Scalable Synthesis of Roflumilast Intermediate via Regioselective Protection Strategy
Introduction to Advanced Roflumilast Intermediate Manufacturing
The pharmaceutical landscape for Chronic Obstructive Pulmonary Disease (COPD) treatments relies heavily on the consistent supply of high-quality active pharmaceutical ingredients, with Roflumilast standing as a cornerstone therapy. The efficient production of its key precursor, 3-cyclopropylmethoxy-4-difluoromethoxybenzoic acid, remains a critical challenge for generic manufacturers seeking market entry. Patent CN102503815A discloses a groundbreaking preparation method that fundamentally restructures the synthetic pathway to overcome historical inefficiencies. This innovation shifts the paradigm from low-yield, purification-intensive batch processes to a streamlined, high-yield sequence suitable for modern industrial demands. By strategically employing a temporary benzyl protecting group, the process achieves exceptional regioselectivity that was previously unattainable in direct functionalization attempts. The result is a robust manufacturing protocol that delivers stable quality without the need for complex chromatographic separations. This technical breakthrough not only enhances chemical efficiency but also aligns perfectly with the rigorous purity standards required by global regulatory bodies for respiratory medications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
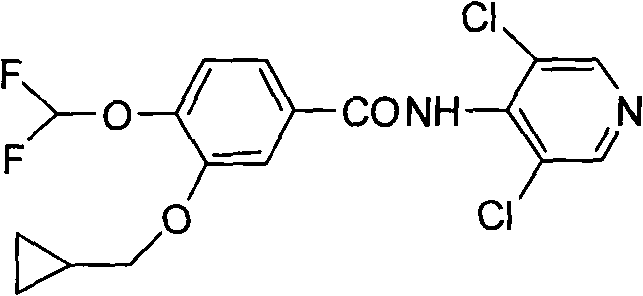
Historically, the synthesis of this critical benzoic acid derivative has been plagued by significant chemical and operational hurdles that hinder cost-effective manufacturing. Traditional routes typically initiate with the direct difluoromethylation of 3,4-dihydroxybenzaldehyde, a step notorious for its poor reaction preference and lack of regiocontrol. This lack of selectivity generates a complex mixture of isomers, necessitating laborious and expensive column chromatography for purification, which drastically reduces the isolated yield to a mere 25% to 30%. Furthermore, subsequent etherification steps in conventional protocols often require excessive amounts of alkylating agents, such as three equivalents of cyclopropylmethyl bromide, and prolonged reaction times extending up to 14 hours. The final oxidation stage in older methods frequently employs sodium chlorite and sulfamic acid, creating violent reaction conditions that are difficult to control safely on a large scale. These cumulative inefficiencies render traditional pathways economically unviable for competitive commercial production, creating a bottleneck for supply chain reliability.
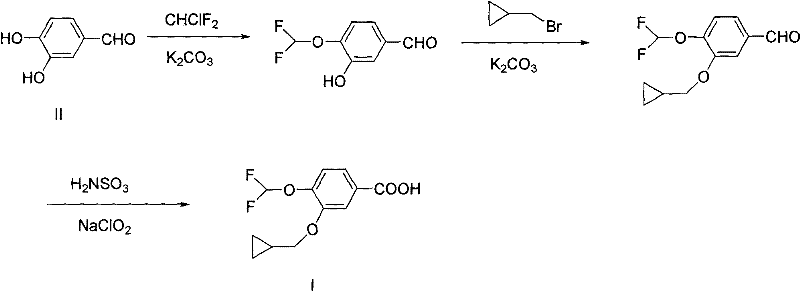
The Novel Approach
In stark contrast, the inventive methodology presented in the patent data introduces a sophisticated five-step sequence that elegantly resolves these regioselectivity and safety issues. The core innovation lies in the initial protection of the 4-hydroxyl group using a benzyl moiety, which effectively masks this position and directs subsequent reactions exclusively to the 3-hydroxyl group. This strategic maneuver allows for the clean installation of the cyclopropylmethoxy group with near-quantitative conversion, eliminating the formation of unwanted isomers. Following this, a mild catalytic hydrogenolysis step removes the protecting group under gentle conditions, restoring the phenolic functionality without degrading the sensitive cyclopropyl ring. The subsequent difluoromethylation proceeds smoothly on the unprotected phenol, and the final oxidation utilizes hydrogen peroxide, a greener and safer oxidant compared to chlorite salts. This holistic approach boosts the overall yield to an impressive 58.4% while completely removing the need for column chromatography, thereby simplifying post-treatment and waste management significantly.
