Scalable Rhodium-Catalyzed Synthesis of Complex 3,3-Spiro-Oxindole Scaffolds for Oncology Applications
Scalable Rhodium-Catalyzed Synthesis of Complex 3,3-Spiro-Oxindole Scaffolds for Oncology Applications
The pharmaceutical industry is constantly seeking efficient pathways to access complex heterocyclic scaffolds that serve as privileged structures in drug discovery. Patent CN103554120A discloses a groundbreaking preparation method for 3,3-spiro(2-tetrahydrofuran)oxindole polycyclic compounds, which represent a highly valuable class of intermediates for developing novel therapeutics. These specific molecular architectures are not merely academic curiosities; they form the core backbone of numerous bioactive natural products and synthetic pharmaceuticals, including potent progesterone receptor inhibitors and antihypertensive agents. The innovation detailed in this patent provides a robust chemical platform for generating diverse compound libraries aimed at inhibiting Aurora Kinase A (AURKA), a pivotal enzyme in cell mitosis whose dysregulation is linked to various cancers. By leveraging a sophisticated rhodium-catalyzed cascade reaction, this technology overcomes historical synthetic bottlenecks, offering a streamlined route to high-value pharmaceutical intermediates that were previously difficult to access with high stereocontrol and efficiency.
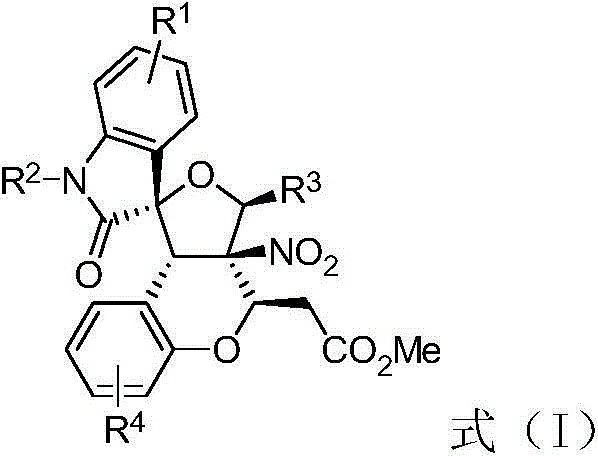
The strategic importance of this synthesis lies in its ability to construct significant molecular complexity rapidly. The general structure, as depicted in the patent documentation, allows for extensive derivatization at multiple positions (R1, R2, R3, and R4), enabling medicinal chemists to fine-tune pharmacokinetic properties and binding affinity. For R&D directors focused on oncology pipelines, access to such a versatile scaffold is crucial for structure-activity relationship (SAR) studies. The presence of the spiro-oxindole motif combined with the tetrahydrofuran ring creates a rigid three-dimensional framework that often enhances binding specificity to protein targets. Furthermore, the patent highlights that these compounds exhibit strong inhibition against AURKA, suggesting their potential utility as lead compounds for next-generation anti-cancer drugs. This positions the technology not just as a synthetic method, but as a gateway to new intellectual property in the competitive field of kinase inhibitors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the construction of 3,3-spiro(2-tetrahydrofuran)oxindole frameworks was fraught with significant challenges that hindered both research and commercial production. Historical methods, such as those reported by Professor Muthusamy in 2004 and the Schreiber group in 2007, relied on multi-step sequences that were often inefficient and operationally cumbersome. These conventional approaches typically suffered from low substrate universality, meaning they failed to accommodate a broad range of functional groups, thereby limiting the chemical diversity accessible to researchers. Moreover, the reaction conditions required were frequently harsh, involving extreme temperatures or sensitive reagents that posed safety risks and increased the complexity of process control. Such limitations resulted in poor overall yields and generated substantial chemical waste, making these routes economically unviable for large-scale API manufacturing. The inability to efficiently establish multiple chiral centers in a single operation also necessitated costly resolution steps or asymmetric syntheses that further drove up production costs and extended lead times.
