Revolutionizing Amide Bond Formation: A Deep Dive into Visible-Light Photocatalytic Dehydrogenation for Commercial Scale-up
Revolutionizing Amide Bond Formation: A Deep Dive into Visible-Light Photocatalytic Dehydrogenation for Commercial Scale-up
The pharmaceutical and fine chemical industries are constantly seeking sustainable alternatives to traditional amide bond formation, a cornerstone reaction in the synthesis of active pharmaceutical ingredients (APIs) and agrochemical intermediates. Patent CN111574477A discloses a groundbreaking synthesis method for amide compounds that leverages visible-light photocatalysis to drive the direct dehydrogenative coupling of benzyl alcohols and amines. This technology represents a paradigm shift from energy-intensive thermal processes to mild, room-temperature reactions mediated by a novel metal-loaded P-C3N4 heterogeneous catalyst. By utilizing abundant feedstocks like benzyl alcohol derivatives and avoiding stoichiometric coupling reagents, this method addresses critical pain points regarding atomic economy and waste generation. For R&D directors and process chemists, understanding the mechanistic nuances of this photocatalytic system is essential for integrating greener pathways into existing manufacturing pipelines.
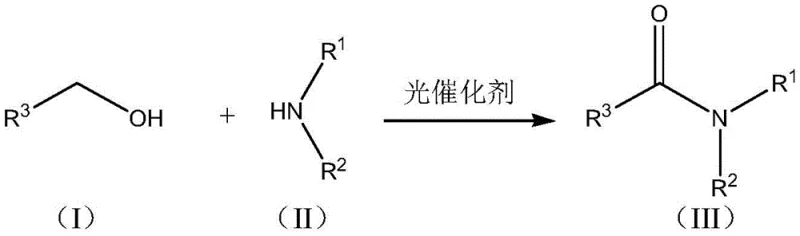
Furthermore, the strategic implementation of this technology offers substantial implications for supply chain resilience and cost structures. As a reliable pharmaceutical intermediate supplier, analyzing such patents allows us to forecast trends towards lower-energy manufacturing protocols. The ability to synthesize complex amide structures under ambient conditions not only reduces the carbon footprint but also mitigates the risks associated with handling hazardous oxidants. This report provides a comprehensive technical analysis of the patented method, evaluating its feasibility for commercial scale-up of complex intermediates and its potential to redefine cost reduction in amide manufacturing through catalyst recyclability and operational simplicity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional amide synthesis has long relied on the activation of carboxylic acids using coupling reagents such as carbodiimides, phosphonium salts, or the conversion of acids into highly reactive acid chlorides. While effective on a small laboratory scale, these conventional methods suffer from severe drawbacks when translated to industrial production. The primary issue lies in poor atomic economy; these reactions generate stoichiometric amounts of waste byproducts, such as urea derivatives or salt wastes, which require complex and costly downstream purification processes. Additionally, many traditional protocols necessitate high reaction temperatures and the use of hazardous solvents, increasing energy consumption and safety risks. The reliance on precious metal homogeneous catalysts in some modern variations further complicates the process, as removing trace metal impurities to meet stringent pharmaceutical purity specifications often requires additional chromatography or scavenging steps, driving up both time and material costs significantly.
The Novel Approach
In stark contrast, the method disclosed in CN111574477A introduces a direct oxidative coupling strategy that bypasses the need for pre-activated acid derivatives. By employing a metal-loaded P-C3N4 photocatalyst, the reaction proceeds via a dehydrogenative pathway where the only byproduct is hydrogen gas (or water if oxygen is used as a terminal oxidant), drastically improving atom economy. This novel approach operates at room temperature under visible light irradiation, eliminating the need for external heating sources and reducing the thermal load on the reactor system. The use of a heterogeneous catalyst means that the solid catalyst can be easily separated from the liquid reaction mixture via simple filtration, solving the persistent industry challenge of catalyst recovery. This transition from homogeneous to heterogeneous photocatalysis not only simplifies post-treatment but also enables the potential for continuous flow processing, a key factor for enhancing supply chain reliability and throughput in modern chemical manufacturing facilities.
