Scalable Synthesis of 1-R-2,3-dihydro-1H-quinoline-4-ketone via Efficient Redox Strategy
The pharmaceutical and fine chemical industries continuously seek robust synthetic pathways for complex heterocyclic scaffolds, particularly quinoline derivatives which serve as critical building blocks for numerous bioactive molecules. Patent CN101429159B introduces a transformative synthesis method for 1-R-2,3-dihydro-1H-quinoline-4-ketone, addressing long-standing challenges associated with traditional manufacturing protocols. This innovation shifts the paradigm from harsh cyclization conditions to a streamlined three-step sequence involving N-alkylation, selective reduction, and controlled oxidation. By leveraging 4-hydroxyquinoline as a readily available starting material, the process circumvents the need for difficult-to-handle reagents and complex purification steps that have historically plagued the production of these valuable intermediates. For R&D directors and procurement specialists alike, this methodology represents a significant leap forward in process chemistry, offering a pathway that balances high purity requirements with operational simplicity and economic efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1-R-2,3-dihydro-1H-quinoline-4-ketone derivatives has relied heavily on intramolecular Friedel-Crafts acylation strategies. As illustrated in the conventional pathway, this typically involves the reaction of mono-substituted anilines with acrylic esters or nitriles, followed by a cyclization step mediated by polyphosphoric acid (PPA). This traditional approach presents severe logistical and technical hurdles for modern manufacturing environments. The reliance on PPA necessitates high reaction temperatures and generates a highly viscous reaction mixture that is notoriously difficult to stir and handle on a large scale. Furthermore, the post-reaction workup is cumbersome, often requiring extensive aqueous processing to remove the acidic medium, which leads to significant product loss due to the water solubility of certain ketone intermediates. These factors collectively result in low overall yields, inconsistent batch quality, and substantial environmental burdens due to waste generation, making the conventional route economically unviable for high-volume commercial production.
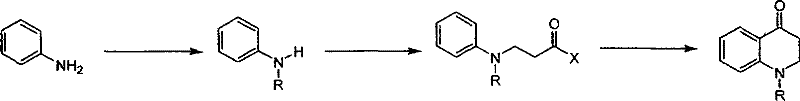
The Novel Approach
In stark contrast to the legacy methods, the novel approach detailed in the patent utilizes a divergent strategy starting from 4-hydroxyquinoline. This route bypasses the problematic cyclization step entirely by constructing the core skeleton first and then modifying the nitrogen substituent and saturation levels through a logical redox sequence. The process begins with the N-alkylation of 4-hydroxyquinoline using alkyl halides or sulfonates to form a stable ketone intermediate. This is followed by a selective reduction step to generate the corresponding tetrahydro-alcohol, and finally, a mild oxidation restores the ketone functionality while maintaining the desired dihydro state of the carbocyclic ring. This methodology operates under much milder conditions, utilizing common organic solvents such as methanol, ethanol, and dichloromethane, which facilitates easy filtration and extraction during workup. The elimination of polyphosphoric acid not only simplifies the equipment requirements but also drastically improves the safety profile and environmental footprint of the synthesis, making it an ideal candidate for reliable pharmaceutical intermediate supplier operations.

Mechanistic Insights into Selective Reduction-Oxidation Sequence
The core chemical ingenuity of this patent lies in the precise control of the oxidation state of the quinoline ring system. The initial N-alkylation step proceeds via a nucleophilic substitution mechanism where the nitrogen atom of the 4-hydroxyquinoline attacks the electrophilic carbon of the alkyl halide, facilitated by a base such as potassium carbonate. This forms a quaternary salt or N-alkylated species that tautomerizes to the stable 1-R-quinolin-4(1H)-one structure. The subsequent reduction step is critical; utilizing hydride sources like sodium borohydride or lithium aluminium hydride allows for the 1,4-reduction of the conjugated enone system within the heterocyclic ring. This selectively saturates the C2-C3 double bond and reduces the C4 ketone to an alcohol, yielding the 1-R-4-hydroxy-1,2,3,4-tetrahydroquinoline intermediate without affecting the aromatic benzene ring fused to the system. This chemoselectivity is paramount for ensuring the structural integrity of the final product.
Following the reduction, the final oxidation step employs mild oxidizing agents such as Dess-Martin periodinane or activated dimethyl sulfoxide (Swern oxidation conditions). The mechanistic goal here is to re-oxidize the secondary alcohol at the C4 position back to a ketone without causing dehydrogenation of the C2-C3 bond, which would revert the molecule to the fully aromatic quinolinone. By carefully controlling the reaction temperature, often keeping it low during the activation of the oxidant, the process ensures that only the alcohol functionality is targeted. This precise control minimizes the formation of over-oxidized byproducts or re-aromatized impurities, resulting in a high-purity crude product that requires minimal downstream purification. For R&D teams, understanding this mechanism highlights the robustness of the route against common side reactions that typically plague quinoline chemistry.
