Scalable Production of 2-Amino-4-Bromopyridine via Novel Ammonolysis-Reduction Strategy
Introduction to Advanced Synthetic Methodologies
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the urgent need for more efficient, cost-effective, and scalable synthetic routes. A pivotal development in this domain is documented in patent CN102603622B, which discloses a robust synthetic method for 2-amino-4-bromopyridine, a critical building block in the design of novel heterocyclic compounds for medicinal and agrochemical applications. This patent addresses the longstanding challenges associated with traditional synthesis pathways, such as prohibitive raw material costs and suboptimal reaction yields that hinder commercial viability. By leveraging a strategic ammonolysis-reduction sequence starting from 2,4-dibromopyridine-N-oxide, the disclosed methodology achieves a remarkable total yield of up to 80.5 percent. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, understanding the mechanistic advantages of this route is essential for optimizing supply chains and reducing the overall cost of goods sold in complex drug manufacturing processes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
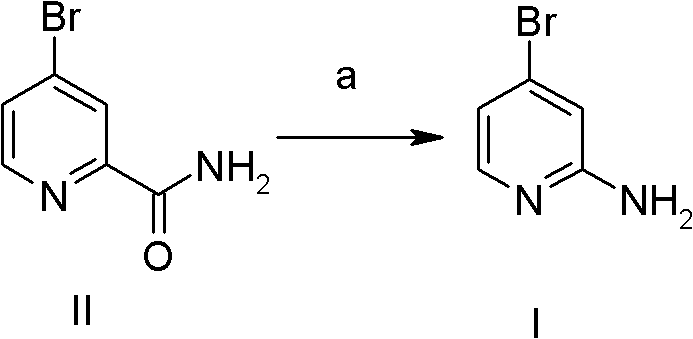
Historically, the synthesis of 2-amino-4-bromopyridine has been plagued by inefficient multi-step sequences that rely on expensive precursors and harsh reaction conditions. One prominent prior art method, referenced in WO2006038116, utilizes an amide-based starting material that undergoes bromination and subsequent hydrolysis. As illustrated in the reaction scheme below, this pathway is not only chemically cumbersome but also economically burdensome due to the low isolated yield of approximately 57.8 percent. Furthermore, alternative routes involving diazotization reactions, such as those reported in WO2008057469, introduce significant safety hazards and operational complexities. These conventional approaches often involve steps with yields as low as 22 percent, creating massive amounts of chemical waste and driving up the unit cost of the final API intermediate. Such inefficiencies make these legacy methods unsuitable for the rigorous demands of modern large-scale industrial preparation, where consistency and atom economy are paramount.
The Novel Approach
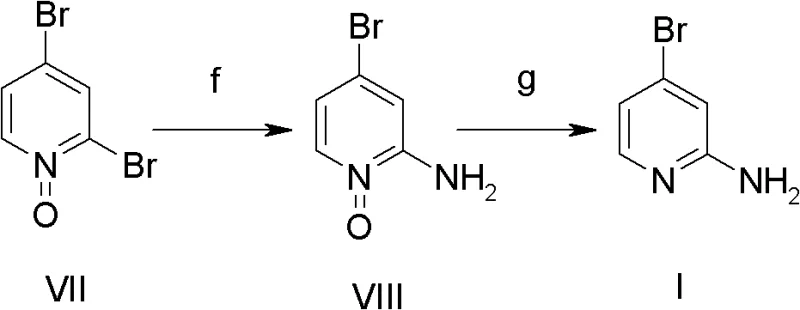
In stark contrast to these legacy methodologies, the novel approach outlined in CN102603622B streamlines the synthesis into a highly efficient two-step process that maximizes atom economy and minimizes waste generation. The strategy employs 2,4-dibromopyridine-N-oxide as a versatile and cost-effective starting material, which undergoes a direct nucleophilic substitution with ammonia water. This transformation is followed by a straightforward reduction step to remove the N-oxide functionality, delivering the target 2-amino-4-bromopyridine with exceptional purity. The simplicity of this route eliminates the need for protecting groups or exotic reagents, significantly lowering the barrier to entry for commercial production. By bypassing the low-yielding hydrolysis and diazotization steps of the past, this new method offers a direct path to high-purity intermediates, making it an ideal candidate for manufacturers aiming to enhance their competitive edge in the global fine chemicals market.
Mechanistic Insights into N-Oxide Activation and Reduction
The success of this synthetic strategy hinges on the unique electronic properties of the pyridine N-oxide moiety, which serves as a powerful activator for nucleophilic aromatic substitution. In the first critical step (Step f), the electron-withdrawing nature of the N-oxide oxygen increases the electrophilicity of the carbon atoms at the 2 and 4 positions of the pyridine ring. This activation allows the relatively weak nucleophile, ammonia, to displace the bromine atom at the 2-position under moderate thermal conditions ranging from 60 to 110 degrees Celsius. The reaction is typically conducted in a sealed pressure vessel to maintain the concentration of ammonia, driving the equilibrium towards the formation of the 2-amino-4-bromopyridine-N-oxide intermediate. This mechanistic feature is crucial because it avoids the need for strong bases or transition metal catalysts often required for unactivated aryl halides, thereby simplifying the workup and reducing the risk of metal contamination in the final product.
Following the ammonolysis, the second phase involves the reductive removal of the N-oxide group to restore the aromatic pyridine system (Step g). This reduction can be achieved through two distinct pathways, offering flexibility for different manufacturing setups. The first option utilizes iron powder in the presence of an acid such as hydrochloric or acetic acid, a method known for its low cost and ease of execution on a multi-ton scale. Alternatively, catalytic hydrogenation using Raney nickel or palladium on carbon (Pd/C) provides a cleaner profile with potentially faster kinetics. Both methods effectively cleave the N-O bond without affecting the sensitive amino or bromo functionalities, ensuring high selectivity. The ability to choose between base metal reduction and catalytic hydrogenation allows process chemists to tailor the operation to their specific safety and environmental compliance requirements while maintaining high product integrity.
