Scalable Manufacturing of AZD6738 Intermediates Using Novel Asymmetric Oxidation Technology
Introduction to Advanced AZD6738 Manufacturing Technologies
The pharmaceutical landscape for oncology treatments is continuously evolving, with AZD6738, also known as Ceralasertib, standing out as a potent ATR kinase inhibitor currently in clinical development for solid tumors and hematological cancers. The efficient production of this complex molecule relies heavily on the robustness of its synthetic route, particularly the establishment of its critical chiral centers. Patent CN111646985A introduces a groundbreaking methodology that shifts away from traditional enzymatic or harsh chemical oxidation processes towards a more economically viable and scalable vanadium-catalyzed asymmetric oxidation system. This technical insight report delves into the specifics of this innovation, highlighting how it addresses the longstanding challenges of cost, safety, and scalability in the manufacturing of high-purity pharmaceutical intermediates.
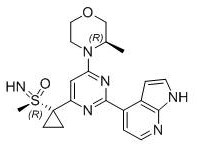
By leveraging a chiral ligand-induced asymmetric oxidation method, this new protocol utilizes inexpensive oxidants like hydrogen peroxide to transform methyl sulfide precursors into the requisite chiral sulfoxide intermediates with high efficiency. Unlike previous iterations that depended on costly biocatalysts or suffered from environmental drawbacks associated with batch processing, this approach integrates green chemistry principles with industrial practicality. For R&D directors and procurement specialists, understanding the nuances of this pathway is essential for evaluating supply chain resilience and potential cost reductions in API manufacturing. The patent further elucidates the application of flow chemistry techniques, which significantly enhance the total synthesis yield compared to conventional batch reactions, marking a substantial leap forward in process intensification for heterocyclic antitumor drugs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of AZD6738 and similar sulfoximine-containing molecules has been plagued by significant technical and economic hurdles. Prior art, such as patents WO2011154737A1 and US2011/306613, predominantly describes batch reaction protocols that are inherently less efficient and more resource-intensive. A major bottleneck in these traditional routes is the introduction of the chiral sulfoxide moiety, often achieved through enzymatic catalysis. While enzymatic methods offer high stereoselectivity, they impose a heavy financial burden due to the high cost of enzymes and the requirement for specialized bioreactors, which complicates the capital expenditure profile for manufacturing facilities. Furthermore, conventional batch processes often struggle with heat and mass transfer limitations, leading to inconsistent reaction profiles and the formation of impurities that require rigorous and costly purification steps, such as preparative chromatography, to meet stringent pharmaceutical standards.
The Novel Approach
The methodology disclosed in CN111646985A presents a paradigm shift by replacing biological catalysts with a chemically robust vanadium-salt and chiral-ligand system. This novel approach catalyzes the asymmetric oxidation of sulfide compounds using affordable oxidants like hydrogen peroxide, effectively bypassing the economic constraints of enzyme procurement. The process is designed to be highly efficient, achieving transformation rates comparable to the original processes but with a drastically simplified purification workflow that eliminates the need for complex chromatographic separations. Additionally, the integration of continuous flow chemistry for multiple steps in the synthesis sequence allows for precise control over reaction parameters such as temperature and residence time. This technological upgrade not only accelerates reaction kinetics but also improves safety profiles by minimizing the inventory of hazardous intermediates, thereby offering a more sustainable and economically attractive route for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Vanadium-Catalyzed Asymmetric Oxidation
At the heart of this synthetic innovation lies the vanadium-catalyzed asymmetric oxidation step, which is responsible for establishing the critical stereochemistry of the sulfoxide group. The mechanism involves the coordination of a vanadium salt, specifically vanadyl acetylacetonate (VO(acac)2), with a bespoke chiral organic ligand derived from the condensation of a nitrobenzaldehyde derivative and an amino alcohol. This chiral complex creates a sterically constrained environment that directs the attack of the oxidant, hydrogen peroxide, onto the sulfur atom of the thioether precursor. The result is the highly selective formation of the desired (S)-sulfoxide enantiomer, which is a prerequisite for the biological activity of the final drug molecule. The use of hydrogen peroxide as the terminal oxidant is particularly advantageous from a green chemistry perspective, as its only byproduct is water, minimizing the generation of toxic waste streams associated with traditional stoichiometric oxidants.
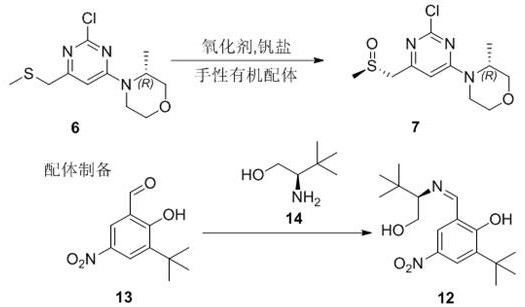
Furthermore, the patent details the optimization of reaction conditions to maximize diastereomeric ratio (dr) and chemical purity. By fine-tuning the molar ratios of the vanadium catalyst and the chiral ligand relative to the substrate, the process achieves a dr value of up to 10:1, which can be further enriched to high purity through simple recrystallization or silica gel chromatography. This level of control is crucial for ensuring the consistency of the intermediate quality, directly impacting the downstream synthesis of the final API. The mechanistic robustness of this system allows it to tolerate various substrates and scales, making it a versatile tool for medicinal chemists and process engineers alike. The ability to generate the chiral ligand in situ or use it as a pre-formed entity adds another layer of operational flexibility, allowing manufacturers to adapt the process based on their specific supply chain capabilities and equipment configurations without compromising the stereochemical integrity of the product.