Mechanistic Insights into Regioselective Protection and Oxidation
The success of this synthetic route is anchored in the precise manipulation of electronic and steric factors through the benzyl protection strategy. In the first step, the reaction between 3,4-dihydroxybenzaldehyde and a benzyl halide under alkaline conditions exploits the subtle differences in acidity and nucleophilicity between the two hydroxyl groups, or simply relies on stoichiometric control to favor mono-protection at the 4-position. This protection is crucial because the 4-hydroxyl group is generally more reactive towards electrophilic attack; by blocking it, the synthesis forces the cyclopropylmethyl bromide to react selectively at the 3-position in the subsequent step. The use of potassium carbonate or similar bases facilitates the formation of the phenoxide ion, which acts as a potent nucleophile for the SN2 displacement of the halide. This mechanistic control ensures that the molecular architecture is built in the correct orientation from the outset, preventing the formation of regioisomers that would otherwise contaminate the final product.
Furthermore, the choice of oxidation reagents in the final step represents a significant advancement in process safety and impurity control. Traditional chlorite oxidations generate chlorine dioxide gas and other hazardous byproducts, requiring specialized scrubbing systems and posing explosion risks. The patented method utilizes hydrogen peroxide in an alkaline medium, likely proceeding through a peroxyhemiacetal intermediate that rearranges to the carboxylic acid. This mechanism is inherently cleaner, producing water as the primary byproduct, which simplifies downstream processing and reduces the environmental footprint. The mild conditions of the hydrogen peroxide oxidation also preserve the integrity of the difluoromethoxy and cyclopropylmethoxy groups, which might be susceptible to degradation under harsher acidic or strongly oxidative conditions found in older protocols. This careful balance of reactivity ensures a high-purity profile essential for pharmaceutical applications.
How to Synthesize 3-Cyclopropylmethoxy-4-difluoromethoxybenzoic Acid Efficiently
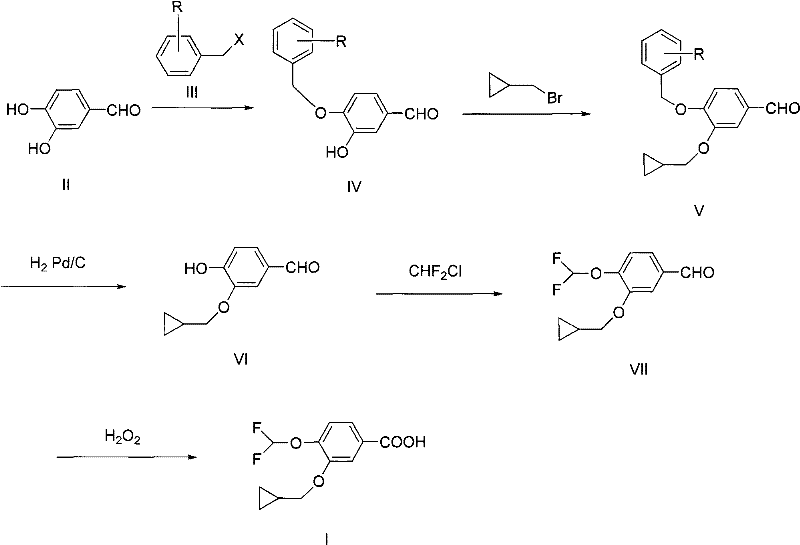
The execution of this synthesis requires strict adherence to the optimized reaction parameters outlined in the patent to maximize yield and purity. The process begins with the protection step in acetone or similar solvents, followed by filtration and extraction to isolate the benzyl-protected intermediate. Subsequent etherification utilizes cyclopropylmethyl bromide in ketone solvents like butanone, ensuring rapid conversion. The catalytic hydrogenolysis is performed at ambient temperature and pressure using palladium on carbon, a standard and easily scalable operation. The difluoromethylation step involves feeding difluorochloromethane gas into the reaction mixture, requiring appropriate gas handling infrastructure. Finally, the oxidation is conducted by adding hydrogen peroxide to an alkaline solution of the aldehyde, followed by acidification to precipitate the product. For the complete standardized operating procedures and specific molar ratios, please refer to the detailed guide below.
