol intermediates using this diastereomeric separation strategy. These insights are derived directly from the patent specifications and are intended to clarify the feasibility and advantages of this specific synthetic route for industry stakeholders. Understanding these nuances is vital for making informed decisions about technology transfer and vendor selection.
Q: How does this process improve purity compared to traditional chromatographic methods?
A: By utilizing the distinct physical properties of silylated diastereomers, where one crystallizes and the other remains oily, the process eliminates the need for expensive and low-throughput chromatographic separation, significantly enhancing scalability and purity.
Q: What is the key advantage of using TBDMS protection in this synthesis?
A: The tert-butyldimethylsilyl (TBDMS) group facilitates the critical separation step by inducing a phase difference between the diastereomers, allowing for efficient isolation via simple crystallization and digestion techniques.
Q: Can this method be scaled for commercial API production?
A: Yes, the reliance on standard unit operations such as crystallization, filtration, and reductive amination makes this route highly amenable to large-scale commercial manufacturing, unlike lab-scale chromatographic methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Nebivolol Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes for high-value cardiovascular drugs like nebivolol. Our technical team has extensively analyzed the diastereomeric cyanohydrin coupling strategy and possesses the expertise to implement this process at an industrial level. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to pilot plant and finally to full-scale manufacturing is seamless. Our facilities are equipped with rigorous QC labs capable of monitoring the critical diastereomeric ratios and ensuring stringent purity specifications are met for every batch of intermediates supplied.
We invite pharmaceutical companies and contract manufacturers to collaborate with us to leverage this advanced technology for their supply chains. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and discuss route feasibility assessments for your upcoming projects. Let us help you optimize your nebivolol supply chain with a solution that balances technical excellence with commercial viability.