The Novel Approach
In stark contrast, the methodology described in patent CN103554120A introduces a paradigm shift by utilizing a one-pot tandem reaction strategy that dramatically simplifies the synthetic landscape. This novel approach employs a rhodium acetate catalyst to facilitate a cascade sequence that constructs five distinct ring systems and five chiral carbon centers simultaneously. This level of complexity generation in a single operational step is rare in organic synthesis and represents a significant leap forward in atom economy. The process operates under mild conditions, typically at room temperature, which eliminates the need for energy-intensive heating or cooling protocols. By integrating the formation of the carbonyl ylide, the [3+2] cycloaddition, and the subsequent intramolecular Michael addition into a continuous flow of chemical transformations, the method minimizes intermediate isolation and purification steps. This consolidation of synthetic operations not only accelerates the timeline from raw materials to final product but also significantly reduces the consumption of solvents and reagents, aligning perfectly with the principles of green chemistry and sustainable fine chemical manufacturing.
Mechanistic Insights into Rhodium-Catalyzed Cascade Cyclization
The core of this technological breakthrough lies in the elegant mechanistic pathway driven by the dirhodium tetraacetate catalyst. The reaction initiates with the decomposition of the isatin diazo compound by the rhodium catalyst, generating a highly reactive metal-carbenoid species. This intermediate undergoes a rapid transformation to form a carbonyl ylide, a dipolar species that serves as the key building block for the subsequent ring-forming events. The carbonyl ylide then engages in a highly stereoselective [3+2] cycloaddition with an ortho-substituted phenylnitroalkene, which acts as the dipolarophile. This step is critical as it establishes the initial spiro-oxindole core and sets the stereochemistry for the adjacent chiral centers. The precision of this cycloaddition is governed by the electronic and steric properties of the catalyst and the substrates, ensuring high diastereoselectivity as evidenced by the d.r. values reported in the experimental data. Following the cycloaddition, the addition of a base such as DBU triggers an intramolecular Michael addition. This final cyclization event closes the fifth ring, locking the molecular architecture into the desired polycyclic conformation and completing the construction of the complex scaffold.
Understanding the impurity profile is essential for scaling this chemistry, and the mechanism offers inherent advantages in this regard. Because the reaction proceeds through a concerted cascade, there are fewer opportunities for side reactions that typically plague stepwise syntheses. The use of molecular sieves in the reaction mixture helps to scavenge water, preventing the hydrolysis of the sensitive diazo intermediate and ensuring high conversion rates. Furthermore, the mild reaction conditions minimize thermal degradation of the product or intermediates, leading to a cleaner crude reaction mixture. This simplifies the downstream purification process, often requiring only standard column chromatography to achieve high purity levels suitable for biological testing. For process chemists, this means a more predictable and controllable manufacturing process with reduced risk of batch-to-batch variability. The ability to tolerate a wide range of substituents on the aldehyde and nitroalkene components without compromising the reaction efficiency further underscores the robustness of this catalytic system, making it an ideal candidate for generating diverse libraries of high-purity pharmaceutical intermediates.
How to Synthesize 3,3-Spiro-Oxindole Derivatives Efficiently
The practical implementation of this synthesis is designed for ease of execution while maintaining high standards of quality and reproducibility. The protocol begins with the preparation of a reaction mixture containing the aldehyde, the ortho-substituted phenylnitroalkene, the rhodium acetate catalyst, and activated molecular sieves in a suitable organic solvent such as dichloromethane. The use of molecular sieves is a critical detail that ensures the anhydrous conditions necessary for the stability of the diazo species. A solution of the isatin diazo compound is then prepared separately and added slowly to the reaction vessel via a peristaltic pump. This controlled addition rate is vital to manage the concentration of the reactive carbenoid intermediate, preventing dimerization or other non-productive decomposition pathways. Once the addition is complete, typically over one hour, the reaction is allowed to stir to ensure full conversion to the cycloaddition intermediate. Subsequently, a base like DBU is introduced to the system to promote the final ring-closing step. The entire process is conducted at ambient temperature, eliminating the need for specialized heating or cooling equipment and reducing energy consumption.
- Prepare a reaction mixture containing aldehyde, ortho-substituted phenylnitroalkene, rhodium acetate catalyst, and molecular sieves in an organic solvent such as dichloromethane.
- Slowly add a solution of isatin diazo compound to the reaction mixture at room temperature over a period of approximately one hour to control the generation of the reactive intermediate.
