Revolutionizing Organometallic Catalysis with One-Pot Eight-Membered Amidine Palladium Compounds
Introduction to Patent CN110981919A
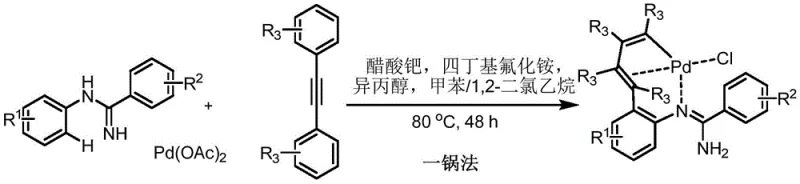
The landscape of organometallic chemistry is constantly evolving, driven by the need for more efficient and sustainable synthetic methodologies. A groundbreaking development in this field is detailed in Chinese Patent CN110981919A, which discloses a novel one-pot method for synthesizing eight-membered amidine ring palladium compounds. This technology represents a significant leap forward in C-H bond activation strategies, utilizing N-phenylbenzamidine derivatives as key synthons. By integrating C-H activation with bimolecular alkyne insertion in a single reaction vessel, this process eliminates the cumbersome isolation steps typically associated with constructing macrocyclic palladacycles. For R&D directors and process chemists, this patent offers a robust pathway to access complex organometallic structures that serve as potent catalysts for cross-coupling reactions, specifically Suzuki couplings. The method operates under mild conditions, ranging from 80°C to 110°C, and demonstrates remarkable tolerance to air and moisture, addressing common stability issues in organometallic synthesis.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of metal cyclopalladated compounds, particularly those with larger ring sizes like eight-membered systems, has been fraught with operational complexity. Traditional literature, such as works by Saxena and Vicente, describes multi-step sequences where a six-membered or five-membered palladacycle intermediate must first be synthesized, isolated, and purified before undergoing further alkyne insertion reactions. This stepwise approach not only increases the total processing time but also leads to significant material loss during each isolation stage, thereby reducing the overall atom economy. Furthermore, the intermediates involved in these classical routes often require stringent inert atmosphere conditions, complicating scale-up efforts and increasing the capital expenditure for specialized equipment. The tedious nature of these protocols makes them less attractive for industrial applications where throughput and cost-efficiency are paramount concerns for supply chain managers.
The Novel Approach
In stark contrast, the methodology presented in CN110981919A streamlines the entire synthesis into a convergent one-pot operation. By leveraging the directing ability of the C=N group in N-phenylbenzamidine, the process facilitates an initial ortho-C-H activation followed immediately by the insertion of two alkyne molecules without interrupting the reaction sequence. This tandem transformation bypasses the need to isolate the six-membered ring intermediate, effectively collapsing what was previously a multi-day procedure into a single heating step lasting 12 to 48 hours. The result is a drastic simplification of the workflow, which translates directly into reduced labor costs and minimized solvent consumption. For procurement teams, this efficiency implies a more reliable supply of high-purity catalysts, as the reduced number of unit operations lowers the probability of batch-to-batch variability and contamination.
Mechanistic Insights into C-H Activation and Alkyne Insertion
The core of this technological advancement lies in the sophisticated interplay between the palladium center and the organic substrates. The mechanism initiates with the coordination of the palladium salt to the nitrogen atom of the amidine group, which acts as a powerful directing group to activate the adjacent ortho-C-H bond on the phenyl ring. This cyclometallation step generates a reactive Pd-C bond, setting the stage for the subsequent insertion events. Unlike simple oxidative additions, this C-H activation is highly selective and proceeds efficiently under the specified thermal conditions. Once the metallacycle is formed, the system undergoes a sequential insertion of two alkyne molecules into the Pd-C bond. This bimolecular insertion is critical for expanding the ring size from a typical five or six-membered structure to the desired eight-membered framework. The electronic properties of the alkyne substituents play a crucial role here, influencing both the rate of insertion and the final regioselectivity of the product.
From an impurity control perspective, the one-pot nature of the reaction inherently limits the formation of side products associated with intermediate handling. Since the reactive palladacycle intermediate is not exposed to the external environment or purification media, degradation pathways such as hydrolysis or oxidation are minimized. The use of specific additives, such as tetrabutylammonium fluoride, further enhances the reaction efficiency by potentially facilitating the C-H cleavage step or stabilizing the transition states involved in alkyne insertion. The final product, an eight-membered amidine cyclic palladium compound, features a stable metal-carbon bond and a coordinated nitrogen donor, creating a rigid structure that is ideal for catalytic turnover. This structural integrity ensures that the catalyst maintains its activity over multiple cycles, a key metric for evaluating the long-term value of any homogeneous catalyst in industrial processes.
