Scalable Manufacturing of Glutamine and Asparagine Cyclic Dipeptides for Global Pharma
Scalable Manufacturing of Glutamine and Asparagine Cyclic Dipeptides for Global Pharma
Introduction to Novel Cyclic Dipeptide Synthesis Technology
The pharmaceutical and biotechnology sectors are constantly seeking robust methods for producing bioactive cyclic peptides, specifically 2,5-diketopiperazines, which exhibit superior metabolic stability and potent biological activities ranging from antiviral to antitumor effects. Patent CN111875668A introduces a groundbreaking synthetic methodology specifically designed for cyclic dipeptides containing L-asparagine or L-glutamine sequences, addressing long-standing challenges in peptide cyclization efficiency. This technology leverages a hybrid solid-phase and liquid-phase strategy that significantly enhances the feasibility of manufacturing these complex intermediates at a commercial scale. By utilizing 2-Chlorotrityl Chloride Resin as a solid support, the process ensures high loading efficiency and mild cleavage conditions, which are critical for preserving the integrity of sensitive amino acid side chains. The subsequent transformation involves a carefully orchestrated sequence of methyl esterification, aqueous alkaline cyclization, and final aminolysis, resulting in target molecules with exceptional purity profiles. This innovation represents a substantial leap forward for reliable pharmaceutical intermediate supplier networks aiming to diversify their portfolio with high-value cyclic peptide scaffolds.
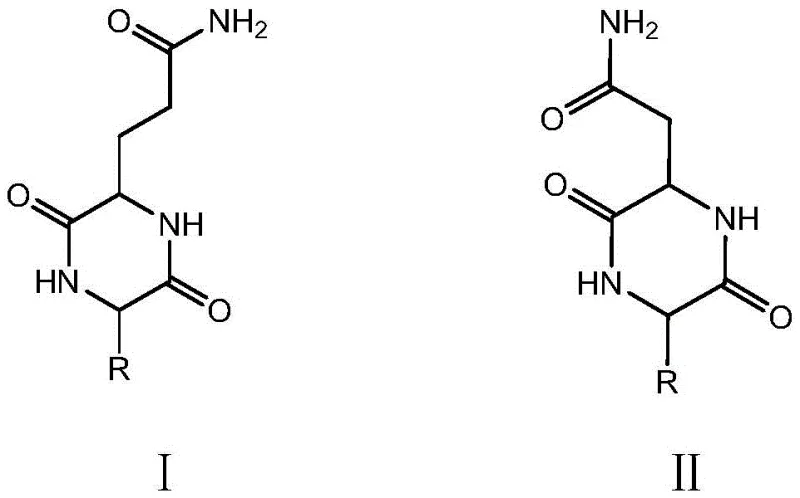
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for cyclic dipeptides containing amide side chains like glutamine and asparagine have historically been plagued by significant technical bottlenecks that hinder commercial viability. Conventional solid-phase methods often rely on expensive linker resins such as Oxime-Resin or amino resins, which not only drive up raw material costs but also introduce complex cleavage steps that can compromise overall yield. Furthermore, liquid-phase strategies typically employ maximum protection schemes involving bulky protecting groups like trityl on the side chains to prevent unwanted side reactions. However, these large steric groups create severe spatial hindrance during the critical cyclization step, frequently leading to incomplete ring closure and the formation of linear oligomers or other difficult-to-remove impurities. The necessity for extensive column chromatography purification in these traditional methods further exacerbates production costs and extends lead times, making them less attractive for high-volume manufacturing. Additionally, the use of excessive organic solvents in liquid-phase cyclization raises environmental compliance concerns and increases the burden on waste treatment facilities, creating a less sustainable supply chain profile for cost reduction in pharmaceutical intermediate manufacturing.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in the patent data utilizes a streamlined workflow that effectively bypasses steric hindrance issues while maintaining high operational safety and simplicity. The core innovation lies in the use of 2-Chlorotrityl Chloride Resin to synthesize linear dipeptide fragments followed by a liquid-phase methyl esterification step that converts carboxylic acids into methyl esters without requiring bulky side-chain protection. This strategic modification allows for a subsequent aqueous alkaline cyclization, where the reduced steric bulk facilitates efficient head-to-tail amide bond formation to create the stable six-membered diketopiperazine ring. The use of an aqueous medium for cyclization not only eliminates the need for large volumes of hazardous organic solvents but also leverages the dilution effect to suppress intermolecular polymerization by-products. This method has been proven to successfully synthesize various sequences, including Cyclo[Glu(OMe)-AA] and Cyclo[Asp(OMe)-AA] precursors, which are then converted to the final glutamine or asparagine products via aminolysis. The result is a process that offers superior cyclization efficiency, easier purification through simple filtration or crystallization, and a significantly greener chemical footprint.