- Add a base such as DBU to the mixture to induce intramolecular Michael addition, followed by solvent removal and column chromatography to isolate the pure polycyclic product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this synthesis method offers compelling economic and logistical benefits that directly impact the bottom line. The primary driver of cost efficiency is the drastic reduction in process steps. Traditional routes to similar polycyclic structures often require multiple isolation and purification stages, each adding labor, time, and material costs. By consolidating these into a single one-pot operation, the new method significantly lowers the operational expenditure associated with manufacturing. Additionally, the raw materials required—isatin derivatives, various aldehydes, and nitroalkenes—are commodity chemicals that are readily available from multiple global suppliers. This abundance ensures a stable supply chain and mitigates the risk of raw material shortages or price volatility. The high atom economy of the reaction means that a larger proportion of the starting mass is incorporated into the final product, reducing waste disposal costs and improving the overall material efficiency of the process. These factors combine to create a manufacturing profile that is both cost-effective and resilient.
- Cost Reduction in Manufacturing: The elimination of intermediate isolation steps and the use of low catalyst loading (1-2 mol%) substantially reduce the consumption of solvents and reagents. This streamlined approach minimizes the physical footprint required for production and lowers the utility costs associated with heating, cooling, and vacuum operations. Furthermore, the simplified workup procedure reduces the labor hours needed per batch, allowing for higher throughput with existing infrastructure. The avoidance of expensive transition metal removal steps, often required in other catalytic processes, further contributes to significant cost savings in the final purification stage.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials ensures that production schedules are not held hostage by niche reagent lead times. The robustness of the reaction conditions, which tolerate a variety of functional groups and operate at room temperature, reduces the likelihood of batch failures due to minor fluctuations in process parameters. This reliability translates to more consistent delivery times for customers and a lower risk of supply disruption. The scalability of the method from milligram to kilogram scales without significant re-optimization further supports a agile supply chain capable of responding quickly to changing market demands.
- Scalability and Environmental Compliance: The green chemistry attributes of this process, including high atom economy and reduced solvent usage, align with increasingly stringent environmental regulations. The mild reaction conditions reduce the energy intensity of the process, contributing to a lower carbon footprint. The generation of less chemical waste simplifies effluent treatment and reduces the environmental burden of manufacturing. These sustainability credentials are becoming a key differentiator in the pharmaceutical supply chain, where partners are increasingly prioritizing environmentally responsible vendors. The ability to scale this process to commercial quantities while maintaining these green standards positions it as a future-proof solution for long-term production needs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of these 3,3-spiro(2-tetrahydrofuran)oxindole compounds. The answers are derived directly from the technical specifications and experimental data provided in the patent literature, ensuring accuracy and relevance for industry professionals. Understanding these details is crucial for evaluating the feasibility of integrating this technology into your own R&D or manufacturing workflows. Whether you are concerned about the specific biological activity, the scalability of the reaction, or the availability of precursors, the information below provides a comprehensive overview based on the documented evidence.
Q: What are the primary structural advantages of this synthesis method?
A: This novel methodology enables the simultaneous construction of five rings and five chiral centers in a single operational step, significantly streamlining the synthesis of complex spiro-oxindole architectures compared to traditional multi-step sequences.
Q: What is the biological significance of the resulting compounds?
A: The synthesized 3,3-spiro(2-tetrahydrofuran)oxindole derivatives have demonstrated potent inhibitory activity against Aurora Kinase A (AURKA), a critical target in cancer therapy for disrupting cell division and inducing tumor cell apoptosis.
Q: Are the raw materials for this process commercially viable?
A: Yes, the process utilizes readily available and cost-effective starting materials including various isatin diazo derivatives, common aldehydes, and nitroalkenes, ensuring a robust and sustainable supply chain for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,3-Spiro-Oxindole Supplier
The development of complex oncology therapeutics requires a partner who understands both the scientific intricacies and the commercial realities of pharmaceutical manufacturing. NINGBO INNO PHARMCHEM stands ready to support your pipeline with our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific requirements of rhodium-catalyzed reactions, ensuring that the high purity specifications and rigorous QC labs standards demanded by the global pharmaceutical industry are consistently met. We recognize that the transition from laboratory discovery to commercial supply is a critical phase, and our team of expert process chemists is dedicated to optimizing this specific synthetic route to maximize yield and minimize cost for your specific application.
We invite you to discuss how this advanced synthesis technology can accelerate your drug discovery programs. Our technical procurement team is available to provide a Customized Cost-Saving Analysis tailored to your volume requirements and quality specifications. By partnering with us, you gain access not only to high-quality intermediates but also to deep technical expertise that can help de-risk your supply chain. Please contact us today to request specific COA data and route feasibility assessments for your next project, and let us demonstrate how our commitment to innovation and quality can drive value for your organization.
