Advanced Synthesis of 6-Bromo-1-Chlorobenzothiophene Pyridine for Commercial Scale-Up
Introduction to Next-Generation Photoelectric Intermediates
The rapid evolution of organic light-emitting diode (OLED) technology and advanced pharmaceutical architectures demands intermediates with exceptional structural precision and purity. Patent CN113956268A introduces a groundbreaking synthetic methodology for 6-bromo-1-chlorobenzothiophene[2,3-c]pyridine, a critical building block for high-performance optoelectronic materials. This compound features a fused thiophene-pyridine skeleton with differentiated halogen handles at the 1 and 6 positions, enabling highly selective downstream functionalization for complex molecular designs. Unlike traditional approaches that struggle with regio-selectivity and hazardous reagents, this novel pathway leverages robust cross-coupling chemistry to deliver a single, high-purity product suitable for rigorous industrial applications. For R&D teams and procurement strategists alike, understanding this shift from unstable thiophenol chemistry to stable boronic acid coupling represents a significant opportunity to optimize supply chain reliability and reduce manufacturing costs in the competitive landscape of electronic chemical production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of benzothienopyridine derivatives has relied heavily on the iodination of 2-chloro-4-aminopyridine as a primary activation step. However, this classical route is plagued by severe regio-selectivity issues that render it economically unviable for large-scale operations. As documented in prior art, the iodination reaction yields a troublesome mixture of three distinct products: 2-chloro-5-iodo-4-aminopyridine, 2-chloro-3-iodo-4-aminopyridine, and di-iodinated species, typically in a chaotic ratio of roughly 45:45:10. Separating these structurally similar isomers requires extensive and costly chromatographic purification, which drastically reduces overall throughput and atom economy. Furthermore, subsequent coupling steps often utilize p-bromothiophenol, a reagent notorious for its high reactivity and tendency to oxidize into 1,2-bis(4-bromophenyl)disulfide, introducing difficult-to-remove sulfur-containing impurities that compromise the quality of the final API or electronic material.
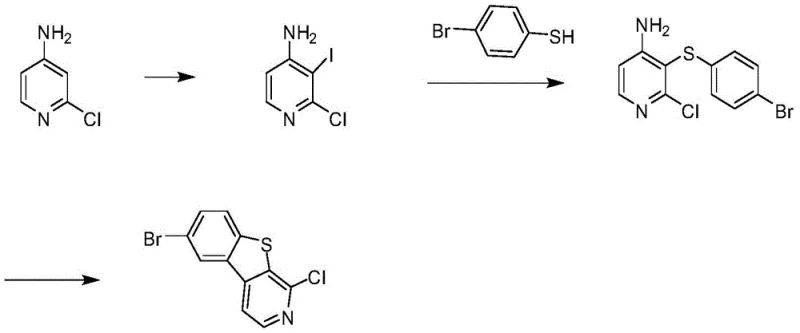
The Novel Approach
In stark contrast, the methodology disclosed in CN113956268A circumvents these historical bottlenecks by employing a Suzuki-Miyaura coupling strategy as the foundation of the synthesis. By utilizing 2-methylthio phenylboronic acid and 4-chloro-3-nitropyridine as starting materials, the process achieves a clean, regio-specific bond formation without generating isomeric byproducts. This strategic pivot eliminates the need for complex isomer separation entirely, streamlining the workflow from day one. Additionally, the use of a methyl-protected thioether group prevents the premature oxidation issues associated with free thiols, ensuring a cleaner reaction profile throughout the sequence. This modern approach not only enhances the chemical purity of the intermediates but also aligns perfectly with green chemistry principles by reducing solvent waste and energy consumption associated with repetitive purification cycles, offering a clear path toward cost reduction in organic photoelectric materials manufacturing.
