Breakthrough Catalytic Strategy for High-Purity UV-1600 and UV-479 Intermediates Manufacturing
The global demand for high-performance ultraviolet (UV) stabilizers in the polymer industry continues to surge, driven by the need for durable materials in construction, photovoltaics, and automotive applications. A pivotal development in this sector is disclosed in Chinese patent CN116199638A, which details a novel preparation method for 2,4-bis(biphenyl-4-yl)-6-(2,4-dihydroxyphenyl)-1,3,5-triazine. This compound serves as the critical core intermediate for manufacturing advanced UV absorbers such as UV-1600 and UV-479. Unlike their simpler aryl-substituted counterparts, these biphenyl-based absorbers offer superior UV absorption efficiency and durability, making them essential for protecting high-transparency polycarbonate plates and films. The patent introduces a groundbreaking two-step Friedel-Crafts alkylation strategy that overcomes the longstanding selectivity and solubility challenges associated with biphenyl substrates, offering a viable pathway for industrial-scale production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2,4,6-triaryl-1,3,5-triazines has relied heavily on direct Friedel-Crafts reactions where the aromatic substrate also functions as the solvent. While this approach works efficiently for liquid aromatics like benzene or xylene, it fails catastrophically when applied to biphenyl. Biphenyl exists as a solid at room temperature with a high melting point, rendering it unusable as a reaction medium. Consequently, additional solvents must be introduced, which dilutes reactant concentration and alters reaction kinetics. More critically, biphenyl exhibits lower reactivity and weaker localization effects compared to simple benzenes. When reacted directly with cyanuric chloride under traditional aluminum chloride catalysis, the process suffers from poor regioselectivity, inevitably generating a complex mixture of isomers, tandem by-products, and unwanted tri-substituted impurities. These side reactions not only depress the yield of the desired 2,4-di(biphenyl-4-yl)-6-chloro-1,3,5-triazine intermediate but also create a purification nightmare that escalates production costs and compromises the optical clarity of the final UV absorber.
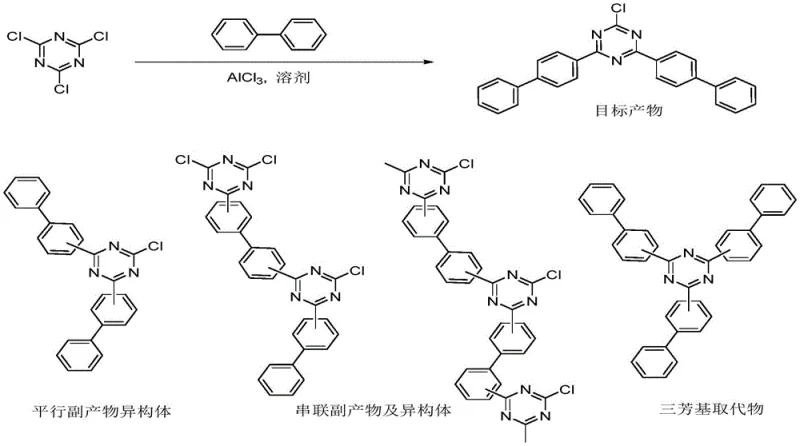
The Novel Approach
To circumvent these intrinsic chemical limitations, the inventors propose a strategic reversal of the synthetic sequence. Instead of attempting to install the difficult biphenyl group first, the new method prioritizes the attachment of the resorcinol moiety to the triazine ring. By utilizing cyanuric chloride and resorcinol as the initial feedstocks, the process leverages a highly selective perfluorosulfonate catalyst to achieve mono-substitution with exceptional precision. This first step generates 2,4-dichloro-6-(2,4-dihydroxyphenyl)-1,3,5-triazine, a stable intermediate that sets the stage for the subsequent introduction of the biphenyl units. Only after this scaffold is established is the biphenyl introduced in a second Friedel-Crafts reaction using standard aluminum trichloride. This "reverse addition" logic effectively decouples the selectivity issues from the solubility constraints, allowing for the use of standard organic solvents like dichlorobenzene in the second step without sacrificing yield or purity.
