Revolutionizing Sulfur-Containing Dihydroisoxazole Production: A Green, Scalable Commercial Strategy
The pharmaceutical and agrochemical industries are constantly seeking more efficient, sustainable, and cost-effective pathways to construct complex heterocyclic scaffolds. A recent breakthrough detailed in patent CN113929637A introduces a novel synthetic strategy for sulfur-containing dihydroisoxazole compounds, a critical structural motif found in numerous bioactive molecules. This technology represents a significant departure from conventional organometallic approaches, leveraging a transition-metal-free protocol that utilizes inexpensive inorganic bases and water as the primary solvent. For R&D directors and procurement managers alike, this innovation promises to streamline the supply chain for high-purity pharmaceutical intermediates while simultaneously addressing the growing demand for green chemistry compliance. By replacing hazardous reagents with benign alternatives, this method not only enhances operational safety but also opens new avenues for the rapid construction of C-S bonds under mild conditions, ensuring robust impurity profiles essential for regulatory approval.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of sulfur-containing dihydroisoxazoles has relied heavily on sophisticated and often hazardous organometallic chemistry. As illustrated in prior art such as WO2010116121A, the construction of the critical C-S bond typically involved the use of Grignard reagents, specifically reacting benzylmagnesium bromide with 3-thiocyano-5,5-dimethyl-4,5-dihydroisoxazole. 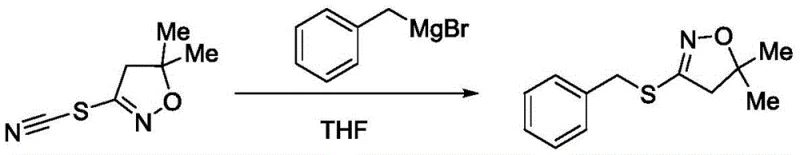 This traditional approach, depicted in Scheme 1, necessitates strictly anhydrous conditions and the handling of pyrophoric reagents, which poses significant safety risks and logistical challenges during commercial scale-up. Furthermore, alternative methods reported by Chantal Belie et al. utilized trimethylsilyl-containing substrates to achieve similar transformations, as shown in Scheme 2.
This traditional approach, depicted in Scheme 1, necessitates strictly anhydrous conditions and the handling of pyrophoric reagents, which poses significant safety risks and logistical challenges during commercial scale-up. Furthermore, alternative methods reported by Chantal Belie et al. utilized trimethylsilyl-containing substrates to achieve similar transformations, as shown in Scheme 2. 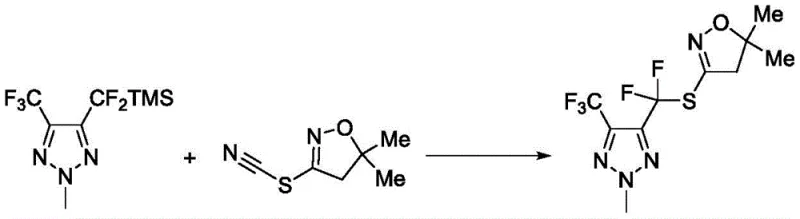 These legacy processes are burdened by high synthesis costs due to the requirement for specialized reagents and complex purification steps to remove metal residues. The reliance on transition metals or sensitive organometallic species often leads to complicated waste streams and increased environmental compliance costs, making these routes less attractive for modern, sustainability-focused manufacturing facilities.
These legacy processes are burdened by high synthesis costs due to the requirement for specialized reagents and complex purification steps to remove metal residues. The reliance on transition metals or sensitive organometallic species often leads to complicated waste streams and increased environmental compliance costs, making these routes less attractive for modern, sustainability-focused manufacturing facilities.
The Novel Approach
In stark contrast to the cumbersome legacy methods, the invention disclosed in CN113929637A offers a streamlined, green alternative that fundamentally changes the reaction mechanism. The new strategy employs a nucleophilic substitution reaction between a thiocyano dihydroisoxazole compound (III) and a halogenated hydrocarbon (IV or V) in the presence of a cheap inorganic base. 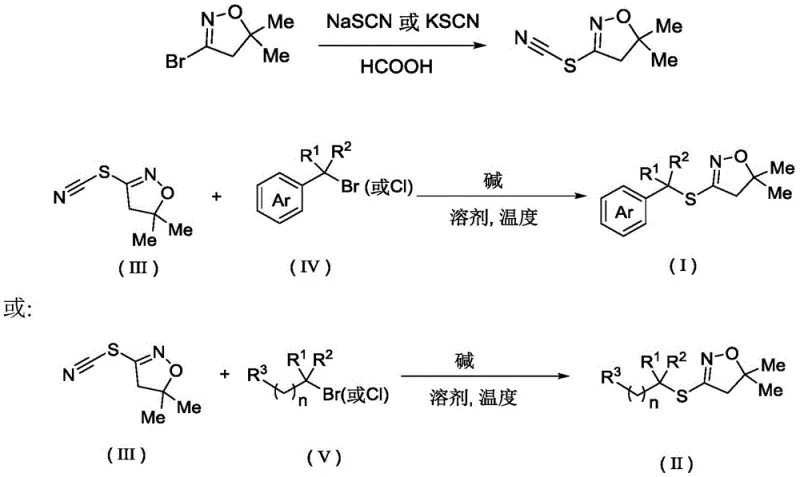 This approach eliminates the need for transition metal catalysts and special reagents entirely, operating effectively under mild conditions with water serving as the preferred solvent. The reaction proceeds efficiently at temperatures ranging from 50°C to 120°C, typically completing within just 1 to 3 hours. This shift from electrophilic organometallic reagents to electrophilic halides reacting with a thiocyanate nucleophile simplifies the process flow dramatically. The result is a robust synthesis capable of tolerating a wide array of functional groups, including halogens, cyano groups, and bulky alkyl chains, thereby providing a versatile platform for generating diverse libraries of sulfur-containing heterocycles without the baggage of heavy metal contamination.
