Scalable Asymmetric Synthesis of D-Citronellal for Global Flavor and Pharma Supply Chains
The global demand for high-purity chiral intermediates in the flavor and fragrance industry continues to drive innovation in asymmetric synthesis technologies. A pivotal development in this sector is detailed in patent CN103724170A, which discloses a novel method for the asymmetric synthesis of D-citronellal. This compound serves as a critical precursor for menthol and various high-value terpenoid aroma compounds. The disclosed technology utilizes citral as a starting material, employing a dihydropyridine derivative as a negative hydrogen source and a chiral amine salt as a chiral auxiliary agent. This dual catalytic system facilitates an asymmetric hydrogenation reaction that achieves a total yield greater than 85% and an ee value exceeding 80%. For R&D Directors and Procurement Managers seeking a reliable D-Citronellal supplier, this metal-free approach represents a significant shift towards more sustainable and cost-effective manufacturing paradigms, offering mild reaction conditions and simplified catalyst recovery suitable for large-scale industrial production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of optically pure D-citronellal has relied on complex multi-step pathways that introduce significant supply chain bottlenecks and cost inefficiencies. One prominent conventional route, developed by Takasago, involves the asymmetric hydrogen migration of N,N-diethylgeranylamine using a (S)-BINAP-Rh+ type catalyst. While this method produces high-quality products, the preparation conditions for the rhodium-based catalyst are notoriously harsh, and the recycling of such precious metal catalysts is technically difficult and economically burdensome. Furthermore, alternative routes, such as the one developed by BASF using isobutylene and formaldehyde, require the rectification of cis-trans citral isomer mixtures to obtain the specific cis-configuration needed for subsequent steps. This distillation process demands high-specification production equipment and results in the generation of trans-citral byproducts that are difficult to reuse, thereby lowering overall atom economy and increasing waste disposal costs for chemical manufacturers.
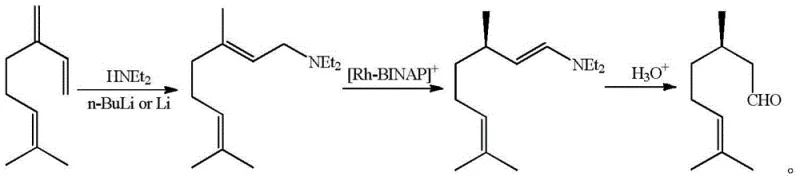
The Novel Approach
In stark contrast to these legacy methods, the novel organocatalytic approach described in the patent data offers a streamlined pathway that bypasses the need for precious metals and rigorous isomer separation. By utilizing a chiral amine salt and a dihydropyridine derivative, the reaction proceeds under significantly milder conditions, typically ranging from -30°C to 100°C, with a preferred range of -20°C to 60°C. This flexibility in temperature control allows for easier thermal management in large-scale reactors, reducing energy consumption associated with extreme cooling or heating. Moreover, the ability to use a mixture of cis-trans citral isomers directly without prior rectification eliminates a major unit operation from the production line. This simplification not only reduces capital expenditure on distillation columns but also shortens the overall production cycle time, providing a robust solution for cost reduction in flavor and fragrance manufacturing while maintaining high stereochemical control.
Mechanistic Insights into Organocatalytic Asymmetric Hydrogenation
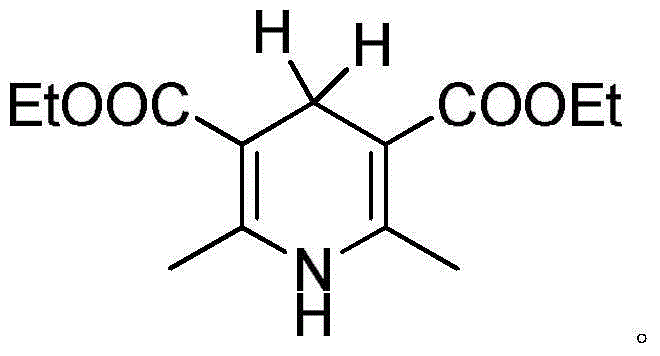
The core of this technological breakthrough lies in the sophisticated interaction between the chiral amine salt auxiliary and the dihydropyridine hydride source. The chiral amine salt, specifically derivatives of (R)-2-[bis(aryl)]methyltetrahydropyrrole, acts as a stereochemical director, creating a chiral environment around the substrate. When combined with the dihydropyridine derivative, such as 2,6-dimethyl-1,4-dihydropyridine-3,5-dicarboxylate diethyl ester, the system facilitates a selective hydrogen transfer. The negative hydrogen is transferred specifically to the double bond of the unsaturated aldehyde, resulting in the saturated beta-substituted aldehyde with high enantioselectivity. This mechanism avoids the formation of racemic mixtures common in non-catalytic hydrogenation, ensuring that the resulting D-citronellal possesses the specific optical rotation required for high-end fragrance applications where scent profiles are sensitive to stereochemistry.
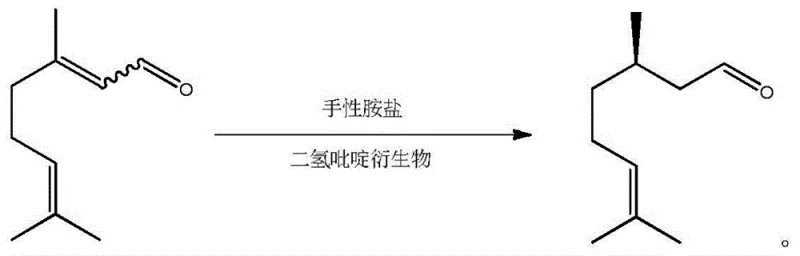
Impurity control is another critical aspect where this mechanism excels, particularly for R&D teams focused on purity specifications. The use of organic small molecule catalysts rather than transition metals inherently reduces the risk of heavy metal contamination, a common regulatory hurdle in pharmaceutical and food-grade intermediate production. The reaction medium, which can include solvents like tetrahydrofuran, toluene, or dichloromethane, allows for efficient solubility of reactants while maintaining the stability of the chiral catalyst. The catalytic amount of the chiral amine salt auxiliary is relatively low, typically between 0.01 to 10 mol%, which minimizes the load of chiral reagents that need to be separated post-reaction. This precise control over reaction parameters ensures that side reactions, such as over-reduction or isomerization of the double bond, are minimized, leading to a cleaner crude product that requires less intensive downstream purification.