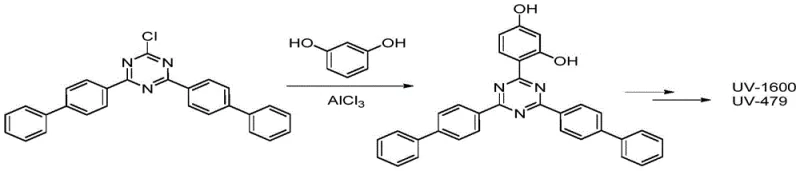
Mechanistic Insights into Perfluorosulfonate-Catalyzed Selective Substitution
The cornerstone of this technological advancement lies in the deployment of specialized perfluorosulfonate metal salts, such as aluminum trifluoromethanesulfonate, during the initial reaction phase. Unlike traditional Lewis acids which may promote uncontrolled poly-substitution or degradation, these superacidic catalysts exhibit a unique affinity for activating the triazine ring towards nucleophilic attack by resorcinol while maintaining strict control over the degree of substitution. The electron-withdrawing nature of the perfluoroalkyl groups enhances the Lewis acidity of the metal center, facilitating the reaction at moderate temperatures ranging from 100°C to 140°C. This controlled activation ensures that the reaction stops predominantly at the mono-substituted stage, preventing the formation of di- or tri-resorcinol adducts which would be detrimental to the final molecular architecture. The mechanism likely involves the formation of a transient coordination complex between the catalyst and the nitrogen atoms of the triazine ring, increasing the electrophilicity of the C-Cl bonds specifically at the 6-position due to steric and electronic factors inherent to the stepwise substitution of cyanuric chloride.
Furthermore, the choice of catalyst directly impacts the impurity profile and downstream processing efficiency. The patent data indicates that these perfluorosulfonate catalysts are not consumed in the reaction but can be recovered from the aqueous filtrate after the first step. By simply washing, drying, and reusing the catalyst, the process minimizes the generation of heavy metal waste and reduces the load on wastewater treatment facilities. This recyclability is a significant mechanistic advantage over stoichiometric reagents or precious metal catalysts that are lost during workup. The high selectivity achieved in this first step simplifies the crystallization of the intermediate, as fewer isomeric impurities are present to interfere with lattice formation. This purity carries through to the second step, where the pre-functionalized intermediate reacts with biphenyl, ensuring that the final UV absorber precursor meets the stringent optical requirements necessary for high-end polymer applications.
How to Synthesize 2,4-Bis(biphenyl-4-yl)-6-(2,4-dihydroxyphenyl)-1,3,5-triazine Efficiently
The synthesis protocol outlined in the patent provides a robust framework for producing this high-value intermediate with consistent quality. The process begins with the careful selection of the perfluorosulfonate catalyst, with aluminum trifluoromethanesulfonate identified as a particularly effective candidate for balancing activity and cost. Operators should maintain the reaction temperature within the specified window of 100°C to 140°C to optimize the trade-off between reaction rate and selectivity. Following the isolation of the mono-substituted intermediate, the second stage requires rigorous control of the aluminum trichloride dosage and reaction time to ensure complete conversion of the remaining chloro groups without inducing thermal degradation. For a comprehensive understanding of the specific operational parameters, safety precautions, and workup procedures, please refer to the detailed standardized synthesis guide below.
- Perform the first Friedel-Crafts reaction between cyanuric chloride and resorcinol using a highly selective perfluorosulfonate catalyst (e.g., aluminum trifluoromethanesulfonate) in an organic solvent like chlorobenzene at 100-140°C to obtain the mono-substituted intermediate.
- Recover and recycle the perfluorosulfonate catalyst from the filtrate via water washing and drying for subsequent batches, significantly reducing material costs.
- Conduct the second Friedel-Crafts reaction by reacting the isolated intermediate with biphenyl in dichlorobenzene using aluminum trichloride as the catalyst at 120-130°C to yield the final target triazine derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this novel catalytic route offers substantial strategic benefits beyond mere technical feasibility. The most immediate impact is seen in the raw material cost structure. By replacing expensive organometallic reagents—such as biphenyl Grignard reagents or boronic acids required in Suzuki couplings—with commodity chemicals like cyanuric chloride, resorcinol, and solid biphenyl, the bill of materials is significantly optimized. These feedstocks are produced on a massive global scale, ensuring price stability and availability even during market fluctuations. Furthermore, the elimination of precious metal catalysts like palladium removes a major cost driver and eliminates the need for complex and costly metal scavenging steps to meet regulatory limits on residual metals in final products.
