Advanced Enzymatic Route for High-Purity Chiral (S)-3-Piperidine Alcohols Manufacturing
The pharmaceutical industry's relentless pursuit of high-purity chiral intermediates has led to significant innovations in biocatalysis, specifically regarding the synthesis of piperidine derivatives which serve as critical scaffolds for numerous therapeutic agents. Patent CN103898178A introduces a groundbreaking enzymatic method for preparing highly chirally pure (S)-3-piperidine alcohols and their derivatives, addressing long-standing challenges in stereochemical control and process efficiency. This technology leverages a sophisticated dual-enzyme system involving recombinant carbonyl reductase and glucose dehydrogenase co-expressed in Escherichia coli, enabling the asymmetric reduction of prochiral ketones under mild aqueous conditions. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, this patent represents a paradigm shift from traditional chemical resolution to sustainable, high-yield biosynthesis. The method not only ensures exceptional optical purity but also drastically simplifies the downstream processing required to meet stringent regulatory standards for active pharmaceutical ingredients (APIs).
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of chiral 3-piperidine alcohols has relied heavily on chemical resolution techniques or multi-step syntheses starting from expensive natural chiral acids such as L-malic acid or L-aspartic acid. These conventional approaches are fraught with inefficiencies; for instance, chiral resolution typically suffers from a theoretical maximum yield of only 50%, necessitating the recycling or disposal of the unwanted enantiomer, which inflates raw material costs and waste generation. Furthermore, processes involving chiral acids often require complex condensation and reduction steps that are difficult to scale industrially due to harsh reaction conditions and the need for extensive purification via silica gel column chromatography. The reliance on stoichiometric amounts of expensive resolving agents and the generation of substantial chemical waste make these traditional routes economically unsustainable for large-scale manufacturing, creating significant bottlenecks in the supply chain for key oncology and cardiovascular drug intermediates.
The Novel Approach
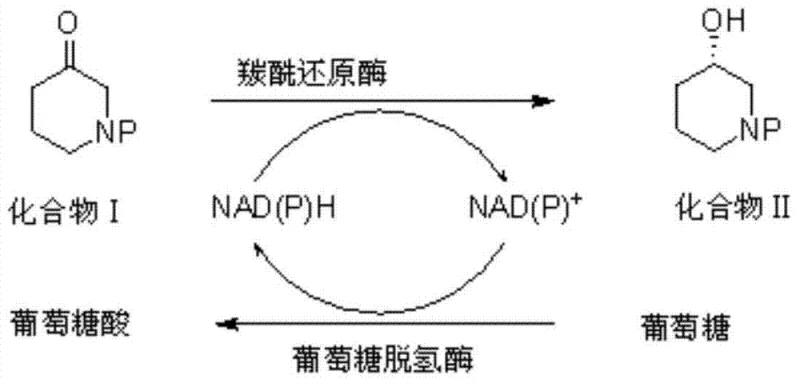
In stark contrast, the novel enzymatic approach detailed in the patent utilizes a highly engineered biocatalytic system that overcomes the yield barriers of kinetic resolution by employing asymmetric reduction. By utilizing a recombinant carbonyl reductase derived from Sporobolomyces salmonicolor and a glucose dehydrogenase from Burkholderia sp., the process achieves near-quantitative conversion of the ketone substrate to the desired (S)-alcohol with an optical purity exceeding 99.9% ee. This method operates at high substrate concentrations, reaching up to 150g/L, which is a substantial improvement over previous biocatalytic attempts that were limited to less than 5g/L. The integration of a coenzyme regeneration cycle eliminates the need for stoichiometric amounts of expensive NADPH, while the use of whole-cell or crude enzyme preparations simplifies the catalyst preparation process. This breakthrough offers a viable pathway for cost reduction in API manufacturing by merging high efficiency with environmental sustainability.
Mechanistic Insights into Dual-Enzyme Catalytic Reduction
The core of this technological advancement lies in the synergistic action of two distinct enzymes working in tandem to drive the thermodynamic equilibrium towards the desired chiral product. The primary catalyst, a mutated carbonyl reductase, selectively reduces the carbonyl group of the 3-piperidone derivative to a hydroxyl group with strict stereocontrol, favoring the formation of the (S)-enantiomer. However, this reduction consumes the reduced cofactor NADPH, converting it to NADP+. To sustain the reaction without prohibitive costs, a second enzyme, glucose dehydrogenase (GDH), is employed to regenerate NADPH from NADP+ by oxidizing glucose to gluconic acid. This creates a closed-loop catalytic cycle where the expensive cofactor is recycled continuously, allowing it to be used in catalytic quantities rather than stoichiometric ones. The genetic engineering of these enzymes, involving specific amino acid mutations to enhance activity and stability in organic solvent mixtures, ensures that the biocatalyst remains robust throughout the extended reaction times required for high conversion rates.
Beyond the primary reduction mechanism, the process incorporates sophisticated impurity control strategies inherent to enzymatic specificity. Unlike chemical reducing agents such as sodium borohydride or lithium aluminum hydride, which may reduce other functional groups or cause racemization under certain conditions, the engineered carbonyl reductase exhibits exquisite chemoselectivity for the ketone moiety even in the presence of N-protecting groups like Boc, Cbz, or Fmoc. The reaction conditions are maintained within a narrow pH window of 6.0 to 7.5 and temperatures between 25°C and 40°C, which minimizes the risk of side reactions such as elimination or epimerization that often plague chemical syntheses. Furthermore, the use of a biphasic system or the addition of organic co-solvents like butyl acetate helps to solubilize the hydrophobic substrate while maintaining enzyme stability, thereby preventing substrate inhibition and ensuring a clean reaction profile that simplifies the final isolation of the high-purity product.