How to Synthesize Eight-Membered Amidine Cyclopalladated Compounds Efficiently
Implementing this synthesis requires precise control over reaction parameters to maximize yield and purity. The standard protocol involves combining N-phenylbenzamidine, a diarylacetylene derivative, palladium acetate, and a fluoride additive in a mixed solvent system typically comprising toluene, 1,2-dichloroethane, and isopropanol. The reaction mixture is then heated to temperatures between 80°C and 110°C for a duration of 12 to 48 hours, depending on the specific substrate reactivity. Upon completion, the solvent is removed under reduced pressure, and the crude product is purified via column chromatography to afford the target orange solid. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility across different laboratory settings.
- Mix N-phenylbenzamidine, alkyne compounds, palladium acetate, tetrabutylammonium fluoride, and solvent system (toluene/DCE/iPrOH).
- Heat the reaction mixture to 80-110°C and maintain for 12-48 hours to facilitate C-H activation and double alkyne insertion.
- Cool to room temperature, remove solvent under reduced pressure, and purify via column chromatography to isolate the target palladacycle.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders focused on the bottom line and supply continuity, the shift to this one-pot methodology offers compelling economic arguments. The elimination of intermediate isolation steps fundamentally alters the cost structure of producing these specialized organometallic catalysts. By removing the need for separate workups, drying, and purification of the six-membered precursor, manufacturers can achieve substantial cost savings in terms of both raw materials and energy consumption. This streamlined process reduces the overall manufacturing footprint, allowing for higher throughput within existing facility constraints. Additionally, the robustness of the reaction conditions, which do not require strict inert gas protection, lowers the barrier for entry for contract manufacturing organizations, thereby diversifying the potential supplier base and enhancing supply chain resilience.
- Cost Reduction in Manufacturing: The convergence of multiple synthetic steps into a single pot drastically reduces the operational expenses associated with labor and solvent usage. Without the need to isolate and purify intermediates, the cumulative yield losses typical of multi-step syntheses are avoided, leading to a higher overall output of the final catalyst per kilogram of starting material. This improvement in atom utilization directly correlates to a lower cost of goods sold, making high-performance palladium catalysts more accessible for large-scale pharmaceutical applications.
- Enhanced Supply Chain Reliability: The insensitivity of this reaction to air and moisture simplifies logistics and storage requirements. Unlike sensitive organometallic reagents that demand cold chain shipping or glovebox handling, these eight-membered palladacycles can be managed with standard chemical handling procedures. This durability reduces the risk of spoilage during transit and storage, ensuring that procurement managers receive materials that meet specification upon arrival. Furthermore, the wide substrate scope allows for flexibility in sourcing raw materials, mitigating risks associated with the shortage of specific niche reagents.
- Scalability and Environmental Compliance: The mild reaction temperatures and simplified workup procedure make this process highly amenable to scale-up from gram to kilogram quantities. The reduction in solvent volume and the avoidance of hazardous intermediate handling steps contribute to a greener manufacturing profile, aligning with increasingly stringent environmental regulations. For supply chain heads, this means easier permitting for production facilities and a reduced environmental liability, supporting sustainable sourcing initiatives that are critical for modern corporate responsibility goals.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and application of this patented technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for technical teams evaluating this route for adoption. Understanding these nuances is essential for assessing the feasibility of integrating this catalyst system into existing production lines.
Q: What is the primary advantage of this one-pot synthesis over traditional methods?
A: Unlike traditional multi-step routes requiring intermediate isolation, this method constructs the eight-membered ring directly via tandem C-H activation and alkyne insertion, significantly simplifying operations and improving atom economy.
Q: Is the resulting palladacycle stable for industrial handling?
A: Yes, the synthesized eight-membered amidine cyclic palladium compounds are insensitive to water and air, allowing for simpler handling and storage compared to sensitive organometallic species.
Q: What is the catalytic application of these compounds?
A: These novel palladacycles serve as highly efficient catalysts for Suzuki coupling reactions, demonstrating excellent activity in forming biaryl structures essential for pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Eight-Membered Amidine Palladium Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced C-H activation technologies in modern drug discovery and process chemistry. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN110981919A can be successfully translated to industrial reality. Our rigorous QC labs and commitment to stringent purity specifications guarantee that every batch of organometallic catalyst meets the exacting standards required for API synthesis. We understand that consistency is key in catalytic applications, and our state-of-the-art facilities are equipped to handle the specific solvent systems and thermal profiles necessary for these palladacycle syntheses.
We invite you to collaborate with us to leverage this cutting-edge chemistry for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in complex organometallic synthesis can drive efficiency and innovation in your supply chain.