- Protect the 4-hydroxyl group of 3,4-dihydroxybenzaldehyde using a benzyl halide under alkaline conditions to form the protected intermediate.
- Perform regioselective etherification at the 3-position using cyclopropylmethyl bromide to install the cyclopropylmethoxy group.
- Execute catalytic hydrogenolysis to remove the benzyl protecting group, yielding 4-hydroxy-3-cyclopropylmethoxybenzaldehyde.
- Etherify the remaining 4-hydroxyl group with difluorochloromethane to introduce the difluoromethoxy moiety.
- Oxidize the aldehyde functionality to the carboxylic acid using hydrogen peroxide under alkaline conditions to obtain the final target acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route translates directly into enhanced operational stability and reduced total cost of ownership. The elimination of column chromatography is perhaps the most significant economic driver, as chromatographic purification is notoriously expensive, solvent-intensive, and difficult to scale beyond kilogram quantities. By replacing this batch-intensive unit operation with simple crystallization and filtration steps, the process drastically reduces solvent consumption and waste disposal costs. Additionally, the use of hydrogen peroxide instead of sodium chlorite lowers raw material costs and mitigates the need for expensive safety engineering controls associated with hazardous oxidants. The higher overall yield means that less starting material is required to produce the same amount of final product, further driving down the cost of goods sold. These factors combine to create a supply chain that is more resilient to raw material price fluctuations and capable of meeting large-volume demands without exponential cost increases.
- Cost Reduction in Manufacturing: The removal of column chromatography and the reduction of reaction times significantly lower processing costs. The use of cheaper, safer oxidants like hydrogen peroxide replaces expensive and hazardous chlorite salts, leading to substantial savings in both raw materials and waste treatment. The high yield of each step minimizes material loss, ensuring that the theoretical maximum output is approached in practice.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as benzyl chloride, cyclopropylmethyl bromide, and hydrogen peroxide ensures a stable supply of raw materials, reducing the risk of shortages associated with specialty reagents. The robustness of the reaction conditions means that production schedules are less likely to be disrupted by failed batches or difficult purifications, guaranteeing consistent delivery timelines for downstream API manufacturers.
- Scalability and Environmental Compliance: The process is explicitly designed for industrial suitability, avoiding operations that are inherently difficult to scale, such as flash chromatography. The greener oxidation step reduces the generation of toxic halogenated waste, simplifying compliance with increasingly stringent environmental regulations. This ease of scale-up allows manufacturers to rapidly increase production capacity from pilot plants to multi-ton commercial facilities without extensive re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation. Understanding these details is crucial for technical teams evaluating the feasibility of technology transfer or process validation.
Q: Why is the benzyl protection strategy superior to direct difluoromethylation?
A: Direct difluoromethylation of 3,4-dihydroxybenzaldehyde suffers from poor regioselectivity, yielding a mixture of isomers that requires difficult column chromatography separation with low yields (25-30%). The benzyl protection strategy ensures exclusive reaction at the desired position, eliminating purification bottlenecks.
Q: What are the safety advantages of the oxidation step in this new process?
A: Conventional methods often utilize sodium chlorite and sulfamic acid, which can lead to violent and hard-to-control reactions. The novel process employs hydrogen peroxide oxidation, which offers milder reaction conditions, easier temperature control, and significantly reduced safety risks for large-scale manufacturing.
Q: Is this synthesis route suitable for multi-ton commercial production?
A: Yes, the route is specifically designed for industrial scalability. It avoids complex operations like column chromatography, utilizes common solvents and reagents, and achieves a high overall yield of approximately 58.4%, making it economically viable for mass production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Cyclopropylmethoxy-4-difluoromethoxybenzoic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and proven manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising yields reported in patent literature are realized in our reactors. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify the identity and quality of every batch. Our commitment to excellence means that we do not just supply chemicals; we deliver validated solutions that integrate seamlessly into your API synthesis workflows, minimizing your regulatory risk and accelerating your time to market.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain specific insights into how switching to our manufactured intermediate impacts your overall production economics. We encourage potential partners to contact us for specific COA data and route feasibility assessments tailored to your unique volume requirements. Let us demonstrate how our technical superiority translates into tangible business value for your organization.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
