Advanced Synthesis of 1-R-4-Amino-1,2,3,4-Tetrahydroquinoline for Commercial API Manufacturing
The pharmaceutical industry constantly seeks robust synthetic pathways for complex heterocyclic scaffolds, particularly those serving as critical building blocks for bioactive molecules. Patent CN101429160A introduces a transformative methodology for synthesizing 1-R-4-amino-1,2,3,4-tetrahydroquinoline derivatives, addressing longstanding inefficiencies in prior art. This innovation pivots away from cumbersome cyclization strategies, instead leveraging 4-hydroxyquinoline as a versatile, industrially available starting material. By employing a sequence of N-alkylation, selective reduction, azidation via diphenylphosphoryl azide (DPPA), and final amine formation, the process achieves high purity and operational simplicity. For R&D directors and procurement specialists, this represents a significant opportunity to optimize the supply chain for quinoline-based active pharmaceutical ingredients (APIs). The following analysis details how this technical breakthrough translates into tangible commercial advantages, ensuring reliable supply and cost efficiency for global pharmaceutical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1-alkyl-4-amino-1,2,3,4-tetrahydroquinolines has relied heavily on intramolecular Friedel-Crafts acylation or alkylation strategies. As illustrated in the traditional pathway, this typically involves reacting mono-substituted anilines with acrylates or nitriles, followed by a cyclization step mediated by polyphosphoric acid. This conventional approach presents severe bottlenecks for commercial scale-up. The use of polyphosphoric acid creates a highly viscous reaction medium that impedes heat transfer and mixing, necessitating rigorous temperature control to prevent thermal runaways. Furthermore, the post-reaction workup is notoriously difficult; quenching large volumes of polyphosphoric acid generates substantial acidic waste streams, complicating environmental compliance and increasing disposal costs. Additionally, the solubility profiles of the intermediate ketones often lead to poor extraction efficiencies, resulting in lower overall yields and inconsistent product quality that fails to meet the stringent purity specifications required for modern drug development.
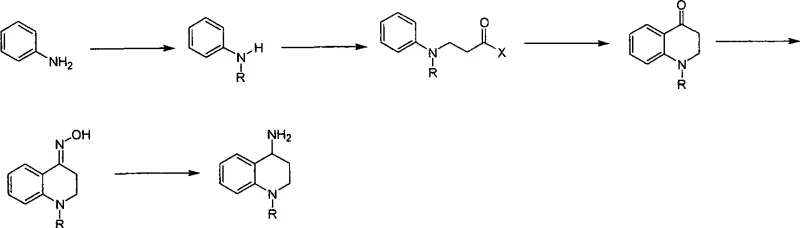
The Novel Approach
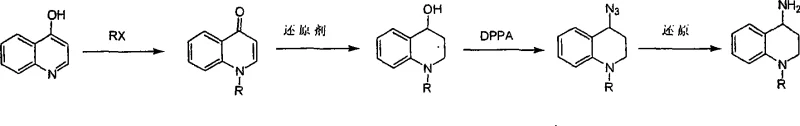
In stark contrast, the novel methodology disclosed in the patent utilizes 4-hydroxyquinoline as the foundational feedstock, bypassing the problematic cyclization step entirely. The process initiates with a straightforward N-alkylation using alkyl halides or sulfonates to generate the quinolinone intermediate. This is followed by a controlled reduction to the alcohol, conversion to an azide using DPPA, and final reduction to the target amine. This linear progression eliminates the need for corrosive polyphosphoric acid and high-temperature cyclization conditions. The reaction conditions are significantly milder, often proceeding at room temperature or standard reflux in common solvents like acetonitrile, methanol, or ethyl acetate. This shift not only simplifies the operational workflow but also dramatically improves the impurity profile, as the stepwise nature of the synthesis allows for intermediate purification. For a reliable pharmaceutical intermediate supplier, this route offers a predictable and scalable alternative that aligns with green chemistry principles by reducing hazardous reagent usage.
Mechanistic Insights into N-Alkylation and Azide Substitution
The core of this synthetic strategy lies in the precise manipulation of the quinoline nitrogen and the C4 position. The initial N-alkylation step exploits the nucleophilicity of the 4-hydroxyquinoline nitrogen atom. In the presence of a base such as potassium carbonate, the nitrogen attacks the electrophilic carbon of the alkyl halide (RX), forming a stable N-alkylated quinolin-4(1H)-one. This step is crucial as it installs the diverse 'R' group (methyl, ethyl, benzyl, etc.) early in the sequence, allowing for late-stage diversification without altering the core reaction conditions. Following this, the carbonyl group at the C4 position is selectively reduced to a hydroxyl group using hydride sources like sodium borohydride or lithium aluminium hydride. This reduction is highly chemoselective, preserving the aromatic integrity of the benzene ring while converting the ketone into a secondary alcohol, which serves as the handle for the subsequent functionalization.
The transformation of the C4-hydroxyl group into an amine is achieved through a clever two-step sequence involving azidation and reduction. The reaction of the alcohol with diphenylphosphoryl azide (DPPA) in the presence of a base like DBU facilitates the substitution of the hydroxyl group with an azide moiety. While DPPA is traditionally associated with the Curtius rearrangement for converting carboxylic acids to amines, in this context, it acts as an efficient activator for nucleophilic substitution on the activated alcohol species. The resulting 4-azido-1,2,3,4-tetrahydroquinoline is then subjected to reduction. This can be accomplished via catalytic hydrogenation using palladium on carbon (Pd/C) or via the Staudinger reduction using triphenylphosphine. Both methods effectively cleave the azide bond to yield the primary amine with high fidelity, minimizing the formation of over-reduced byproducts or ring-opened impurities that often plague harsher amination protocols.
