Advanced Ambient Pressure Synthesis of 2,6-Dihydroxyterephthalic Acid for High-Performance Polymers
Advanced Ambient Pressure Synthesis of 2,6-Dihydroxyterephthalic Acid for High-Performance Polymers
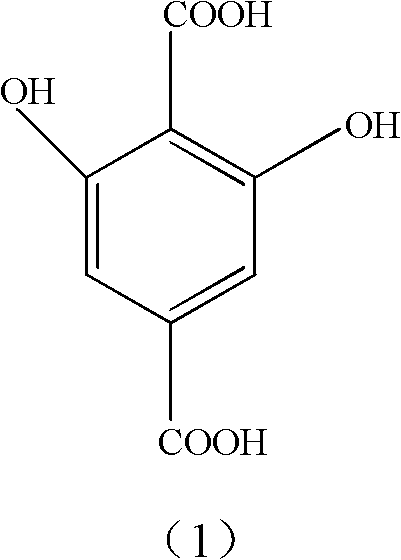
The global demand for high-performance polymeric materials, particularly those utilized in aerospace and advanced textile applications, has necessitated the development of robust synthetic routes for key monomers like 2,6-dihydroxyterephthalic acid. Patent CN101875606A introduces a groundbreaking methodology that addresses the longstanding challenges associated with the industrial production of this critical intermediate. By leveraging a novel molten salt system, this technology enables the efficient carboxylation of 3,5-dihydroxybenzoic acid under ambient pressure conditions, marking a significant departure from the hazardous high-pressure protocols of the past. This innovation not only enhances the safety profile of the manufacturing process but also delivers exceptional product purity and yield, making it an ideal candidate for reliable agrochemical intermediate supplier networks and pharmaceutical supply chains alike. The structural integrity of the target molecule, as depicted below, is crucial for its function in modifying DH-PBO fibers, where the specific positioning of hydroxyl groups dictates the material's mechanical and thermal properties.
Furthermore, the economic implications of adopting this synthesis route are profound for stakeholders focused on cost reduction in electronic chemical manufacturing and specialty polymer production. The ability to operate at normal pressure drastically reduces the capital expenditure associated with reactor infrastructure, removing the need for specialized high-pressure vessels capable of withstanding forces up to 80MPa. This shift allows for a more flexible and scalable production environment, ensuring that supply chain heads can secure consistent volumes of high-purity 2,6-dihydroxyterephthalic acid without the bottlenecks typical of batch processes requiring extensive safety interlocks. As we delve deeper into the technical specifics, it becomes evident that this patent represents a pivotal advancement in the field of fine chemical intermediates, offering a sustainable pathway for the next generation of high-performance materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2,6-dihydroxyterephthalic acid has been plagued by severe operational constraints that hindered its widespread commercial adoption. Early methodologies, such as those described by Hauptschein, relied on the carboxylation of 5-trifluoromethyl resorcinol followed by hydrolysis, a multi-step sequence that suffered from low overall yields and the scarcity of fluorinated starting materials. More recent attempts, documented in patents like US7345195, utilized 3,5-dihydroxybenzoic acid but required extreme reaction conditions, specifically maintaining carbon dioxide pressures as high as 80MPa in glycerin solvents. These high-pressure environments necessitate the use of expensive, heavy-duty autoclaves and rigorous safety protocols, which significantly inflate the operational costs and extend the lead time for high-purity pharmaceutical intermediates. Moreover, the harsh conditions often lead to side reactions and decomposition, resulting in suboptimal yields, as evidenced by comparative data showing net yields hovering around 40% in traditional high-pressure setups. Such inefficiencies create substantial supply chain vulnerabilities, making it difficult for procurement managers to forecast costs accurately or guarantee delivery schedules for downstream polymer manufacturers.
