Advanced Synthesis of 6-Halo-Caproic Acid: Scalable Routes for Global Pharmaceutical Supply Chains
The global demand for high-purity halogenated fatty acid intermediates continues to surge, driven by their critical role in the synthesis of complex pharmaceuticals and agrochemicals. Patent CN102633619A introduces a robust and economically viable preparation method for 6-halo-caproic acid, specifically addressing the longstanding challenges of selectivity and scalability in this chemical class. By utilizing 1,6-hexanediol as a primary feedstock, this technology offers a streamlined pathway that bypasses the need for exotic reagents or extreme reaction conditions. For R&D directors and procurement specialists alike, this represents a significant opportunity to optimize supply chains for key intermediates used in polymer additives, dye precursors, and medicinal chemistry. The methodology described herein not only simplifies the synthetic route but also inherently reduces environmental impact through efficient solvent management and high atom economy.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for generating mono-halogenated long-chain alcohols and acids often suffer from poor regioselectivity, leading to significant quantities of di-halogenated byproducts that are difficult and costly to separate. In many legacy processes, the use of homogeneous acid catalysts without effective phase separation results in over-halogenation, where both hydroxyl groups of the diol are substituted, drastically lowering the yield of the desired mono-substituted intermediate. Furthermore, conventional oxidation steps frequently rely on stoichiometric amounts of heavy metal oxidants or harsh acidic conditions that generate substantial hazardous waste streams, complicating downstream processing and increasing disposal costs. These inefficiencies create bottlenecks in manufacturing, leading to inconsistent batch quality and extended lead times for final API production. The inability to effectively isolate the mono-halo intermediate often necessitates complex purification protocols such as repeated crystallization or chromatography, which are impractical for multi-ton commercial scale-up.
The Novel Approach
The innovative strategy outlined in the patent data leverages a clever solvent engineering approach to achieve high selectivity during the initial halogenation step. By employing toluene as the reaction medium, the process exploits the differential solubility of the reactants and products; the starting 1,6-hexanediol and hydrogen halide remain largely in the aqueous phase or interface, while the generated 6-halo-hexanol preferentially dissolves in the toluene layer. This in-situ extraction mechanism effectively removes the product from the reactive environment, thereby kinetically suppressing the second halogenation event and ensuring high purity of the mono-substituted alcohol. Following this, the oxidation step utilizes a crown ether-catalyzed system with potassium permanganate in dichloromethane, offering a milder and more controlled oxidation profile compared to traditional strong acid oxidations. This two-stage sequence ensures that the structural integrity of the carbon chain is maintained while efficiently converting the terminal hydroxyl group to a carboxylic acid functionality.
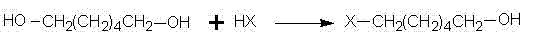
The first stage of the synthesis involves the reaction of 1,6-hexanediol with a hydrogen halide (HX), where X represents either chlorine or bromine, conducted under reflux conditions between 80°C and 100°C. The molar ratio of diol to hydrogen halide is carefully controlled between 1:1 and 1:1.5 to further minimize the risk of di-substitution while ensuring complete conversion of the limiting reagent. The use of toluene serves a dual purpose: it acts as a solvent for the organic product and functions similarly to a phase-transfer agent by sequestering the 6-halo-hexanol away from the aqueous acid layer. After the reaction reaches completion, typically within 6 to 9 hours, the solvent is removed under normal pressure, and the crude product is washed and distilled under reduced pressure to isolate pure 6-halo-hexanol. This precise control over reaction parameters and phase behavior is critical for achieving the high purity required for subsequent pharmaceutical applications.
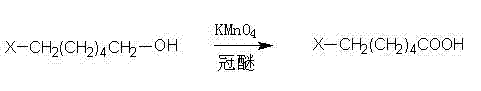
In the second transformative stage, the isolated 6-halo-hexanol undergoes oxidation to form the target 6-halo-caproic acid. This step is performed in dichloromethane using potassium permanganate as the oxidant, facilitated by a catalytic amount of crown ether. The crown ether complexes with the potassium cation, solubilizing the permanganate anion in the organic phase and significantly enhancing its oxidative power towards the primary alcohol. The oxidant is added in small batches to manage the exothermic nature of the reaction and prevent over-oxidation or degradation of the halo-alkyl chain. Reaction progress is monitored via thin-layer chromatography (TLC) to ensure optimal endpoint determination. Upon completion, the mixture is filtered to remove manganese dioxide byproducts, and the organic phase is washed with alkali to extract the carboxylic acid as its water-soluble salt. Finally, acidification of the aqueous layer precipitates the pure 6-halo-caproic acid, which is then dried to yield the final commercial product.
Mechanistic Insights into Selective Halogenation and Crown Ether Catalysis
The core mechanistic advantage of this process lies in the thermodynamic and kinetic control exerted during the halogenation phase. In a standard homogeneous system, the statistical probability of reacting with either hydroxyl group is equal, and once one group reacts, the electronic effects might even favor the second substitution. However, by introducing toluene, the system creates a dynamic equilibrium where the product is continuously partitioned into the organic layer. This physical separation acts as a driving force, shifting the equilibrium towards the mono-substituted product according to Le Chatelier's principle. The solubility difference is the key discriminator; since 1,6-hexanediol is highly water-soluble and 6-halo-hexanol is lipophilic, the toluene layer essentially acts as a 'product sink.' This eliminates the need for protecting group chemistry, which would otherwise add at least two additional synthetic steps (protection and deprotection), thereby reducing overall process mass intensity and cost.