Mechanistic Insights into Suzuki Coupling and Low-Temperature Chlorination
The core of this innovative synthesis lies in its five-step cascade, beginning with a palladium-catalyzed Suzuki coupling performed in a dioxane-water biphasic system. Under inert atmosphere protection, 2-methylthio phenylboronic acid reacts with 4-chloro-3-nitropyridine in the presence of an inorganic base such as potassium phosphate and a catalyst like tetrakis(triphenylphosphine)palladium(0). This step proceeds efficiently at moderate temperatures of 85-95°C, yielding the biaryl intermediate with high fidelity. Following this, a controlled bromination using elemental bromine in dichloroethane introduces the necessary halogen handle at the 5-position of the phenyl ring. The subsequent reduction of the nitro group to an amine is achieved safely using sodium hydrosulfite, avoiding the use of explosive hydrogenation catalysts. The cyclization to form the benzothiophene core is then triggered via a diazotization reaction using tert-butyl nitrite, which facilitates the intramolecular C-S bond formation under mild acidic conditions.
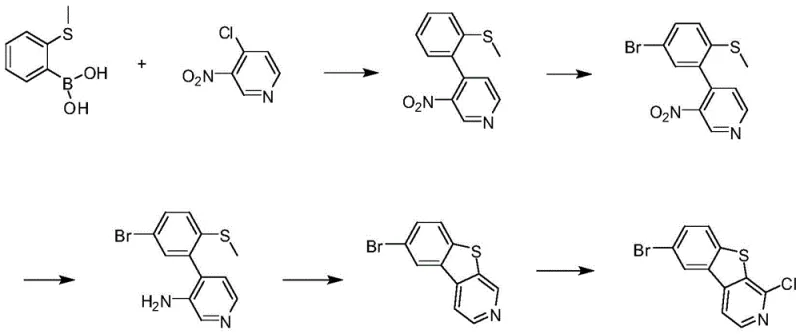
The final and perhaps most critical transformation is the selective chlorination at the 1-position of the pyridine ring. This step requires precise thermal management to ensure chemoselectivity, as the molecule contains multiple reactive sites. The process utilizes a strong non-nucleophilic base, such as lithium 2,2,6,6-tetramethylpiperidide (LTMP), to deprotonate the specific carbon adjacent to the nitrogen atom at temperatures between -20°C and 0°C. Subsequently, hexachloroethane is introduced as the chlorinating agent at cryogenic temperatures ranging from -75°C to -85°C. This extreme cold is essential to suppress side reactions and ensure that the chlorine atom is installed exclusively at the desired position without affecting the bromine substituent or the sensitive thiophene ring. This level of control demonstrates a sophisticated understanding of heterocyclic reactivity, resulting in a final product with purity exceeding 99%, which is paramount for high-end electronic applications where trace impurities can quench luminescence or alter charge transport properties.
How to Synthesize 6-Bromo-1-Chlorobenzothiophene[2,3-c]Pyridine Efficiently
Implementing this synthesis requires strict adherence to the optimized parameters regarding stoichiometry, temperature gradients, and solvent systems outlined in the patent data. The transition from laboratory bench scale to commercial production hinges on maintaining the integrity of the low-temperature chlorination step and ensuring efficient workup procedures after the Suzuki coupling. Operators must be trained to handle the cryogenic conditions safely while monitoring reaction progress via liquid chromatography to prevent over-reaction or decomposition. The following guide summarizes the critical operational phases required to achieve the high yields and purity specifications reported in the technical disclosure, serving as a foundational reference for process engineers planning the commercial scale-up of complex electronic chemical intermediates.
- Perform Suzuki coupling between 2-methylthio phenylboronic acid and 4-chloro-3-nitropyridine using Pd catalyst and inorganic base at 85-95°C.
- Conduct bromination of the intermediate using bromine in dichloroethane at 30-50°C to introduce the bromine substituent.
- Reduce the nitro group to an amine using sodium hydrosulfite in ethanol/water at 45-55°C.
- Execute diazotization and ring closure using tert-butyl nitrite in acetic acid/THF to form the benzothiophene core.