Mechanistic Insights into Metal-Loaded P-C3N4 Photocatalytic Dehydrogenation
The core innovation of this technology lies in the sophisticated design of the photocatalyst, specifically the phosphorus-doped graphitic carbon nitride (P-C3N4) loaded with transition metals such as silver, ruthenium, or copper. Under visible light irradiation, the P-C3N4 semiconductor absorbs photons to generate electron-hole pairs. The doping with phosphorus modifies the band structure, enhancing visible light absorption and charge separation efficiency. The loaded metal nanoparticles act as electron traps, effectively preventing the recombination of photogenerated electrons and holes, thereby extending the lifetime of the charge carriers. These active species facilitate the abstraction of hydrogen atoms from the hydroxyl group of the benzyl alcohol and the amino group of the amine. This dual activation mechanism allows for the direct formation of the C-N bond with the concurrent release of hydrogen, a process that is thermodynamically favorable yet kinetically challenging without the precise electronic environment provided by the metal-semiconductor interface.
From an impurity control perspective, this photocatalytic mechanism offers superior selectivity compared to thermal oxidation methods. Traditional thermal catalysis often suffers from over-oxidation, where the intermediate aldehyde is further oxidized to the corresponding carboxylic acid, leading to difficult-to-separate impurity profiles. However, the mild redox potential of the photogenerated holes in this system is tuned to stop at the amide formation stage, minimizing the formation of acid byproducts. Furthermore, the reaction conditions are mild enough to preserve sensitive functional groups on the aromatic rings or heterocyclic systems of the substrates, which might otherwise degrade under harsh acidic or basic thermal conditions. This high level of chemoselectivity ensures that the resulting high-purity amide compounds meet the rigorous quality standards required for pharmaceutical applications, reducing the burden on downstream purification units and increasing overall process yield.
How to Synthesize Amide Compounds Efficiently
The synthesis protocol outlined in the patent provides a robust framework for producing a wide variety of amide derivatives, ranging from simple N-benzylamides to complex heterocyclic amides. The process begins with the preparation of the catalyst, involving the crystallization of melamine with phosphoric acid followed by calcination to form the P-C3N4 support, which is subsequently loaded with metal salts and calcined again. In the reaction phase, benzyl alcohol and the amine are mixed in an organic solvent such as toluene or THF, with an optional base like potassium tert-butoxide added to facilitate proton transfer. The mixture is then irradiated with a standard LED light source (15W to 300W) at room temperature. Detailed standardized synthesis steps see the guide below.
- Prepare the heterogeneous photocatalyst by calcining melamine with phosphoric acid to form P-C3N4, followed by metal loading (e.g., Ag, Ru, Au) and secondary calcination.
- Combine benzyl alcohol, amine compound, organic solvent (e.g., toluene), optional base, and the metal-loaded photocatalyst in a reaction vessel.
- Stir the mixture under visible light irradiation (15W-300W LED) at room temperature for 60-600 minutes, then filter to recover the catalyst and isolate the amide product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this photocatalytic technology translates into tangible strategic advantages beyond mere technical novelty. The shift towards room-temperature reactions significantly lowers utility costs associated with heating and cooling large-scale reactors, contributing to substantial cost savings in amide manufacturing. Moreover, the elimination of stoichiometric coupling reagents and hazardous oxidants like peroxides reduces the raw material inventory costs and mitigates the regulatory burdens associated with transporting and storing dangerous chemicals. The simplified work-up procedure, driven by the ease of filtering the heterogeneous catalyst, shortens the batch cycle time, allowing for faster turnover and improved responsiveness to market demands. These factors collectively enhance the economic viability of producing high-value intermediates, making the supply chain more agile and cost-competitive in a global market.