Regarding impurity control, the oxidation step utilizing the crown ether/potassium permanganate system offers superior selectivity for primary alcohols over secondary positions or sensitive functional groups like the alkyl halide moiety. Traditional oxidants such as chromic acid or nitric acid can be overly aggressive, potentially leading to chain cleavage or elimination reactions that generate olefinic impurities. The mild basicity of the permanganate/crown ether system preserves the carbon-halogen bond, which is crucial for the downstream utility of the molecule in nucleophilic substitution reactions. Furthermore, the workup procedure involving alkaline extraction effectively separates the desired carboxylic acid from any unreacted alcohol or neutral byproducts, as only the acid forms a water-soluble salt. This inherent purification capability during the workup phase significantly reduces the burden on final crystallization steps, ensuring a cleaner impurity profile for the final API intermediate.
How to Synthesize 6-Halo-Caproic Acid Efficiently
To implement this synthesis effectively in a pilot or production plant, operators must strictly adhere to the temperature profiles and addition rates specified in the patent embodiments to ensure safety and reproducibility. The initial halogenation requires efficient water removal, often achieved via a Dean-Stark trap or similar azeotropic distillation setup, to drive the reaction to completion and prevent hydrolysis of the formed alkyl halide. Detailed standardized operating procedures for the batch-wise addition of oxidant and the subsequent pH adjustments during workup are essential for maintaining consistent quality across different production scales.
- Perform selective mono-halogenation of 1,6-hexanediol using hydrogen halide in toluene solvent at reflux temperatures (80-100°C) to yield 6-halo-hexanol.
- Oxidize the resulting 6-halo-hexanol using potassium permanganate in dichloromethane with a crown ether catalyst to form the corresponding carboxylic acid salt.
- Acidify the aqueous phase with mineral acid to precipitate the final 6-halo-caproic acid product, followed by drying and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible benefits in terms of cost stability and raw material security. The reliance on 1,6-hexanediol, a commodity chemical produced on a massive global scale, ensures a stable and competitive supply base, mitigating the risks associated with sourcing niche or proprietary starting materials. The elimination of complex protecting group strategies and expensive transition metal catalysts translates directly into reduced raw material costs and simplified inventory management. Moreover, the use of common industrial solvents like toluene and dichloromethane facilitates easy solvent recovery and recycling, further driving down the variable costs per kilogram of the final product. This process efficiency allows manufacturers to offer more competitive pricing structures without compromising on the stringent purity specifications required by regulated industries.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by removing the need for precious metal catalysts and complex multi-step protection sequences. By utilizing inexpensive reagents like hydrogen halides and potassium permanganate, the direct material cost is drastically lowered compared to routes requiring palladium or platinum. Additionally, the high selectivity of the reaction minimizes the formation of hard-to-remove byproducts, reducing the loss of yield during purification and maximizing the output from each batch of raw materials.
- Enhanced Supply Chain Reliability: Sourcing reliability is greatly improved as the key feedstock, 1,6-hexanediol, is widely available from multiple global suppliers, reducing dependency on single-source vendors. The robustness of the reaction conditions, which do not require cryogenic temperatures or ultra-high pressures, means that production can be maintained consistently across different manufacturing sites with standard equipment. This flexibility ensures continuous supply availability even during market fluctuations or logistical disruptions affecting specialized reagents.
- Scalability and Environmental Compliance: The methodology is inherently designed for scale-up, utilizing unit operations such as reflux, distillation, and filtration that are standard in fine chemical plants. The reduction in hazardous waste generation, particularly by avoiding heavy metal contaminants and minimizing di-halo byproducts, simplifies compliance with increasingly strict environmental regulations. This 'green' aspect of the process not only lowers waste disposal costs but also aligns with the sustainability goals of major pharmaceutical and agrochemical customers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 6-halo-caproic acid derivatives based on the patented technology. Understanding these details helps stakeholders evaluate the feasibility of integrating this intermediate into their specific value chains.
Q: How does this method prevent the formation of di-halo byproducts?
A: The process utilizes toluene as a solvent which creates a biphasic system. Since the mono-halo product is soluble in toluene while the starting diol and hydrogen halide are water-soluble, the product is effectively extracted into the organic phase as it forms, preventing further reaction to the di-halo species.
Q: What are the advantages of using Potassium Permanganate with Crown Ether for oxidation?
A: This combination allows for efficient oxidation under mild conditions in an organic solvent (dichloromethane). The crown ether acts as a phase-transfer catalyst, enhancing the reactivity of the permanganate ion in the organic phase, leading to high conversion rates without requiring harsh acidic conditions typical of other oxidants.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the patent explicitly highlights the method's suitability for large-scale production due to simple operational steps, readily available raw materials like 1,6-hexanediol, and the use of standard solvents like toluene and dichloromethane which are easily recovered and recycled in industrial settings.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6-Halo-Caproic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of next-generation therapeutics and crop protection agents. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale discovery to full-scale manufacturing is seamless and efficient. We are committed to delivering 6-halo-caproic acid and related derivatives with stringent purity specifications, supported by our rigorous QC labs that employ advanced analytical techniques to verify every batch. Our facility is equipped to handle the specific solvent systems and reaction conditions required by this patent, guaranteeing a consistent supply of material that meets the exacting standards of the global pharmaceutical industry.
We invite you to collaborate with us to leverage this advanced synthesis technology for your upcoming projects. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing processes can enhance your supply chain efficiency and reduce your overall cost of goods sold.
