Scalable Synthesis of N-n-butylsulfonyl-O-(4-(4-pyridyl)-butyl)-L-tyrosine Alkyl Ester for Global Pharma Supply
Scalable Synthesis of N-n-butylsulfonyl-O-(4-(4-pyridyl)-butyl)-L-tyrosine Alkyl Ester for Global Pharma Supply
The pharmaceutical landscape for cardiovascular therapeutics continues to evolve, with Tirofiban hydrochloride remaining a critical non-peptide GPIIb/IIIa receptor antagonist for treating unstable angina and preventing myocardial infarction. As demand for high-purity active pharmaceutical ingredients (APIs) grows, the efficiency of synthesizing key intermediates becomes a paramount concern for global supply chains. Patent CN102285912A introduces a transformative methodology for preparing N-n-butylsulfonyl-O-(4-(4-pyridyl)-butyl)-L-tyrosine alkyl ester, a pivotal precursor in the Tirofiban synthesis pathway. This innovation addresses long-standing challenges in process chemistry by replacing hazardous reagents and complex purification steps with a streamlined, two-step sequence that utilizes mild reaction conditions and phase transfer catalysis. For R&D directors and procurement specialists, understanding the nuances of this patent is essential for securing a reliable pharmaceutical intermediate supplier capable of delivering consistent quality at scale.
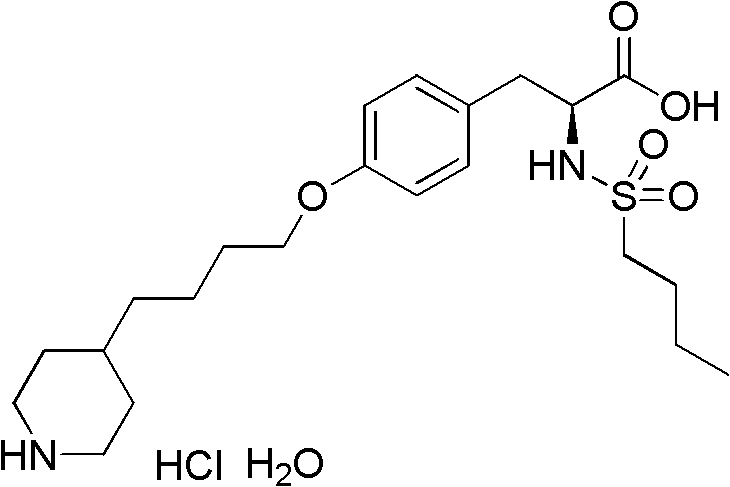
Tirofiban hydrochloride, originally developed by Merck, represents a cornerstone in antiplatelet therapy, yet its complex molecular architecture has historically necessitated convoluted synthetic routes. The structural integrity of the tyrosine backbone coupled with the specific alkylation of the phenolic oxygen requires precise chemical control to avoid racemization and impurity formation. The method disclosed in CN102285912A offers a robust solution by focusing on the efficient construction of the ether linkage between the tyrosine derivative and the pyridyl-butyl chain. By shifting away from traditional activation methods that rely on heavy metals or cryogenic conditions, this technology opens new avenues for cost reduction in API manufacturing while adhering to stricter environmental regulations. The ability to produce this intermediate with fewer steps and higher total yield directly translates to enhanced supply chain reliability for downstream drug manufacturers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Tirofiban and its intermediates has been plagued by operational inefficiencies and safety hazards inherent in early-generation protocols. For instance, Patent US750647 describes an original route originating from 4-piperidine-2-ethanol that spans eleven distinct steps, involving harsh transformations such as Swern oxidation, Wittig reactions, and borane reductions. This lengthy sequence inevitably results in a low overall yield, and critically, the tenth step requires column chromatography purification, a technique that is notoriously difficult to translate to industrial tonnage production. Similarly, Patent CN1844099 attempts to streamline the process but introduces significant risks by utilizing highly reactive sodium metal (99.5%) for reduction steps, which poses severe safety liabilities in a plant environment. Furthermore, the reliance on 4-(4-pyridyl) butyl iodide generated in situ can lead to reproducibility issues and inconsistent batch quality.