How to Synthesize 2-Amino-4-Bromopyridine Efficiently
Implementing this synthesis requires careful attention to reaction parameters to ensure optimal conversion and safety. The process begins with the preparation of the reaction mixture in a pressure-resistant reactor, where the stoichiometry of ammonia water to the N-oxide substrate is carefully controlled, typically within a mass ratio of 1:2.5 to 1:5.0. Maintaining the temperature between 90 and 95 degrees Celsius during the overnight stirring period ensures complete consumption of the starting material, yielding a crude intermediate that often requires no further purification before the next step. For the reduction phase, whether using iron powder or catalytic hydrogenation, the reaction is monitored until completion, followed by standard filtration and concentration techniques. The detailed standardized operating procedures, including specific solvent volumes, stirring rates, and safety protocols for handling pressurized ammonia and hydrogen, are critical for successful technology transfer.
- Conduct an ammonolysis reaction on 2,4-dibromopyridine-N-oxide using ammonia water in a sealed pressure reactor at 60-110°C to obtain the amino-intermediate.
- Perform a reduction reaction on the intermediate using either iron powder with acid or catalytic hydrogenation (Raney Nickel/Pd-C) to yield the final 2-amino-4-bromopyridine product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route translates into tangible strategic advantages that extend far beyond simple chemical yield. The primary benefit lies in the drastic simplification of the supply chain; by utilizing 2,4-dibromopyridine-N-oxide, a commercially accessible bulk chemical, manufacturers can avoid the volatility and high costs associated with specialized amide precursors or unstable diazonium salts. This shift to readily available raw materials significantly de-risks the procurement process, ensuring a steady flow of inputs even during periods of market fluctuation. Furthermore, the high total yield of over 80 percent means that less raw material is required to produce the same amount of finished goods, directly improving the margin structure. The elimination of complex purification steps for the intermediate also reduces solvent consumption and waste disposal costs, contributing to a more sustainable and economically viable manufacturing footprint.
- Cost Reduction in Manufacturing: The economic impact of this process is profound, primarily driven by the replacement of expensive starting materials with cost-effective alternatives and the substantial increase in overall yield. By avoiding the low-yielding steps characteristic of older methods, the amount of wasted reagents and solvents is minimized, leading to a significant decrease in the variable cost per kilogram of the final product. Additionally, the option to use iron powder for reduction instead of precious metal catalysts further lowers the operational expenditure, as it removes the need for expensive catalyst recovery systems or the purchase of noble metals. This cumulative effect results in a much more competitive pricing structure for the final API intermediate, allowing downstream pharmaceutical companies to optimize their own production budgets.
- Enhanced Supply Chain Reliability: From a logistics perspective, the robustness of this synthetic route ensures a more reliable supply of high-purity pharmaceutical intermediates. The use of stable, non-hazardous reagents like ammonia water and iron powder simplifies storage and transportation requirements compared to the hazardous reagents needed for diazotization. Moreover, the high conversion rates and simple workup procedures reduce the likelihood of batch failures or delays caused by difficult purifications. This reliability is crucial for maintaining continuous production schedules in large-scale facilities, preventing costly downtime and ensuring that downstream drug development timelines are met without interruption due to material shortages.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, having been validated from laboratory scales up to multi-kilogram batches with consistent results. The operational simplicity, involving standard heating and stirring without the need for cryogenic conditions or ultra-high pressures, makes it easily adaptable to existing industrial reactors. Environmentally, the reduction in step count and the potential to use greener reduction methods align with increasingly stringent global regulations on chemical waste and emissions. The ability to produce high-quality intermediates with a smaller environmental footprint enhances the corporate social responsibility profile of the manufacturer, making it a preferred partner for multinational corporations committed to sustainable sourcing practices.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of this technology, we have compiled answers to common questions regarding the process specifics and its industrial applicability. These insights are derived directly from the experimental data and technical disclosures found in the patent literature, providing a clear picture of what to expect during technology transfer. Understanding these nuances is vital for R&D directors planning pilot runs and for quality assurance teams establishing specification limits. The following responses address the core technical differentiators that set this method apart from conventional synthesis strategies.
Q: Why is the N-oxide route superior to conventional amide hydrolysis methods?
A: The N-oxide route utilizes readily available raw materials and achieves a total yield of over 80%, whereas conventional amide hydrolysis routes often suffer from lower yields (around 57%) and require more expensive starting materials.
Q: What reduction methods are compatible with this synthesis?
A: The process offers flexibility, allowing for reduction via inexpensive iron powder in acidic conditions or through catalytic hydrogenation using Raney Nickel or Pd/C, enabling manufacturers to choose based on their existing infrastructure.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the method is specifically designed for scale-up, featuring simple operational steps, mild reaction conditions, and the elimination of complex purification procedures for intermediates, ensuring high throughput.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Amino-4-Bromopyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this high-yield synthesis are fully realized in practice. Our state-of-the-art facilities are equipped to handle both the pressure reactions required for ammonolysis and the specialized hydrogenation or metal reduction steps, all while adhering to stringent purity specifications. With our rigorous QC labs and commitment to process optimization, we guarantee the delivery of high-purity pharmaceutical intermediates that meet the exacting standards of the global healthcare industry.
We invite you to collaborate with us to leverage this advanced synthetic technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this route can improve your bottom line. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing you to make informed decisions about integrating this efficient 2-amino-4-bromopyridine supply into your value chain.