This approach eliminates the need for transition metal catalysts and special reagents entirely, operating effectively under mild conditions with water serving as the preferred solvent. The reaction proceeds efficiently at temperatures ranging from 50°C to 120°C, typically completing within just 1 to 3 hours. This shift from electrophilic organometallic reagents to electrophilic halides reacting with a thiocyanate nucleophile simplifies the process flow dramatically. The result is a robust synthesis capable of tolerating a wide array of functional groups, including halogens, cyano groups, and bulky alkyl chains, thereby providing a versatile platform for generating diverse libraries of sulfur-containing heterocycles without the baggage of heavy metal contamination.
Mechanistic Insights into Base-Promoted Nucleophilic Substitution
The core of this technological advancement lies in the mechanistic inversion of the C-S bond formation process. In the conventional Grignard route, the alpha-carbon of the reagent acts as a carbanion (nucleophile), attacking the sulfur center. However, in this novel invention, the electronic roles are reversed: the alpha-carbon of the halogenated hydrocarbon (Compound IV or V) exhibits carbon positivity, acting as the electrophilic site. The sulfur atom in the thiocyano group of Compound (III) serves as the nucleophile, facilitated by the deprotonation or activation provided by the inorganic base such as sodium hydroxide or potassium carbonate. 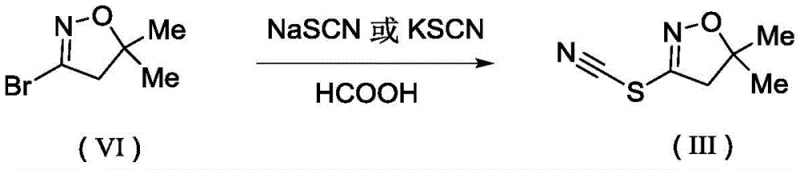 The preparation of the key intermediate (III) itself is optimized, reacting a bromo-dihydroisoxazole with sodium thiocyanate in formic acid at mild temperatures (0°C to 50°C). This two-step sequence ensures high purity of the starting material before the final coupling. The use of water as a solvent is particularly ingenious; it not only dissolves the inorganic base effectively but also helps to stabilize the transition state through hydrogen bonding, accelerating the reaction rate without the need for expensive phase transfer catalysts. This mechanistic clarity allows chemists to predict reactivity patterns accurately, ensuring that electron-withdrawing or donating groups on the aromatic ring of the halide do not inhibit the reaction, thus guaranteeing consistent yields across different substrates.
The preparation of the key intermediate (III) itself is optimized, reacting a bromo-dihydroisoxazole with sodium thiocyanate in formic acid at mild temperatures (0°C to 50°C). This two-step sequence ensures high purity of the starting material before the final coupling. The use of water as a solvent is particularly ingenious; it not only dissolves the inorganic base effectively but also helps to stabilize the transition state through hydrogen bonding, accelerating the reaction rate without the need for expensive phase transfer catalysts. This mechanistic clarity allows chemists to predict reactivity patterns accurately, ensuring that electron-withdrawing or donating groups on the aromatic ring of the halide do not inhibit the reaction, thus guaranteeing consistent yields across different substrates.
From an impurity control perspective, this metal-free pathway offers distinct advantages for pharmaceutical applications. Traditional transition-metal catalyzed couplings often leave behind trace amounts of palladium, copper, or nickel, which are strictly regulated in final drug substances due to toxicity concerns. Removing these metals requires additional purification steps, such as scavenging resins or recrystallization, which inevitably reduce overall yield and increase processing time. By completely avoiding transition metals, this new method inherently produces a cleaner crude product profile. The primary byproducts are simple inorganic salts (e.g., sodium bromide), which are easily removed during the aqueous workup and extraction phases described in the patent examples. This intrinsic cleanliness reduces the burden on QC labs and minimizes the risk of batch failures due to heavy metal specifications, directly contributing to a more reliable supply of high-purity pharmaceutical intermediates.