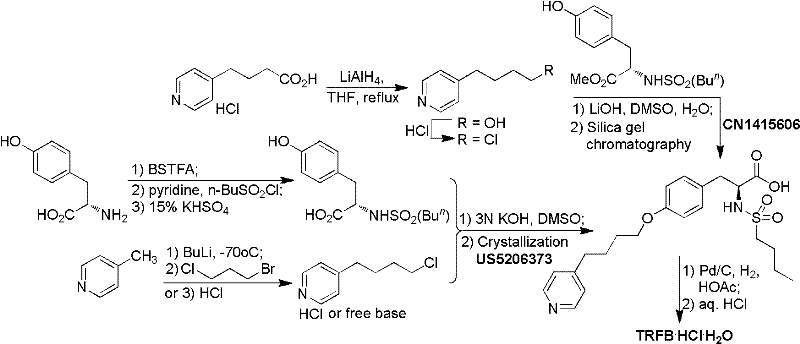
Another significant bottleneck in conventional synthesis is exemplified by Patent US5206373 and CN1415606, which utilize n-Butyl Lithium for the preparation of key halide intermediates. This reagent demands strictly anhydrous conditions and cryogenic temperatures around -70°C, creating substantial energy costs and engineering challenges for commercial scale-up. Additionally, the Mitsunobu reaction approach detailed in Patent CN1040534, while chemically elegant, suffers from poor atom economy. It requires stoichiometric amounts of triphenylphosphine and diisopropyl azodicarboxylate (DIAD), generating massive quantities of triphenylphosphine oxide as a byproduct. This waste stream complicates downstream processing, requiring extensive washing and purification to meet pharmaceutical purity standards, thereby inflating production costs and extending lead times for high-purity pharmaceutical intermediates.
The Novel Approach
In stark contrast to these legacy methods, the technology outlined in CN102285912A presents a streamlined, two-step strategy that fundamentally reimagines the construction of the ether bond. The process initiates with the mesylation of 4-pyridine butanol using methanesulfonyl chloride (MsCl) under mild alkaline conditions, typically between -10°C and 30°C. This activation step converts the hydroxyl group into a highly reactive mesylate leaving group without the need for hazardous lithiation or reduction chemistry. Crucially, the resulting methylsulfonyl pyridine butanol intermediate does not require isolation or rigorous purification; it can be telescoped directly into the subsequent condensation reaction. This "one-pot" capability significantly reduces solvent consumption and processing time, addressing the pain points of both procurement managers and supply chain heads who prioritize operational efficiency.
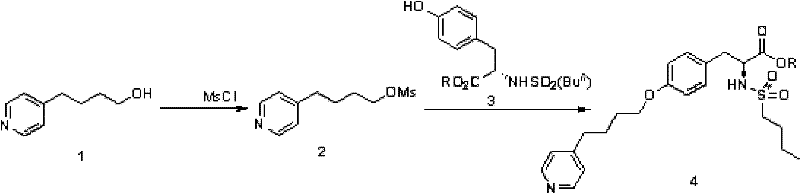
The second phase of this novel approach involves the condensation of the crude mesylate with N-n-butylsulfonyl L-tyrosine alkyl ester in the presence of a phase transfer catalyst. By employing common inorganic bases such as potassium carbonate or sodium hydride in solvents like acetonitrile or DMF, the reaction proceeds smoothly at moderate temperatures ranging from 50°C to 80°C. This eliminates the need for cryogenic cooling and allows for the use of standard stainless steel reactors found in most fine chemical facilities. The integration of phase transfer catalysis ensures efficient mixing of reagents across phases, driving the reaction to completion with high conversion rates. This methodological shift not only enhances the safety profile of the manufacturing process by avoiding dangerous reagents like n-BuLi and LiAlH4 but also drastically simplifies the workup procedure, making it ideally suited for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Mesylate Activation and Nucleophilic Substitution
The core chemical innovation of this patent lies in the strategic selection of the mesylate group as the electrophilic partner for the etherification reaction. Mechanistically, the reaction of 4-pyridine butanol with methanesulfonyl chloride proceeds via a nucleophilic attack of the alcohol oxygen on the sulfur atom of the sulfonyl chloride, facilitated by an organic base such as triethylamine or diisopropylethylamine. This base serves a dual purpose: it scavenges the hydrochloric acid byproduct to drive the equilibrium forward and activates the alcohol by deprotonation. The resulting sulfonate ester is an exceptional leaving group, superior to halides in many contexts due to its stability and reactivity profile. This stability allows the intermediate to be handled without immediate degradation, providing a wider operational window for the subsequent coupling step compared to more labile alkyl halides generated in other routes.