- Cost Reduction in Manufacturing: The economic viability of this process is bolstered by the recyclability of the perfluorosulfonate catalyst. In traditional homogeneous catalysis, the catalyst is often quenched and discarded, representing a sunk cost and a waste disposal liability. In this new method, the catalyst is recovered from the aqueous phase with high efficiency and reused in subsequent batches. This circular approach drastically reduces the consumption of specialty chemicals per kilogram of product. Additionally, the high selectivity of the reaction minimizes the formation of by-products, which translates to higher overall yields and reduced solvent usage for purification. The simplified downstream processing, characterized by straightforward filtration and recrystallization rather than complex chromatography or distillation, further lowers utility and labor costs associated with manufacturing.
- Enhanced Supply Chain Reliability: Supply chain resilience is critically dependent on the robustness of the manufacturing process. Previous methods relying on Grignard reagents necessitated strictly anhydrous and oxygen-free environments, requiring specialized equipment and posing significant safety risks that could lead to unplanned downtime. The new Friedel-Crafts based approach operates under much more forgiving conditions, utilizing standard glass-lined or stainless steel reactors common in fine chemical plants. The use of stable, non-pyrophoric reagents reduces the risk of batch failures due to moisture ingress or handling errors. This operational stability ensures consistent lead times and allows suppliers to commit to larger volume contracts with greater confidence, securing the supply of critical UV absorber intermediates for long-term projects in the solar and construction sectors.
- Scalability and Environmental Compliance: As environmental regulations tighten globally, the ability to scale production while minimizing waste is a key competitive differentiator. This process inherently generates less hazardous waste by avoiding the use of strong bases like sodium hydride or sodium amide, which produce hydrogen gas and require careful quenching. The aqueous workup allows for the separation of the catalyst and inorganic salts in a controlled manner, facilitating easier treatment of effluent streams. The high atom economy of the Friedel-Crafts reactions, combined with the solvent recovery systems implied by the use of high-boiling solvents like dichlorobenzene, aligns well with green chemistry principles. This makes the technology attractive for expansion into regions with strict environmental compliance standards, ensuring long-term operational continuity without the threat of regulatory shutdowns.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on performance metrics and operational requirements.
Q: Why is the traditional Friedel-Crafts method unsuitable for synthesizing biphenyl-substituted triazines?
A: Traditional methods rely on benzene or xylene acting as both solvent and reactant. Since biphenyl is a solid with a high melting point, it cannot serve as a solvent. Furthermore, its lower reactivity and weak localization effects in direct reactions with cyanuric chloride lead to significant formation of isomers, tandem by-products, and tri-substituted impurities, complicating purification.
Q: What represents the primary cost advantage of this new catalytic route?
A: The process utilizes commodity raw materials such as cyanuric chloride, resorcinol, and biphenyl, avoiding expensive organometallic reagents like Grignard reagents or palladium catalysts. Additionally, the perfluorosulfonate catalyst used in the first step is highly recoverable and reusable, drastically lowering the cost of goods sold (COGS).
Q: How does this method improve supply chain reliability for UV absorber production?
A: By eliminating the need for harsh anhydrous and oxygen-free conditions required by Grignard or Suzuki coupling routes, the process becomes significantly safer and easier to operate on a large scale. The robust nature of the catalyst and the use of standard solvents facilitate consistent batch-to-batch quality and reliable delivery schedules.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,4-Bis(biphenyl-4-yl)-6-(2,4-dihydroxyphenyl)-1,3,5-triazine Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of advanced polymer additives hinges on the reliability and purity of the underlying intermediates. Our technical team has thoroughly analyzed the catalytic mechanisms described in CN116199638A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of 2,4-bis(biphenyl-4-yl)-6-(2,4-dihydroxyphenyl)-1,3,5-triazine meets the exacting standards required for high-performance UV absorbers like UV-1600 and UV-479. Our commitment to quality assurance guarantees that your downstream polymer formulations will perform consistently in the field.
We invite you to collaborate with us to leverage this innovative synthesis route for your supply chain. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate how our optimized manufacturing capabilities can drive value and security for your polymer additive sourcing strategy.