How to Synthesize D-Citronellal Efficiently
Implementing this synthesis route requires careful attention to the stoichiometry and reaction environment to maximize yield and enantiomeric excess. The process begins with the preparation of the reaction mixture under an inert atmosphere, typically nitrogen, to prevent oxidation of the sensitive aldehyde and hydride species. The detailed standardized synthesis steps involve specific molar ratios of citral to the dihydropyridine derivative, often ranging from 1:1 to 1:8, depending on the desired conversion rate. Reaction times are optimized between 5 to 48 hours, with a preferred window of 10 to 24 hours, balancing throughput with complete conversion. For technical teams looking to scale this process, the following guide outlines the critical operational parameters derived from the patent examples, ensuring reproducibility and safety in a commercial setting.
- Prepare the reaction mixture by dissolving citral, chiral amine salt auxiliary, and dihydropyridine hydride source in an organic solvent such as toluene or THF.
- Maintain the reaction temperature between -20°C and 60°C under nitrogen protection, stirring for 10 to 24 hours to ensure complete asymmetric hydrogenation.
- Remove the solvent under reduced pressure and purify the crude product via column chromatography to isolate D-Citronellal with high enantiomeric excess.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this organocatalytic method offers tangible strategic advantages beyond mere technical feasibility. The elimination of expensive transition metal catalysts, such as rhodium complexes, directly translates to substantial cost savings in raw material procurement. Additionally, the simplified workup procedure, which avoids complex metal scavenging steps, reduces the consumption of auxiliary chemicals and lowers waste treatment costs. The robustness of the reaction conditions allows for the use of standard stainless steel reactors without the need for specialized lining required for highly corrosive or high-pressure metal-catalyzed processes. This compatibility with existing infrastructure accelerates the timeline for technology transfer and commercial scale-up of complex flavor intermediates, ensuring a more reliable supply chain for downstream customers.
- Cost Reduction in Manufacturing: The removal of precious metal catalysts from the synthesis route eliminates a significant cost driver associated with volatile metal markets and complex recovery systems. By relying on organic small molecules that are easier to synthesize and handle, manufacturers can achieve a drastically simplified cost structure. Furthermore, the ability to utilize mixed isomer citral feedstocks without prior separation reduces the energy intensity of the process, leading to lower utility bills and improved overall margin profiles for high-purity D-Citronellal production.
- Enhanced Supply Chain Reliability: The reagents required for this process, including dihydropyridine derivatives and chiral amine salts, are more readily available and stable compared to sensitive organometallic catalysts. This availability reduces the risk of supply disruptions caused by geopolitical issues affecting rare metal mining or specialized catalyst manufacturing. The mild reaction conditions also enhance operational safety, reducing the likelihood of unplanned shutdowns due to thermal runaways or pressure incidents, thereby ensuring consistent delivery schedules for global clients.
- Scalability and Environmental Compliance: The process generates significantly less hazardous waste, particularly heavy metal residues, which simplifies compliance with increasingly stringent environmental regulations in major chemical manufacturing hubs. The solvents used, such as toluene and THF, are standard industrial solvents with well-established recovery and recycling protocols. This environmental friendliness not only reduces disposal costs but also aligns with the sustainability goals of major multinational corporations, making the supply of these intermediates more attractive for green chemistry initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric synthesis technology. These insights are derived directly from the patent specifications and are intended to clarify the operational feasibility and quality outcomes of the process. Understanding these details is crucial for technical procurement teams evaluating the viability of this route for their specific product portfolios and quality standards.
Q: What are the advantages of this organocatalytic route over traditional metal-catalyzed methods?
A: This method eliminates the need for expensive transition metal catalysts like Rhodium, significantly reducing raw material costs and simplifying the removal of heavy metal residues from the final product.
Q: Does this process require pure cis-citral as a starting material?
A: No, the process is robust enough to use a mixture of cis-trans citral isomers directly, removing the need for energy-intensive rectification steps to separate isomers before reaction.
Q: What is the expected enantiomeric excess (ee) and yield for this synthesis?
A: According to patent data, the total yield exceeds 85% with an enantiomeric excess (ee) value greater than 80%, ensuring high optical purity suitable for premium fragrance applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable D-Citronellal Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic routes in securing a stable supply of high-value intermediates. Our CDMO expertise allows us to adapt complex pathways like the organocatalytic synthesis of D-Citronellal from laboratory scale to full commercial production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high yields and ee values reported in patent literature are maintained at an industrial level. Our rigorous QC labs and stringent purity specifications guarantee that every batch meets the exacting standards required by the global flavor and pharmaceutical industries, providing our partners with a secure and high-quality source of critical chiral building blocks.
We invite procurement leaders and R&D directors to collaborate with us on optimizing their supply chains for chiral intermediates. By leveraging our technical capabilities, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments for D-Citronellal and related terpenoid compounds. Let us help you navigate the complexities of chiral synthesis to achieve greater efficiency and reliability in your manufacturing operations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