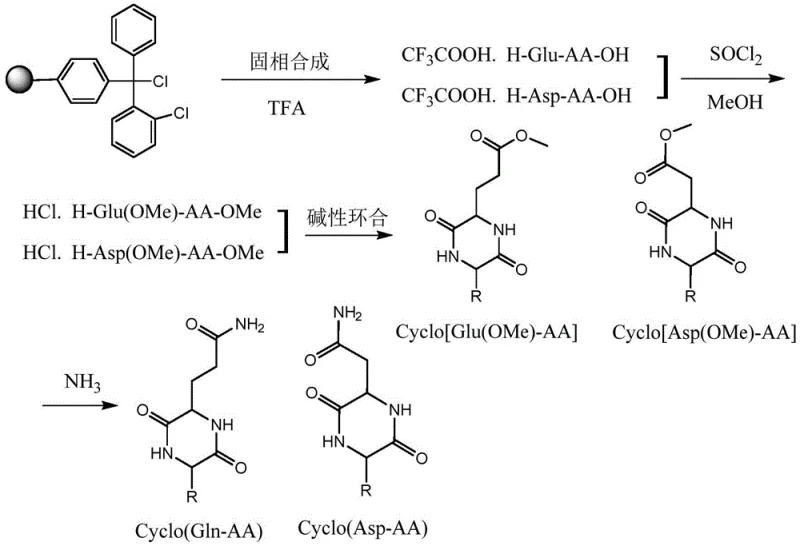
Mechanistic Insights into Aqueous Alkaline Cyclization and Aminolysis
The mechanistic elegance of this synthesis lies in the precise control of reaction conditions during the cyclization and aminolysis phases, which are critical for ensuring high purity and yield. During the aqueous alkaline cyclization step, the linear dimethyl ester dipeptide hydrochloride is dissolved in water and treated with an inorganic base such as sodium bicarbonate to adjust the pH to a neutral or slightly alkaline range of 7 to 8. Under these conditions, the free amino group performs a nucleophilic attack on the ester carbonyl carbon, preferentially forming a stable conformation that leads to the intramolecular closure of the 2,5-diketopiperazine ring. The aqueous environment plays a dual role: it solvates the inorganic salts generated during the reaction, allowing for their easy removal, and it minimizes the solubility of the hydrophobic cyclic products, often causing them to precipitate directly from the solution for easy filtration. For more hydrophilic sequences, the product remains in solution but can be isolated through concentration and solvent exchange, demonstrating the versatility of this mechanism across different amino acid side chains. This controlled cyclization effectively avoids the formation of linear dimers or higher oligomers that are common in concentrated organic solvent systems, thereby simplifying the downstream purification process significantly.
Following the formation of the methyl ester-protected cyclic dipeptide, the final transformation involves an aminolysis reaction to convert the side-chain esters into the desired amide functionalities of glutamine or asparagine. This step is conducted using a solution of ammonia in methanol or 1,4-dioxane, which serves to dissolve the cyclic intermediate while providing a high concentration of nucleophilic ammonia. The reaction mechanism involves the nucleophilic substitution of the methoxy group by the amino group, driven by the equilibrium shift towards the amide product. To ensure complete conversion and prevent the accumulation of partially reacted intermediates, the protocol recommends a supplementary addition of ammonia water after an initial reaction period, which maintains the driving force for the reaction to proceed to completion. This high-efficiency aminolysis ensures that the final crude product contains minimal ester impurities, facilitating the subsequent purification steps such as reverse-phase chromatography or freeze-drying. The stability of the diketopiperazine ring under these ammonolysis conditions is a key factor, as it prevents ring-opening side reactions that could otherwise degrade the target molecule, ensuring the structural integrity of the high-purity pharmaceutical intermediate.
How to Synthesize Cyclic Dipeptides Efficiently
The synthesis of these complex cyclic structures requires a disciplined adherence to the four-step protocol outlined in the patent to ensure reproducibility and quality. The process begins with the solid-phase assembly of the linear dipeptide on 2-Chlorotrityl Chloride Resin, followed by cleavage to obtain the trifluoroacetate salt, which is then subjected to methyl esterification using thionyl chloride in methanol at controlled low temperatures. The resulting diester is then cyclized in an aqueous phase using inorganic bases, and finally, the side-chain esters are converted to amides via ammonolysis. This sequence is designed to maximize yield while minimizing the formation of difficult-to-remove impurities. For detailed operational parameters, reagent ratios, and specific purification techniques required to replicate this high-efficiency synthesis, please refer to the standardized technical guide provided below.
- Synthesize linear dipeptide trifluoroacetates using 2-Chlorotrityl Chloride Resin and Fmoc-protected amino acids via solid-phase methods.
- Perform methyl esterification on the linear dipeptide salts using thionyl chloride in methanol to generate dimethyl ester hydrochlorides.