How to Synthesize 3-Benzylthio-5,5-dimethyl-4,5-dihydroisoxazole Efficiently
The practical implementation of this synthesis is straightforward and amenable to standard laboratory and plant equipment. The process begins by charging a reactor with the thiocyano precursor (Compound III), an appropriate inorganic base like sodium hydroxide, and water. To this mixture, the electrophile, such as benzyl bromide, is added. The reaction is then heated to reflux (approximately 100°C) and monitored via TLC until completion, typically within a few hours. Upon cooling, the product is extracted into an organic solvent like ethyl acetate, washed, and purified via column chromatography if necessary, although the high selectivity often allows for simpler crystallization methods in larger scales.
- Prepare the reaction mixture by combining 3-thiocyanato-4,5-dihydroisoxazole (Compound III), an inorganic base (e.g., NaOH), and a suitable solvent (preferably water) in a reactor.
- Add the electrophilic partner, such as a benzyl halide (Compound IV) or alkyl halide (Compound V), to the reactor under stirring.
- Heat the mixture to 50°C - 120°C (optimally 100°C) for 1-3 hours, monitor via TLC, and purify the crude product via extraction and column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route translates into tangible strategic benefits beyond mere chemical elegance. The elimination of expensive organometallic reagents and transition metal catalysts results in a drastic simplification of the raw material portfolio. Sourcing common inorganic bases and simple alkyl halides is far more stable and cost-predictable than managing the supply of sensitive Grignard reagents, which require special storage and handling protocols. This shift significantly reduces the total cost of ownership for the manufacturing process, as it removes the need for specialized containment systems and expensive waste disposal services associated with hazardous organometallic byproducts.
- Cost Reduction in Manufacturing: The economic impact of switching to this aqueous, metal-free protocol is profound. By removing the dependency on precious metal catalysts and anhydrous solvents, the direct material costs are substantially lowered. Furthermore, the simplified workup procedure—often requiring only extraction and evaporation rather than complex chromatographic purification or metal scavenging—reduces labor hours and solvent consumption. This efficiency gain allows for a more competitive pricing structure for the final sulfur-containing dihydroisoxazole intermediates, enabling downstream partners to optimize their own COGS (Cost of Goods Sold) in API manufacturing.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness of the reaction conditions. The tolerance for water and the use of stable inorganic bases mean that the process is less susceptible to fluctuations in raw material quality or environmental humidity compared to moisture-sensitive organometallic routes. This reliability ensures consistent batch-to-batch quality and reduces the risk of production delays caused by reagent degradation. Additionally, the short reaction time of 1-3 hours increases throughput capacity, allowing manufacturers to respond more agilely to market demand spikes without requiring significant capital investment in new reactor volume.
- Scalability and Environmental Compliance: From an EHS (Environment, Health, and Safety) perspective, this method is superior. The absence of pyrophoric reagents and the use of water as a solvent drastically lower the fire and explosion hazards associated with large-scale production. This safety profile simplifies the permitting process for new manufacturing lines and reduces insurance premiums. Moreover, the generation of benign inorganic salt waste aligns perfectly with modern green chemistry initiatives and stringent environmental regulations, facilitating smoother regulatory approvals and enhancing the corporate sustainability profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
To further clarify the technical and commercial implications of this patent, we have compiled answers to common inquiries regarding its implementation. These insights are derived directly from the experimental data and technical disclosures within CN113929637A, ensuring accuracy and relevance for industry professionals evaluating this technology for their pipelines.
Q: What are the primary advantages of this new synthesis method over traditional Grignard routes?
A: Unlike traditional methods requiring expensive Grignard reagents and strict anhydrous conditions, this patent utilizes cheap inorganic bases and water as a solvent. This eliminates the need for transition metal catalysts and special reagents, drastically simplifying the workflow and reducing raw material costs while maintaining high yields (up to 83%).
Q: Is this process scalable for industrial manufacturing of pharmaceutical intermediates?
A: Yes, the process is highly scalable. The use of water as a solvent and simple inorganic bases like sodium hydroxide removes the safety hazards associated with large-scale organometallic reactions. The mild reaction temperatures (50°C - 120°C) and short reaction times (1-3 hours) facilitate efficient commercial scale-up.
Q: What is the substrate scope for this C-S bond construction strategy?
A: The strategy demonstrates excellent universality. It tolerates various functional groups including halogens (Cl, F), cyano groups, and bulky substituents like tert-butyl. It works effectively with substituted phenyl, naphthalene, and heterocyclic halides, making it versatile for diverse drug molecule synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sulfur-Containing Dihydroisoxazole Supplier
The technological potential of sulfur-containing dihydroisoxazoles in drug discovery is immense, yet realizing this potential requires a manufacturing partner with deep technical expertise and scalable infrastructure. NINGBO INNO PHARMCHEM stands at the forefront of this field, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our commitment to quality is unwavering, supported by stringent purity specifications and rigorous QC labs that ensure every batch meets the exacting standards required for pharmaceutical intermediates. We understand that consistency is key, and our advanced process control systems guarantee that the benefits of this green synthesis method are fully realized in every kilogram we deliver.
We invite you to explore how this innovative synthesis route can optimize your supply chain and reduce costs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific project needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us collaborate to bring your next-generation therapeutics to market faster, safer, and more economically.