- Cost Reduction in Manufacturing: The implementation of a reusable heterogeneous catalyst fundamentally alters the cost structure of amide production. Unlike homogeneous catalysts that are lost in the work-up or require expensive removal processes, the metal-loaded P-C3N4 catalyst can be recovered by simple filtration and reused multiple times without significant loss of activity, as demonstrated in the patent data. This recyclability drastically reduces the cost per kilogram of the catalyst consumed. Additionally, the avoidance of expensive coupling reagents and the reduction in solvent usage due to simpler purification protocols contribute to a leaner manufacturing process. The energy efficiency gained from operating at room temperature rather than reflux conditions further compounds these savings, offering a compelling economic case for scaling this technology.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the availability of specialized reagents and the complexity of multi-step syntheses. This photocatalytic method utilizes readily available starting materials like benzyl alcohols and amines, which are commodity chemicals with stable supply lines. The robustness of the catalyst and the mild reaction conditions reduce the risk of batch failures due to thermal runaways or sensitivity to moisture and oxygen, which are common issues in traditional amide synthesis. By simplifying the process flow and reducing the dependency on fragile reagent supply chains, manufacturers can ensure a more consistent and reliable output of high-purity intermediates, safeguarding against production delays and ensuring on-time delivery to downstream clients.
- Scalability and Environmental Compliance: As environmental regulations become increasingly stringent, the ability to scale processes that align with green chemistry principles is a critical competitive advantage. This photocatalytic route generates minimal waste, primarily hydrogen gas, and avoids the discharge of heavy metal contaminants or toxic organic byproducts associated with traditional methods. The heterogeneous nature of the catalyst facilitates compliance with strict limits on residual metals in pharmaceutical products. Furthermore, the modularity of LED light sources allows for flexible reactor design, enabling easy scale-up from pilot plants to commercial production volumes. This scalability ensures that the technology can meet growing demand without compromising on environmental standards or requiring massive capital investment in new infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this photocatalytic amide synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on catalyst performance, substrate scope, and process safety. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this method for their specific product portfolios.
Q: How does the metal-loaded P-C3N4 catalyst improve reaction efficiency compared to traditional thermal methods?
A: The metal-loaded P-C3N4 catalyst utilizes visible light to generate electron-hole pairs, facilitating direct dehydrogenation at room temperature. This avoids the high thermal energy input required by traditional methods, significantly reducing side reactions and improving selectivity for the target amide bond without needing stoichiometric oxidants.
Q: Is the photocatalyst reusable for industrial scale production?
A: Yes, the patent data indicates that the heterogeneous nature of the metal-loaded P-C3N4 allows for simple filtration and recovery. Experimental results demonstrate that the catalyst can be reused multiple times (up to 6 cycles in tested examples) without significant loss of catalytic activity, which is crucial for cost-effective manufacturing.
Q: What are the safety advantages of this photocatalytic method over peroxide-based oxidations?
A: Traditional thermal catalytic couplings often rely on excessive peroxides which are thermally unstable and pose explosion hazards. This photocatalytic method operates under mild conditions using oxygen or air as the terminal oxidant (or via direct dehydrogenation), eliminating the need for hazardous peroxide reagents and enhancing overall process safety.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Amide Compounds Supplier
The technological advancements detailed in patent CN111574477A underscore the evolving landscape of chemical synthesis, where efficiency and sustainability are paramount. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to bring such innovative methods to life. Our state-of-the-art facilities are equipped to handle complex photocatalytic processes, ensuring that every batch meets stringent purity specifications through our rigorous QC labs. We understand that transitioning to new synthetic routes requires a partner with deep technical expertise and a commitment to quality, and we are dedicated to supporting our clients through every stage of development and commercialization.
We invite you to explore how our capabilities can optimize your supply chain and reduce your manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific amide synthesis needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our advanced photocatalytic technologies can deliver high-purity intermediates with unmatched reliability and efficiency. Let us collaborate to build a more sustainable and profitable future for your chemical projects.
