Scalable Synthesis of Asymmetric Diaminofluorene Derivatives for Next-Gen OLED Display Materials
The rapid evolution of the organic light-emitting diode (OLED) industry has created an insatiable demand for novel electroluminescent materials that offer superior thermal stability and fluorescence quantum efficiency. At the forefront of this technological shift is the development of specialized fluorene derivatives, which serve as critical building blocks for high-performance display technologies. Patent CN103664647A introduces a groundbreaking preparation method for 2,3-diamino-7-bromo-9,9'-dialkylfluorene, a sophisticated intermediate that addresses long-standing challenges in molecular design. This innovation represents a significant leap forward by successfully synthesizing a series of asymmetric ortho-diaminofluorene structures that were previously difficult to access with high regioselectivity. By integrating a bromine substituent directly into the 2,3-diamino-9,9'-dialkylfluorene framework, this technology unlocks new possibilities for downstream derivatization, making it an invaluable asset for researchers aiming to optimize the band-gap energy and charge transport properties of next-generation OLED devices.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of functionalized fluorene derivatives for electronic applications has been plagued by issues related to regioselectivity and the limited diversity of substitution patterns. Conventional methods often struggle to introduce multiple functional groups, such as amino and halogen substituents, at specific positions on the fluorene ring without causing unwanted side reactions or forming complex mixtures of isomers. For instance, attempting to introduce amino groups at the 2 and 3 positions while maintaining a reactive halogen at the 7 position typically requires harsh conditions that can degrade the sensitive fluorene backbone or lead to poor yields. Furthermore, existing routes frequently lack the versatility to accommodate various alkyl chains at the 9-position, limiting the solubility and processability of the final OLED materials. These limitations not only increase the cost of raw materials due to low efficiency but also create significant bottlenecks in the supply chain for high-purity electronic chemicals, forcing manufacturers to rely on expensive purification techniques to remove stubborn impurities.
The Novel Approach
The methodology outlined in the patent data offers a robust solution by employing a stepwise strategy that prioritizes selectivity and modularity. Instead of attempting simultaneous substitutions, the process begins with a readily available 2-bromo-7-nitrofluorene scaffold, which serves as a stable foundation for sequential functionalization. This approach allows for the precise installation of alkyl groups at the 9-position under mild basic conditions, followed by a carefully controlled reduction of the nitro group. A key innovation lies in the use of an acetyl protection group, which shields the newly formed amino functionality during subsequent nitration steps, ensuring that the second nitrogen-containing group is introduced exclusively at the desired 3-position. This logical progression not only maximizes the overall yield but also simplifies the purification process, as each intermediate can be isolated with high purity. By decoupling the introduction of different functional groups, this novel route provides a reliable pathway for producing a diverse library of asymmetric fluorene derivatives tailored for specific optoelectronic requirements.
Mechanistic Insights into Multi-Step Functionalization and Protection Strategies
The core of this synthesis lies in its meticulous management of reactivity through a six-step sequence that balances nucleophilic substitutions, reductions, and electrophilic aromatic substitutions. The process initiates with the alkylation of 2-bromo-7-nitrofluorene using a mineral alkali and a bromoalkane in a polar aprotic solvent like dimethyl sulfoxide (DMSO). This step effectively installs the solubilizing alkyl chains at the 9-position, which are crucial for the processing characteristics of the final OLED material. Following alkylation, the nitro group at the 7-position is selectively reduced to an amino group using versatile reducing systems such as iron powder with hydrochloric acid or catalytic hydrogenation with Raney Nickel. This transformation is critical as it converts an electron-withdrawing group into an electron-donating one, altering the electronic landscape of the molecule for subsequent reactions. The resulting amine is then protected via acetylation with acetic anhydride, a strategic move that deactivates the ring towards over-nitration and directs the incoming nitronium ion to the specific 3-position adjacent to the protected amine.
Following the regioselective nitration, the synthesis proceeds with the hydrolysis of the acetyl protecting group using concentrated sulfuric acid, regenerating the free amino group at the 2-position while retaining the newly installed nitro group at the 3-position. The final step involves the reduction of this second nitro group to an amino group, completing the formation of the 2,3-diamino motif. Throughout this cascade, the bromine atom at the 7-position remains intact, serving as a dormant handle for future cross-coupling reactions. This preservation of the halogen substituent is paramount, as it enables the molecule to participate in palladium-catalyzed coupling reactions like Suzuki or Heck couplings in later stages of OLED material synthesis. The ability to maintain this reactive site while building up the complex amino-substituted core demonstrates a sophisticated understanding of orthogonal reactivity, ensuring that the final product is not just a static molecule but a dynamic precursor ready for further molecular engineering.
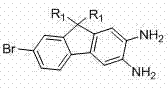
How to Synthesize 2,3-Diamino-7-Bromo-9,9'-Dialkylfluorene Efficiently
The synthesis of this high-value intermediate requires precise control over reaction parameters to ensure the integrity of the fluorene backbone and the correct positioning of substituents. The process leverages common industrial reagents and standard separation techniques, making it highly adaptable for scale-up operations. Operators must pay close attention to the stoichiometry of the alkylating agents and the temperature profiles during the reduction steps to prevent the formation of byproducts. The use of protective groups adds an extra layer of complexity but is essential for achieving the required purity levels for electronic grade materials. Detailed standard operating procedures regarding solvent choices, work-up protocols, and crystallization conditions are critical for reproducibility. For a comprehensive breakdown of the specific reaction conditions, molar ratios, and isolation techniques validated in the patent examples, please refer to the standardized synthesis guide below.