How to Synthesize 1-R-4-Amino-1,2,3,4-Tetrahydroquinoline Efficiently
Implementing this synthesis requires careful attention to solvent selection and stoichiometry to maximize yield and safety. The process begins with the N-alkylation of 4-hydroxyquinoline, where maintaining anhydrous conditions can enhance the reaction rate, although the patent demonstrates success in various polar aprotic and protic solvents. The subsequent reduction steps must be monitored to ensure complete conversion of the ketone and azide intermediates, as residual starting materials can complicate downstream purification. The use of column chromatography is described in the examples for laboratory-scale purification, but for industrial applications, crystallization or extractive workups would be optimized to reduce solvent consumption. The detailed standardized synthesis steps, including specific molar ratios and temperature profiles for each stage, are outlined below to guide process chemists in replicating this high-efficiency route.
- React 4-hydroxyquinoline with alkyl halides or sulfonates in solvent to form the N-alkylated quinolinone ketone.
- Reduce the resulting ketone using sodium borohydride or lithium aluminium hydride to obtain the corresponding 4-hydroxy tetrahydroquinoline alcohol.
- Convert the alcohol to an azide intermediate using diphenylphosphoryl azide (DPPA) in anhydrous tetrahydrofuran with a base.
- Perform final reduction of the azide to the primary amine using hydrogenation with Pd/C or chemical reduction with triphenylphosphine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers profound strategic benefits beyond mere technical feasibility. The elimination of polyphosphoric acid removes a major logistical and safety hazard from the manufacturing process, significantly reducing the burden on waste treatment facilities and lowering the total cost of ownership for the production line. Furthermore, the reliance on 4-hydroxyquinoline as a starting material leverages a commodity chemical that is readily available from multiple global suppliers, thereby mitigating the risk of raw material shortages that often plague specialized aniline derivatives. The modularity of the synthesis allows for the rapid production of various analogues by simply changing the alkylating agent, providing supply chain agility to meet fluctuating market demands for different API variants without requiring extensive process re-validation.
- Cost Reduction in Manufacturing: The new process drastically simplifies the operational workflow by removing the need for high-temperature cyclization and the associated energy costs. By avoiding the use of expensive and hazardous polyphosphoric acid, the facility saves on both reagent procurement and the significant costs associated with neutralizing and disposing of acidic waste. The use of common reducing agents like sodium borohydride and catalysts like Pd/C, which can often be recovered and recycled, further drives down the variable cost per kilogram. Additionally, the improved yield and purity reduce the need for extensive reprocessing or recycling of off-spec material, leading to substantial overall cost savings in pharmaceutical intermediate manufacturing.
- Enhanced Supply Chain Reliability: Sourcing 4-hydroxyquinoline is far more stable than sourcing complex substituted anilines required for the traditional Friedel-Crafts route. This shift ensures a more resilient supply chain, less susceptible to disruptions from niche chemical suppliers. The reaction conditions, which primarily operate at room temperature or standard reflux, reduce the dependency on specialized high-pressure or high-temperature reactor infrastructure. This compatibility with standard glass-lined or stainless steel reactors means that the process can be easily transferred between different manufacturing sites or contract manufacturing organizations (CMOs) without significant capital investment, ensuring continuous supply continuity for critical drug programs.
- Scalability and Environmental Compliance: The absence of viscous polyphosphoric acid makes the process inherently more scalable, as heat and mass transfer issues are minimized in large-scale reactors. The solvents employed, such as ethyl acetate, methanol, and ethanol, are widely accepted in green chemistry initiatives and are easier to recover and recycle compared to chlorinated solvents often used in older methodologies. The reduction in hazardous waste generation aligns with increasingly strict environmental regulations, reducing the regulatory burden on the manufacturing site. This environmental compliance not only safeguards the company's reputation but also future-proofs the manufacturing process against tightening global emissions and waste disposal standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation. Understanding these nuances is critical for technical teams evaluating the feasibility of adopting this route for their specific pipeline projects. The answers highlight the robustness of the chemistry and its alignment with modern manufacturing standards.
Q: Why is the 4-hydroxyquinoline route superior to the traditional Friedel-Crafts method?
A: The traditional Friedel-Crafts cyclization requires polyphosphoric acid, high temperatures, and generates significant waste, making scale-up difficult. The new route uses mild N-alkylation and reduction steps that are easier to control and purify.
Q: What reducing agents are suitable for the final step of this synthesis?
A: The patent specifies two effective methods: catalytic hydrogenation using 10% Pd/C in ethyl acetate at room temperature, or chemical reduction using triphenylphosphine in THF/water, offering flexibility based on equipment availability.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the process avoids viscous polyphosphoric acid and uses common solvents like methanol, ethanol, and ethyl acetate. The reaction conditions range from room temperature to reflux, which are standard for large-scale reactor operations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-R-4-Amino-1,2,3,4-Tetrahydroquinoline Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of life-saving medications. Our technical team has thoroughly analyzed the potential of the 4-hydroxyquinoline-based route and is fully prepared to support your project from gram-scale development to commercial production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab to plant is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 1-R-4-amino-1,2,3,4-tetrahydroquinoline delivered meets the highest international standards for pharmaceutical applications.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next-generation therapeutics. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you optimize your supply chain and accelerate your time to market with our reliable and cost-effective manufacturing solutions.