The Novel Approach
In stark contrast, the methodology outlined in CN101875606A revolutionizes the production landscape by utilizing an alkali metal formate melt as the reaction medium under normal atmospheric pressure. This approach eliminates the need for dangerous high-pressure equipment, allowing the reaction to proceed smoothly at temperatures between 150°C and 300°C with a continuous flow of CO2. The use of formate salts, such as potassium formate or sodium formate, serves a dual purpose: acting as a solvent to dissolve the reactants and facilitating the carboxylation mechanism through effective heat transfer and ionic interaction. This modification results in a dramatic improvement in process efficiency, with experimental data indicating net yields exceeding 90% and HPLC purities reaching upwards of 99.6%. By simplifying the reaction engineering requirements, this novel approach offers a viable path for the commercial scale-up of complex polymer additives, ensuring that manufacturers can meet the growing demand for advanced materials like modified DH-PBO fibers without compromising on safety or economic viability.
Mechanistic Insights into Formate-Mediated Kolbe-Schmitt Carboxylation
The core of this technological breakthrough lies in the unique mechanistic role played by the alkali metal formate solvent during the Kolbe-Schmitt carboxylation process. Unlike traditional organic solvents or glycerin, the molten formate salt creates a highly polar ionic environment that stabilizes the phenoxide intermediate formed from the deprotonation of 3,5-dihydroxybenzoic acid by the alkali metal carbonate. This stabilization is critical for directing the electrophilic attack of carbon dioxide to the ortho-positions relative to the hydroxyl groups, ensuring the specific formation of the 2,6-disubstituted product rather than unwanted isomers. The elevated temperature range of 160°C to 270°C ensures that the formate remains in a liquid state, providing excellent mass transfer properties that allow CO2 to penetrate the reaction mixture effectively without the need for mechanical pressurization. Furthermore, the presence of the carbonate base facilitates the regeneration of the active phenoxide species, creating a catalytic cycle that drives the reaction to completion with minimal byproduct formation. This precise control over the reaction microenvironment is what enables the exceptional selectivity and yield observed in the patent examples, distinguishing it from less controlled high-pressure alternatives.
Impurity control is another critical aspect where this mechanism excels, particularly concerning the suppression of polycondensation side reactions that can occur at excessive temperatures. The patent specifies a strict upper temperature limit of 300°C, beyond which the 2,6-dihydroxybenzoic acid intermediates may undergo undesirable condensation, leading to polymeric tars that are difficult to separate. By maintaining the reaction within the optimal window of 160°C to 270°C, the process minimizes thermal degradation while maximizing the conversion of the starting material. Additionally, the subsequent workup procedure involves a sophisticated pH-controlled precipitation strategy, where the crude salt is first precipitated at a mildly acidic pH of 5-6 to remove bulk inorganic salts, followed by a second acidification to pH 1-3 to isolate the final free acid. This multi-stage purification leverages the differential solubility of the product and impurities at varying pH levels, ensuring that the final 2,6-dihydroxyterephthalic acid meets the stringent purity specifications required for high-end optical and electronic applications.
How to Synthesize 2,6-Dihydroxyterephthalic Acid Efficiently
The practical implementation of this synthesis route involves a straightforward yet precisely controlled sequence of mixing, heating, and purification steps that can be easily adapted for pilot or commercial scale operations. The process begins with the charging of 3,5-dihydroxybenzoic acid and the alkali metal formate solvent into a reaction vessel, followed by the addition of the carbonate base, after which CO2 is introduced at a controlled flow rate. Maintaining the temperature within the specified range is paramount to ensuring the solvent remains molten and the reaction kinetics favor the desired carboxylation. Following the reaction period, the mixture undergoes a carefully designed isolation protocol involving dissolution in hot sulfite solutions, decolorization with activated carbon, and sequential acid precipitation to achieve the target purity. For R&D teams looking to replicate or optimize this process, the detailed standardized synthesis steps are provided below to ensure reproducibility and safety.
- Mix 3,5-dihydroxybenzoic acid with alkali metal formate (solvent) and alkali metal carbonate under normal pressure.
- Introduce CO2 gas flow and maintain reaction temperature between 150-300°C for 1-8 hours.