- Execute aqueous alkaline cyclization to form the protected cyclic dipeptide, followed by aminolysis to yield the final glutamine or asparagine product.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement and supply chain perspective, this synthetic methodology offers compelling advantages that directly address the pain points of cost, reliability, and scalability in the production of specialty peptide intermediates. By eliminating the need for expensive and specialized resins like Oxime-Resin, the raw material costs are significantly reduced, allowing for more competitive pricing structures in the final supply of cyclic dipeptides. The shift to aqueous cyclization drastically simplifies the solvent recovery and waste treatment processes, leading to substantial cost savings in environmental compliance and operational overhead. Furthermore, the robustness of the reaction conditions, which tolerate a variety of amino acid side chains without requiring complex protection group strategies, enhances the flexibility of the manufacturing line to accommodate diverse product portfolios. This adaptability ensures that supply chain managers can rely on a single, versatile platform for producing multiple variants of cyclic dipeptides, thereby reducing the complexity of inventory management and vendor qualification processes. The high purity achieved through this method also reduces the burden on quality control laboratories, as fewer purification cycles are needed to meet stringent pharmaceutical specifications.
- Cost Reduction in Manufacturing: The elimination of costly linker resins and the reduction of organic solvent usage during the cyclization step lead to a direct decrease in the bill of materials and waste disposal costs. The process avoids the need for extensive column chromatography by leveraging precipitation and crystallization for isolation, which significantly lowers the operational expenditure associated with purification media and solvent consumption. Additionally, the use of common reagents like thionyl chloride and ammonia ensures that raw material procurement is straightforward and not subject to the supply volatility often seen with specialized peptide coupling agents. These factors combine to create a manufacturing process that is inherently more cost-efficient, allowing for better margin management and competitive pricing for the final high-purity pharmaceutical intermediate.
- Enhanced Supply Chain Reliability: The reliance on widely available commodity chemicals and standard solid-phase synthesis equipment ensures that the production of these cyclic dipeptides is not bottlenecked by the scarcity of niche reagents. The robustness of the aqueous cyclization step means that the process is less sensitive to minor variations in reaction conditions, leading to consistent batch-to-batch quality and reducing the risk of production failures or delays. This stability is crucial for maintaining continuous supply lines to downstream pharmaceutical customers who require just-in-time delivery of critical intermediates. By simplifying the synthesis route, the lead time for producing complex cyclic dipeptides is effectively reduced, enabling faster response to market demands and ensuring that supply chain heads can meet their delivery commitments with greater confidence and reliability.
- Scalability and Environmental Compliance: The transition from organic solvent-based cyclization to an aqueous system represents a significant improvement in the environmental profile of the manufacturing process, aligning with global trends towards greener chemistry and sustainability. This reduction in hazardous waste generation simplifies regulatory compliance and reduces the environmental footprint of the production facility, which is increasingly important for corporate social responsibility goals. The process has been demonstrated to work effectively on a batch basis, and the simplicity of the workup procedures suggests that it can be readily scaled up from laboratory to commercial production volumes without significant re-engineering. This scalability ensures that as demand for these bioactive cyclic peptides grows, the manufacturing capacity can be expanded efficiently to meet market needs while maintaining strict adherence to environmental and safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of these cyclic dipeptides, based on the detailed data provided in the patent documentation. These answers are designed to clarify the mechanistic advantages and practical benefits of this novel method for stakeholders involved in process development and procurement. Understanding these details is essential for evaluating the feasibility of integrating this technology into existing manufacturing workflows.
Q: How does this method overcome steric hindrance in cyclic dipeptide formation?
A: The method utilizes a specific sequence of methyl esterification followed by aqueous alkaline cyclization. This approach minimizes the steric bulk associated with traditional side-chain protecting groups like trityl, allowing for efficient head-to-tail amide bond formation and high cyclization yields.
Q: What are the purity levels achievable with this synthesis route?
A: The patent data demonstrates HPLC purities exceeding 93% to 99% for various derivatives such as Cyclo(Gln-Phe) and Cyclo(Asn-Pro). The aqueous workup and specific purification steps effectively remove inorganic salts and by-products.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the protocol avoids expensive resins like Oxime-Resin and utilizes common reagents such as thionyl chloride and ammonia. The aqueous cyclization step significantly reduces organic solvent consumption, making it environmentally friendly and cost-effective for scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cyclic Dipeptide Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis and manufacturing, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is uniquely qualified to adapt the advanced synthesis methods described in patent CN111875668A to meet your specific project requirements, ensuring that stringent purity specifications are met through our rigorous QC labs. We understand the critical nature of peptide intermediates in drug development and are committed to providing a seamless transition from process optimization to full-scale commercial supply. Our facility is equipped to handle the specific reagents and conditions required for solid-phase synthesis and aqueous cyclization, guaranteeing a consistent and high-quality supply of cyclic dipeptides for your pharmaceutical applications.
We invite you to engage with our technical procurement team to discuss how we can support your supply chain optimization goals through a Customized Cost-Saving Analysis. By leveraging our expertise in process chemistry, we can help you evaluate the feasibility of this route for your specific target molecules and provide specific COA data and route feasibility assessments tailored to your needs. Contact us today to request a comprehensive evaluation and discover how our manufacturing capabilities can enhance your product development timeline and reduce overall production costs.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