How to Synthesize 1-R-2,3-dihydro-1H-quinoline-4-ketone Efficiently
The synthesis protocol outlined in the patent provides a clear, reproducible framework for generating high-quality 1-R-2,3-dihydro-1H-quinoline-4-ketone derivatives. The procedure is designed to be operationally simple, avoiding the need for specialized high-pressure equipment or cryogenic conditions beyond standard laboratory capabilities. The key to success lies in the stoichiometric control of the alkylating agent and the careful selection of the reducing agent to match the specific R-group being installed. Whether synthesizing the methyl, ethyl, or benzyl variants, the general workflow remains consistent, allowing for a platform approach to manufacturing multiple analogues. Detailed standardized synthesis steps see the guide below.
- React 4-hydroxyquinoline with alkyl halides or sulfonates in solvents like acetonitrile or THF to form the N-alkylated quinolinone intermediate.
- Dissolve the intermediate ketone in alcohol solvents and treat with reducing agents such as sodium borohydride to obtain the corresponding tetrahydro-alcohol.
- Oxidize the alcohol intermediate using Dess-Martin periodinane or activated DMSO to yield the final 1-R-2,3-dihydro-1H-quinolin-4-one product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthesis route offers profound benefits for supply chain stability and cost management. The shift away from polyphosphoric acid eliminates a major bottleneck in production scheduling, as the lengthy and difficult workup associated with PPA is replaced by straightforward solvent evaporation and crystallization or extraction techniques. This transition significantly reduces the turnaround time between batches, allowing manufacturing facilities to increase throughput without expanding their physical footprint. Furthermore, the use of commodity chemicals like 4-hydroxyquinoline and common alkyl halides ensures that raw material sourcing is secure and less susceptible to market volatility compared to specialized cyclization precursors. This reliability is crucial for maintaining continuous supply lines for downstream API production.
- Cost Reduction in Manufacturing: The economic impact of this process is driven primarily by the simplification of the unit operations. By removing the requirement for corrosion-resistant reactors needed for hot polyphosphoric acid, capital expenditure for equipment can be optimized. Additionally, the improved yield profile across the three steps means that less raw material is wasted per kilogram of final product, directly lowering the cost of goods sold. The avoidance of complex chromatographic purifications, relying instead on standard recrystallization or column chromatography on silica, further reduces operational expenses related to consumables and labor.
- Enhanced Supply Chain Reliability: The robustness of the chemical transformations ensures consistent batch-to-batch quality, which is a critical metric for qualifying suppliers in the pharmaceutical industry. Since the reaction conditions are mild and the reagents are stable, the risk of batch failure due to thermal runaways or reagent degradation is minimized. This predictability allows supply chain managers to forecast delivery timelines with greater accuracy, reducing the need for excessive safety stock and enabling a more lean inventory model. The scalability of the process from gram to kilogram scales has been demonstrated, providing confidence for long-term procurement contracts.
- Scalability and Environmental Compliance: Environmental regulations are increasingly stringent regarding the disposal of acidic waste streams. This new method generates significantly less hazardous waste compared to the PPA route, simplifying effluent treatment and reducing disposal costs. The solvents used, such as ethyl acetate and alcohols, are easier to recover and recycle, contributing to a greener manufacturing profile. This alignment with green chemistry principles not only mitigates regulatory risk but also enhances the corporate sustainability credentials of the manufacturing partner, a factor that is becoming increasingly important in vendor selection processes for multinational corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for potential partners. Understanding these details is essential for evaluating the feasibility of integrating this intermediate into your specific drug development pipeline.
Q: What are the primary advantages of this synthesis route over traditional Friedel-Crafts methods?
A: This novel route eliminates the need for polyphosphoric acid (PPA), which is corrosive, difficult to handle, and generates significant waste. The new method uses mild liquid-phase reactions with standard workups, significantly improving scalability and environmental compliance.
Q: What starting materials are required for this process?
A: The process utilizes industrially available 4-hydroxyquinoline as the core scaffold, reacting it with common alkylating agents like methyl iodide, ethyl iodide, or benzyl bromide, followed by standard reducing and oxidizing agents.
Q: Is this method suitable for large-scale commercial production?
A: Yes, the patent explicitly addresses the difficulty of amplification in older methods. By avoiding high-viscosity reagents like PPA and utilizing standard solvent systems (MeOH, EtOH, DCM), the process is highly amenable to kilogram and ton-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-R-2,3-dihydro-1H-quinoline-4-ketone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality heterocyclic intermediates play in the development of next-generation therapeutics. Our technical team has thoroughly analyzed the synthesis method described in CN101429159B and validated its potential for industrial application. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to pilot plant is seamless. Our facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 1-R-2,3-dihydro-1H-quinoline-4-ketone delivered meets the exacting standards required for pharmaceutical grade materials.
We invite you to collaborate with us to leverage this advanced synthetic route for your projects. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our manufacturing capabilities can optimize your supply chain and accelerate your time to market.