How to Synthesize (S)-N-Boc-3-Piperidine Alcohol Efficiently
The practical implementation of this synthesis involves a straightforward protocol that can be adapted for both laboratory screening and industrial fermentation. The process begins with the preparation of a buffered aqueous solution, typically using sodium phosphate buffers adjusted to a pH of roughly 6.5, which provides the optimal environment for enzyme activity. The substrate, such as N-Boc-3-piperidone, is introduced into the reaction vessel along with a molar excess of glucose, which serves as the hydrogen donor for the cofactor regeneration cycle. The reaction is initiated by the addition of the biocatalyst, which can be supplied as a lyophilized powder, a liquid crude enzyme extract, or immobilized cells, offering flexibility depending on the specific manufacturing setup. Detailed standardized synthesis steps for replicating this high-efficiency transformation are provided in the technical guide below.
- Prepare a reaction mixture containing the ketone substrate (e.g., N-Boc-3-piperidone), glucose as a co-substrate, and a buffered solution maintained at pH 6.0-7.5.
- Add the recombinant enzyme system comprising carbonyl reductase and glucose dehydrogenase along with a catalytic amount of NADP+ cofactor.
- Maintain the reaction at 25-40°C with stirring until conversion exceeds 99%, then extract the product using organic solvents like ethyl acetate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this enzymatic platform offers profound strategic benefits that extend beyond simple yield improvements. The elimination of expensive chiral resolving agents and the reduction in solvent usage directly translate to a leaner cost structure, making the final intermediate more price-competitive in a volatile market. Moreover, the ability to operate at high substrate loadings means that reactor volume utilization is maximized, significantly increasing throughput without the need for proportional capital expenditure on new equipment. The robustness of the engineered enzymes allows for consistent batch-to-batch quality, reducing the risk of production delays caused by failed batches or out-of-specification results that often disrupt supply continuity for critical drug substances.
- Cost Reduction in Manufacturing: The enzymatic process fundamentally alters the cost equation by replacing stoichiometric chiral reagents with catalytic biological systems. Since the cofactor NADPH is regenerated in situ using inexpensive glucose, the material cost associated with oxidation-reduction chemistry is drastically minimized. Additionally, the high stereoselectivity removes the need for costly and time-consuming chiral chromatography or multiple recrystallization steps typically required to upgrade optical purity in chemical routes. This streamlined downstream processing not only saves on consumables but also reduces labor and utility costs, resulting in substantial overall savings for the production of complex pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Reliance on natural chiral pool materials often exposes manufacturers to agricultural volatility and price fluctuations; however, this biocatalytic route relies on fermentation-derived enzymes and commodity chemicals like glucose, which are readily available globally. The patent demonstrates successful scale-up in 100L fermenters, proving that the supply of the biocatalyst itself can be secured through established industrial fermentation practices. This decentralization of raw material risk ensures a more stable and predictable supply chain, allowing pharmaceutical companies to plan long-term production schedules with greater confidence and reduced exposure to raw material shortages.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this green chemistry approach aligns perfectly with modern sustainability goals. The reaction proceeds in aqueous buffers with mild pH and temperature conditions, significantly reducing the energy footprint compared to high-temperature chemical reductions. The waste stream is primarily composed of biodegradable gluconic acid and biomass, avoiding the heavy metal contamination or toxic solvent waste associated with traditional catalytic hydrogenation or borohydride reductions. This cleaner profile simplifies waste treatment compliance and facilitates easier regulatory approval for the manufacturing process, accelerating the time to market for new drug applications relying on this key intermediate.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this enzymatic technology. These answers are derived directly from the experimental data and embodiments disclosed in the patent documentation, providing a clear understanding of the process capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing workflows.
Q: What is the optical purity achievable with this enzymatic method?
A: The patented enzymatic process consistently achieves an optical purity (ee value) greater than 99.9%, significantly surpassing traditional chemical resolution methods which often struggle to exceed 98% without multiple recrystallizations.
Q: How does the coenzyme recycling system function in this synthesis?
A: The system utilizes glucose dehydrogenase (GDH) to regenerate the reduced cofactor NADPH from NADP+ using glucose as a sacrificial donor, allowing the expensive cofactor to be used in catalytic rather than stoichiometric amounts.
Q: Can this process be scaled for industrial production?
A: Yes, the patent demonstrates successful fermentation in 100L tanks with substrate concentrations up to 150g/L, indicating robust scalability for commercial manufacturing of pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (S)-3-Piperidine Alcohols Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality chiral intermediates play in the development of next-generation therapeutics, particularly in the fields of oncology and cardiovascular health. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent supply of materials that meet the most rigorous quality standards. Our state-of-the-art facilities are equipped with advanced fermentation capabilities and stringent purity specifications, supported by rigorous QC labs that utilize chiral HPLC and other analytical techniques to verify optical purity greater than 99.9% ee for every batch produced.
We invite potential partners to engage with our technical procurement team to discuss how this enzymatic technology can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this biocatalytic route for your specific API synthesis. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our commitment to delivering superior value and reliability as your trusted partner in fine chemical manufacturing.