- Complete the synthesis via low-temperature chlorination (-75°C to -85°C) using hexachloroethane and an organic alkali base.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers tangible strategic benefits that extend beyond simple chemical yield. By shifting away from the problematic iodination-thiophenol pathway, manufacturers can secure a more stable and predictable supply of raw materials. The starting materials, specifically 2-methylthio phenylboronic acid and 4-chloro-3-nitropyridine, are commodity chemicals available from multiple global suppliers, reducing the risk of single-source dependency. Furthermore, the elimination of difficult isomer separations translates directly into reduced processing time and lower solvent consumption, which are major cost drivers in fine chemical manufacturing. This efficiency gain allows for a more competitive pricing structure without compromising on the stringent quality standards required by downstream clients in the OLED and pharmaceutical sectors.
- Cost Reduction in Manufacturing: The new process significantly lowers production costs by removing the need for expensive and time-consuming chromatographic separation of iodinated isomers. Since the Suzuki coupling produces a single regio-isomer, the downstream purification burden is drastically simplified, requiring only standard crystallization or extraction techniques. Additionally, the avoidance of hazardous hydrogenation catalysts and the use of inexpensive reducing agents like sodium hydrosulfite further contribute to substantial cost savings in reagent procurement and waste disposal.
- Enhanced Supply Chain Reliability: Supply continuity is bolstered by the use of robust, shelf-stable starting materials that do not suffer from the rapid oxidation issues seen with free thiophenols. This stability ensures consistent batch-to-batch quality and reduces the likelihood of production delays caused by degraded raw materials. The simplified reaction sequence also means shorter cycle times, enabling manufacturers to respond more agilely to fluctuating market demand and reducing lead time for high-purity photoelectric intermediates.
- Scalability and Environmental Compliance: The entire five-step sequence operates under atmospheric pressure and avoids extreme high-temperature or high-pressure conditions, making it inherently safer and easier to scale from pilot plants to multi-ton reactors. The absence of violent exotherms and the use of manageable solvents facilitate compliance with increasingly strict environmental and safety regulations. This operational safety profile minimizes the need for specialized high-pressure equipment, thereby lowering capital expenditure requirements for facility upgrades and ensuring long-term operational sustainability.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and advantages of this specific synthetic pathway. These insights are derived directly from the comparative data and experimental results presented in the patent literature, aiming to clarify the operational benefits for technical decision-makers. Understanding these nuances is crucial for evaluating the feasibility of integrating this intermediate into your existing product pipelines and for assessing the potential return on investment associated with switching to this superior manufacturing protocol.
Q: Why is the conventional iodination route unsuitable for industrial production?
A: The conventional method produces a complex mixture of three iodinated isomers in a 45:45:10 ratio, making separation extremely difficult and lowering overall yield significantly.
Q: How does the new Suzuki-based route improve impurity control?
A: By utilizing 2-methylthio phenylboronic acid instead of thiophenol, the process avoids the formation of disulfide byproducts and eliminates regio-isomer issues during the initial coupling step.
Q: What are the critical conditions for the final chlorination step?
A: The final chlorination requires strict temperature control between -75°C and -85°C using hexachloroethane and lithium tetramethylpiperidide to ensure selective substitution without degrading the heterocyclic core.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6-Bromo-1-Chlorobenzothiophene[2,3-c]Pyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of advanced organic photoelectric materials relies on a partner who can bridge the gap between innovative patent chemistry and reliable industrial production. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the intricate low-temperature chlorination and sensitive coupling steps are executed with precision. We maintain stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 6-bromo-1-chlorobenzothiophene[2,3-c]pyridine meets the exacting standards required for high-performance OLED displays and pharmaceutical applications. Our commitment to quality assurance means that you receive a product that is not only chemically pure but also consistent in its physical properties, facilitating smoother downstream processing for your R&D teams.
We invite you to collaborate with us to leverage this cutting-edge synthetic technology for your next project. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that enhance your supply chain resilience and drive innovation in your product development pipeline. Let us be your trusted partner in delivering high-value chemical solutions that power the future of electronics and medicine.