In the condensation step, the phenolic oxygen of the N-n-butylsulfonyl L-tyrosine alkyl ester acts as the nucleophile. Under basic conditions, the phenol is deprotonated to form a phenoxide anion, which then attacks the terminal carbon of the butyl chain in the mesylate intermediate via an SN2 mechanism. The presence of a phase transfer catalyst, such as tetrabutylammonium bromide (TBAB), is critical here; it solubilizes the inorganic base and the phenoxide anion in the organic phase, dramatically increasing the reaction rate and yield. This mechanistic pathway effectively suppresses side reactions such as elimination or racemization of the chiral center on the tyrosine moiety, ensuring the stereochemical integrity of the final product. By controlling the stoichiometry and temperature, manufacturers can minimize the formation of impurities, thereby achieving the stringent purity specifications required for regulatory approval.
How to Synthesize N-n-butylsulfonyl-O-(4-(4-pyridyl)-butyl)-L-tyrosine Alkyl Ester Efficiently
The implementation of this synthesis route requires careful attention to reagent quality and reaction parameters to maximize yield and minimize impurities. The process begins with the activation of the alcohol component, followed immediately by the coupling with the amino acid derivative. This telescoped approach reduces the number of unit operations and solvent exchanges, which is a key factor in reducing the overall cost of goods sold (COGS). Operators should maintain strict temperature control during the mesylation step to prevent over-reaction or decomposition, while the condensation step benefits from elevated temperatures to overcome activation energy barriers. The following guide outlines the standardized protocol derived from the patent examples, serving as a foundational reference for process development teams aiming to adopt this technology.
- React 4-pyridine butanol with methanesulfonyl chloride (MsCl) in an organic solvent (e.g., DCM) using an organic base like triethylamine at 0°C to 20°C to form methylsulfonyl pyridine butanol.
- Without purifying the intermediate, react the crude methylsulfonyl pyridine butanol with N-n-butylsulfonyl L-tyrosine alkyl ester in a solvent like acetonitrile or DMF.