- Alkylation of 2-bromo-7-nitrofluorene using mineral alkali and bromoalkane in DMSO to form the 9,9-dialkyl precursor.
- Selective reduction of the nitro group to an amino group using iron powder/hydrochloric acid or catalytic hydrogenation systems.
- Protection of the amino group via acetylation, followed by regioselective nitration at the 3-position and subsequent hydrolysis and reduction to yield the final diamino product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this synthesis route offers compelling advantages driven by the use of commodity chemicals and robust process chemistry. The reliance on widely available starting materials such as nitrofluorenes, common bromoalkanes, and standard mineral acids significantly mitigates the risk of raw material shortages that often plague specialty chemical supply chains. Furthermore, the reaction conditions operate within a moderate temperature range, eliminating the need for extreme cryogenic cooling or high-pressure equipment that would otherwise drive up capital expenditure and operational costs. The modular nature of the synthesis allows for flexible production scheduling, where intermediates can be stockpiled if necessary, enhancing the overall resilience of the supply network. By optimizing the yield at each step through selective protection strategies, the process minimizes waste generation and reduces the burden on downstream purification units, leading to substantial cost savings in the overall manufacturing lifecycle.
- Cost Reduction in Manufacturing: The elimination of exotic catalysts and the use of inexpensive reducing agents like iron powder or hydrazine hydrate drastically lower the direct material costs associated with production. Additionally, the high selectivity of the reaction sequence reduces the formation of difficult-to-remove impurities, which in turn lowers the consumption of expensive chromatography media or recrystallization solvents. This efficiency translates directly into a more competitive pricing structure for the final electronic chemical, allowing downstream OLED manufacturers to optimize their own bill of materials without compromising on performance specifications.
- Enhanced Supply Chain Reliability: Because the synthesis does not depend on single-source proprietary reagents or unstable intermediates, the supply chain is inherently more stable and less susceptible to geopolitical or logistical disruptions. The ability to source alkylating agents and solvents from multiple global suppliers ensures continuity of supply even in volatile market conditions. Moreover, the scalability of the process means that production volumes can be ramped up quickly to meet surging demand from the display industry, reducing lead times for high-purity OLED material precursors and enabling faster time-to-market for new device architectures.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing unit operations that are standard in fine chemical manufacturing facilities, such as liquid-liquid extraction and vacuum filtration. This compatibility with existing infrastructure facilitates a smoother technology transfer from laboratory to pilot plant and finally to commercial scale. From an environmental standpoint, the potential to use catalytic hydrogenation or iron-based reductions offers alternatives to more hazardous stoichiometric reducers, aligning with increasingly stringent global regulations on industrial emissions and waste disposal, thereby reducing the environmental compliance burden for manufacturers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this specialized fluorene derivative. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity on the feasibility and benefits of adopting this synthesis route. Understanding these details is crucial for R&D teams evaluating new material platforms and procurement officers assessing supplier capabilities. The answers reflect a commitment to transparency and technical excellence, ensuring that all stakeholders have the accurate information needed to make informed decisions about integrating this intermediate into their supply chains.
Q: What is the primary advantage of introducing a bromine substituent in this fluorene derivative?
A: The introduction of a bromine substituent at the 7-position significantly enriches the reaction sites of the fluorene molecule. Unlike simple diaminofluorenes, this asymmetric structure allows for further functionalization via metal coupling reactions such as Heck, Suzuki, and Sonogashira couplings, providing greater flexibility in designing complex OLED emissive layers.
Q: How does this synthesis method ensure high purity for electronic applications?
A: The process utilizes a strategic protection-deprotection strategy involving acetylation. By protecting the 2-amino group before nitration, the method prevents unwanted side reactions and ensures that the nitro group is introduced selectively at the 3-position. Subsequent hydrolysis and reduction steps are optimized to minimize impurities, resulting in a high-purity intermediate suitable for sensitive optoelectronic applications.
Q: Is this process scalable for industrial production of OLED materials?
A: Yes, the synthesis relies on standard unit operations such as solvent extraction, filtration, and crystallization, using commercially available reagents like iron powder, hydrazine hydrate, and common mineral acids. The reaction conditions, ranging from room temperature to moderate heating (up to 100°C), are easily manageable in large-scale reactors, facilitating commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3-Diamino-7-Bromo-9,9'-Dialkylfluorene Supplier
As the demand for high-performance OLED materials continues to accelerate, partnering with a technically proficient manufacturer is essential for securing a competitive edge in the market. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging deep expertise in complex organic synthesis to deliver superior electronic chemical intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous volume requirements of global display panel manufacturers. We are committed to maintaining stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify the structural integrity and impurity profile of every batch, guaranteeing that our products perform consistently in your final device applications.
We invite you to collaborate with us to explore how this innovative synthesis route can enhance your product portfolio and reduce your overall manufacturing costs. Our technical sales team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production needs, demonstrating exactly how our efficient processes can improve your bottom line. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, allowing you to validate the quality and suitability of our 2,3-diamino-7-bromo-9,9'-dialkylfluorene for your next-generation OLED projects with confidence.