How to Synthesize AZD6738 Intermediates Efficiently
The comprehensive synthetic route outlined in the patent provides a clear roadmap for producing AZD6738 from readily available starting materials. The process begins with the N-alkylation of a chiral morpholine derivative, followed by a series of functional group transformations including reduction, mesylation, and thiolation to construct the pyrimidine scaffold. The pivotal asymmetric oxidation step then installs the chiral sulfoxide, followed by imination to form the sulfoximine. Subsequent cyclopropanation and a final Suzuki coupling reaction complete the assembly of the target molecule. This modular approach allows for the isolation and characterization of key intermediates, ensuring quality control at every stage of the synthesis. The detailed procedural steps provided in the patent serve as a valuable reference for laboratories aiming to replicate or adapt this technology for their own production needs.
- Perform N-alkylation of (R)-2-methylmorpholine with methyl 2,6-dichloropyrimidine-4-carboxylate to form the pyrimidine core.
- Reduce the ester group to an alcohol, followed by mesylation and nucleophilic substitution with sodium methanethiolate to introduce the thioether moiety.
- Execute the key asymmetric oxidation using a vanadium salt and chiral ligand with hydrogen peroxide to generate the chiral sulfoxide intermediate.
- Convert the sulfoxide to sulfoximine, perform cyclopropanation, and finalize the synthesis via Suzuki coupling with the pyrrolopyridine boronic acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route offers tangible strategic benefits that extend beyond mere technical feasibility. The primary advantage lies in the substantial cost reduction potential driven by the replacement of expensive biocatalysts with inexpensive chemical reagents. By eliminating the dependency on proprietary enzymes and the specialized infrastructure required to support them, manufacturers can significantly lower their variable production costs. This shift also mitigates supply chain risks associated with the sourcing of biological materials, which can be subject to availability fluctuations and strict storage requirements. The use of common chemical oxidants like hydrogen peroxide ensures a stable and reliable supply of critical reagents, enhancing the overall resilience of the manufacturing operation against market volatility.
- Cost Reduction in Manufacturing: The transition from enzymatic to chemical catalysis fundamentally alters the cost structure of AZD6738 production. Enzymes are often high-value consumables that contribute significantly to the cost of goods sold (COGS). By substituting this with a vanadium-based catalytic system, the process leverages abundant and low-cost transition metals. Additionally, the simplification of the purification process, which no longer requires preparative chromatography for the chiral sulfoxide intermediate, reduces solvent consumption and processing time. These efficiencies translate directly into lower operational expenditures, allowing for more competitive pricing strategies in the global pharmaceutical market while maintaining healthy profit margins for suppliers.
- Enhanced Supply Chain Reliability: Supply chain continuity is paramount in the pharmaceutical industry, where delays can have cascading effects on clinical trial timelines and patient access. This new method enhances reliability by utilizing a robust chemical process that is less sensitive to environmental variations compared to biological systems. The compatibility of the synthesis with continuous flow chemistry further strengthens supply security by enabling rapid scale-up and flexible production scheduling. Flow reactors can be easily paralleled or scaled in number to meet increasing demand without the need for massive single-vessel infrastructure investments. This modularity ensures that suppliers can respond swiftly to changes in order volumes, providing a dependable source of high-purity pharmaceutical intermediates for downstream API manufacturers.
- Scalability and Environmental Compliance: As regulatory pressures regarding environmental sustainability intensify, the green chemistry attributes of this process become a significant commercial asset. The use of hydrogen peroxide generates water as a benign byproduct, reducing the burden on waste treatment facilities and lowering disposal costs. Furthermore, the implementation of flow chemistry improves safety by minimizing the hold-up volume of reactive intermediates, thereby reducing the risk of thermal runaways and accidents. This enhanced safety profile facilitates easier regulatory approval for manufacturing sites and reduces insurance premiums. The process is designed for seamless scale-up from laboratory to commercial production, ensuring that the environmental and safety benefits observed at small scale are maintained and amplified at the multi-ton level, aligning with corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common inquiries regarding the technical and commercial implications of the patented synthesis method for AZD6738. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing clarity on performance metrics and operational considerations. Understanding these details is vital for stakeholders evaluating the feasibility of integrating this technology into their existing manufacturing portfolios or sourcing strategies.
Q: What is the primary advantage of the vanadium-catalyzed oxidation method over enzymatic methods for AZD6738?
A: The vanadium-catalyzed method eliminates the need for expensive biocatalysts and specialized reactors, significantly reducing raw material costs while maintaining comparable yields and simplifying purification without preparative chromatography.
Q: How does the flow chemistry approach impact the overall yield of AZD6738 synthesis?
A: According to the patent data, integrating continuous flow technology for specific steps increases the total synthesis yield from approximately 16.6% in batch mode to over 25%, enhancing process efficiency and throughput.
Q: Is the chiral ligand used in this process commercially available or synthesized in situ?
A: The process allows flexibility; the chiral ligand can be prepared separately by condensing compound 13 and 14, or generated in situ during the reaction, offering operational convenience for scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable AZD6738 Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes for next-generation oncology therapeutics like AZD6738. Our team of expert chemists has thoroughly analyzed the innovations presented in CN111646985A and possesses the technical capability to implement these advanced vanadium-catalyzed oxidation and flow chemistry protocols. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with unwavering consistency. Our state-of-the-art facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of intermediate we deliver meets the highest industry standards for safety and efficacy.
We invite you to collaborate with us to leverage these cutting-edge manufacturing technologies for your project. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you optimize your supply chain and accelerate the development of life-saving medications through our commitment to innovation, quality, and reliability in the production of complex pharmaceutical intermediates.