- Add an inorganic base (e.g., K2CO3) and a phase transfer catalyst (e.g., TBAB), then heat the mixture to 50°C to 80°C for 4-12 hours to obtain the final ester product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the CN102285912A methodology offers tangible benefits that extend beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the supply chain for raw materials. By eliminating the need for specialized, hazardous reagents like n-Butyl Lithium, Lithium Aluminum Hydride, or large quantities of Triphenylphosphine, manufacturers can source common, commodity-grade chemicals that are readily available from multiple vendors. This diversification of the supply base mitigates the risk of shortages and price volatility associated with niche reagents. Furthermore, the removal of column chromatography purification steps from the intermediate stage significantly reduces the consumption of silica gel and solvents, leading to substantial cost savings in waste disposal and material procurement.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the reduction in unit operations and the avoidance of expensive purification technologies. Traditional routes often require cryogenic reactors and specialized handling for pyrophoric reagents, which incur high capital and operational expenditures. In contrast, this novel method operates at near-ambient or moderately elevated temperatures using standard glass-lined or stainless steel equipment. The ability to telescope the intermediate without purification means that solvents do not need to be completely removed and exchanged between steps, saving significant energy and time. Additionally, the higher overall yield resulting from fewer steps directly lowers the cost per kilogram of the final intermediate, providing a competitive edge in pricing negotiations for API contracts.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness of the reaction conditions. Processes that rely on moisture-sensitive reagents or extreme temperatures are prone to batch failures and delays, which can disrupt the entire production schedule. The mild conditions described in this patent—using stable bases like potassium carbonate and solvents like acetonitrile—ensure high reproducibility and batch-to-batch consistency. This reliability allows for more accurate forecasting and inventory management, reducing the need for excessive safety stock. Moreover, the reduced environmental footprint of the process, characterized by less hazardous waste generation, simplifies regulatory compliance and permits faster throughput in facilities with strict environmental controls.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to commercial production often reveals hidden bottlenecks, particularly regarding heat transfer and mixing efficiency. The exothermic nature of mesylation and the heterogeneous nature of the condensation reaction are well-managed in this protocol through the use of phase transfer catalysts and controlled addition rates. This makes the transition from pilot plant to multi-ton production seamless. From an environmental perspective, the avoidance of heavy metal catalysts and the reduction of phosphine oxide waste align with green chemistry principles. This not only reduces the burden on wastewater treatment facilities but also enhances the sustainability profile of the final pharmaceutical product, a factor increasingly valued by end-users and regulatory bodies alike.
Frequently Asked Questions (FAQ)
Understanding the technical specifics of this synthesis route is vital for stakeholders evaluating its potential for integration into existing manufacturing lines. The following questions address common concerns regarding process safety, purity control, and regulatory alignment. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear picture of what partners can expect when adopting this technology. By clarifying these points, we aim to facilitate informed decision-making for technical teams assessing the feasibility of this route for their specific production needs.
Q: What are the primary advantages of the mesylate route over the Mitsunobu reaction for Tirofiban intermediates?
A: The mesylate route described in CN102285912A avoids the use of expensive and hazardous reagents like triphenylphosphine and DIAD required in the Mitsunobu reaction. Furthermore, it eliminates the generation of large quantities of triphenylphosphine oxide waste, significantly simplifying downstream purification and reducing environmental impact.
Q: Is purification of the methylsulfonyl pyridine butanol intermediate required before the condensation step?
A: No, one of the key innovations of this patent is that the intermediate methylsulfonyl pyridine butanol does not require purification after simple treatment. It can be directly used in the next condensation reaction, which streamlines the process and improves overall yield.
Q: How does this method improve industrial scalability compared to prior art using n-Butyl Lithium?
A: Prior art methods often rely on n-Butyl Lithium at cryogenic temperatures (-70°C), which poses significant safety risks and operational costs for large-scale production. This new method operates at mild temperatures ranging from -10°C to 100°C using standard bases like potassium carbonate, making it far safer and more suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tirofiban Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex cardiovascular drugs relies on a foundation of robust and scalable intermediate synthesis. Our technical team has extensively analyzed the methodology presented in CN102285912A and possesses the expertise to implement this route with precision. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with unwavering consistency. Our facilities are equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of N-n-butylsulfonyl-O-(4-(4-pyridyl)-butyl)-L-tyrosine alkyl ester meets the highest industry standards for chirality and chemical purity.
We invite you to collaborate with us to leverage this advanced synthesis technology for your upcoming projects. Our commitment to innovation allows us to offer a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this optimized route can improve your bottom line. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. By partnering with NINGBO INNO PHARMCHEM, you secure not just a supplier, but a strategic ally dedicated to advancing your pharmaceutical pipeline through superior process chemistry and reliable delivery.