- Separate the crude alkali metal salt, followed by acid precipitation and refinement to obtain pure 2,6-dihydroxyterephthalic acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this ambient pressure synthesis route offers transformative benefits that extend far beyond simple chemical yield improvements. The elimination of high-pressure reactors fundamentally alters the cost structure of production, removing the need for specialized, high-maintenance equipment that typically commands a premium in both capital investment and operational upkeep. This shift allows for a more agile manufacturing setup where production capacity can be scaled linearly without the exponential cost increases associated with high-pressure engineering constraints. Consequently, this leads to substantial cost savings in the overall manufacturing budget, enabling more competitive pricing strategies for downstream customers in the polymer and pharmaceutical sectors. The robustness of the process also means fewer unplanned downtimes due to equipment failure or safety incidents, ensuring a more reliable supply of critical intermediates for global clients.
- Cost Reduction in Manufacturing: The removal of high-pressure requirements significantly lowers the barrier to entry for production facilities, as standard glass-lined or stainless steel reactors can be utilized instead of custom-built autoclaves. This reduction in equipment complexity translates directly into lower depreciation costs and reduced maintenance expenditures over the lifecycle of the plant. Furthermore, the high yield achieved (>90%) minimizes raw material waste, ensuring that every kilogram of 3,5-dihydroxybenzoic acid is converted efficiently into valuable product, thereby optimizing the cost per unit of output. The use of common, inexpensive reagents like formates and carbonates further contributes to a leaner bill of materials, enhancing the overall margin profile for manufacturers adopting this technology.
- Enhanced Supply Chain Reliability: Operating under normal pressure drastically improves the safety profile of the facility, reducing the regulatory burden and insurance costs associated with handling high-pressure gases. This safety advantage ensures continuous operation without the frequent inspections and shutdowns required for high-pressure systems, leading to more consistent delivery schedules for customers. Additionally, the raw materials required for this process are commodity chemicals with stable global supply chains, mitigating the risk of shortages that often plague specialized fluorinated precursors used in older methods. This stability allows supply chain heads to plan long-term procurement strategies with greater confidence, knowing that the input stream is secure and resilient against market volatility.
- Scalability and Environmental Compliance: The simplified reaction conditions facilitate easier scale-up from laboratory benchtop to multi-ton commercial production, as heat and mass transfer issues are less pronounced in a molten salt system compared to viscous high-pressure slurries. From an environmental perspective, the process generates less hazardous waste and avoids the use of toxic solvents, aligning with increasingly stringent global regulations on green chemistry and emissions. The ability to recycle the formate solvent and the efficient use of CO2 as a reagent further enhance the sustainability credentials of the process, making it an attractive option for companies aiming to reduce their carbon footprint while maintaining high production volumes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of 2,6-dihydroxyterephthalic acid, derived directly from the insights provided in patent CN101875606A. These answers are designed to clarify the operational advantages and chemical specifics for technical decision-makers evaluating this technology for integration into their supply chains. Understanding these nuances is essential for assessing the feasibility of adopting this method for large-scale manufacturing of high-performance polymer precursors.
Q: What are the primary advantages of the formate melt method over traditional high-pressure synthesis?
A: The formate melt method operates at normal pressure, eliminating the need for expensive high-pressure autoclaves (80MPa) required in traditional glycerin-based methods, thereby significantly reducing equipment costs and safety risks while improving yield from ~40% to over 90%.
Q: How is high purity achieved in the final 2,6-dihydroxyterephthalic acid product?
A: High purity (>99%) is achieved through a dual-stage purification process involving initial precipitation at pH 5-6 to remove bulk impurities, followed by redissolution and precise acidification to pH 1-3, often utilizing activated carbon decolorization steps.
Q: Is this synthesis route scalable for industrial production of polymer intermediates?
A: Yes, the process uses readily available raw materials like 3,5-dihydroxybenzoic acid and common inorganic salts, and the ambient pressure conditions make it highly suitable for large-scale commercial manufacturing without complex engineering controls.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,6-Dihydroxyterephthalic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of next-generation materials, and we are uniquely positioned to support your production needs with our advanced manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements whether you are in the pilot phase or full-scale manufacturing. We adhere to stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of 2,6-dihydroxyterephthalic acid meets the exacting standards required for high-performance polymer and pharmaceutical applications. Our commitment to quality and consistency makes us a trusted partner for global enterprises seeking to secure their supply chains against market fluctuations.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits of switching to this ambient pressure method for your specific application. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive efficiency and profitability in your operations. Let us collaborate to bring your high-performance material projects to fruition with speed, precision, and reliability.
