Revolutionizing N-benzylideneaniline Production: A Safe, High-Yield Catalytic Transfer Hydrogenation Strategy for Global Supply Chains
The global demand for high-purity N-benzylideneaniline derivatives continues to surge, driven by their critical role as versatile intermediates in the pharmaceutical and agrochemical sectors. These compounds serve as pivotal precursors for synthesizing anti-anxiety medications, sedative-hypnotics, penicillin-class antibiotics, and various organophosphorus insecticides. However, traditional manufacturing routes have long been plagued by safety hazards, high raw material costs, and environmental concerns. A groundbreaking technological advancement detailed in patent CN103214392A offers a transformative solution to these industry-wide challenges. This patent discloses a novel, one-pot synthetic method that utilizes nitrobenzene compounds and benzyl alcohol under the catalytic action of supported noble metals. By shifting away from hazardous hydrogen gas and unstable aniline feedstocks, this innovation establishes a new benchmark for safety and efficiency in fine chemical manufacturing. For R&D directors and procurement leaders seeking a reliable pharmaceutical intermediate supplier, understanding the mechanistic depth and commercial viability of this catalytic transfer hydrogenation process is essential for securing a competitive edge in the supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of N-benzylideneaniline compounds has relied heavily on the direct condensation of aniline derivatives with benzaldehyde. While conceptually simple, this legacy approach suffers from severe economic and operational drawbacks that hinder large-scale optimization. Firstly, the raw materials—anilines and benzaldehydes—are significantly more expensive than their nitrobenzene and benzyl alcohol counterparts, directly inflating the cost of goods sold (COGS). Furthermore, aniline compounds are notoriously unstable and possess high toxicity profiles, necessitating rigorous containment protocols and increasing operational overhead. Alternative methods attempting to utilize nitrobenzenes often require external hydrogen gas sources for catalytic hydrogenation with benzaldehyde. This introduces substantial safety risks associated with high-pressure hydrogen handling and storage, alongside the complexity of managing explosive atmospheres within the plant. Additionally, previous attempts using gold catalysts with alcohols often resulted in over-reduction, yielding N-benzylaniline instead of the desired imine, thereby compromising yield and purity.
The Novel Approach
The methodology outlined in patent CN103214392A fundamentally reengineers the synthesis pathway by employing a catalytic transfer hydrogenation strategy. Instead of relying on external hydrogen or expensive aldehydes, this process utilizes benzyl alcohol as a dual-function reagent: it acts as the carbon skeleton source and simultaneously serves as the hydrogen donor. Under the influence of a supported noble metal catalyst—such as palladium or ruthenium dispersed on porous carbon or silica—the nitro group of the starting material is selectively reduced while the alcohol is oxidized in situ. This elegant cascade reaction occurs under mild nitrogen atmospheres at temperatures ranging from 50 to 240°C, with optimal performance observed between 160 and 180°C. The result is a highly selective formation of the target imine bond with minimal over-reduction. This approach not only drastically simplifies the reaction setup by eliminating high-pressure hydrogenation equipment but also leverages cheaper, more stable feedstocks, representing a paradigm shift in cost reduction in pharmaceutical intermediate manufacturing.
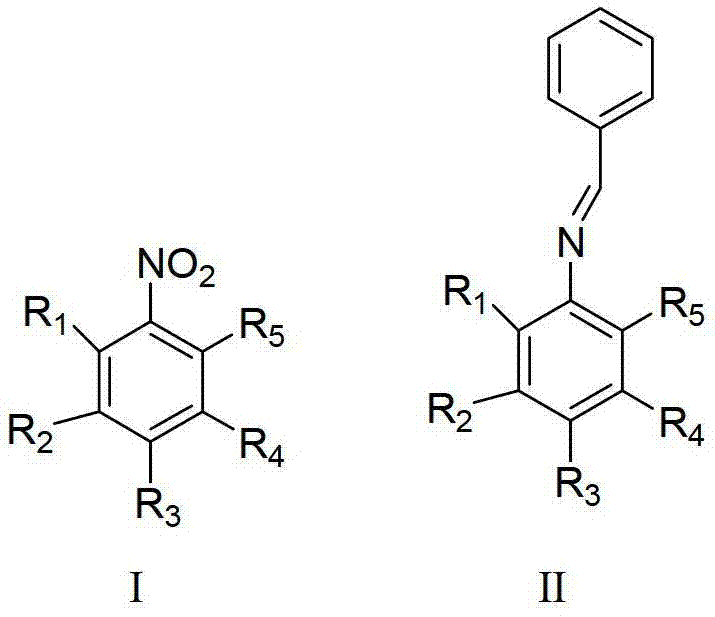
Mechanistic Insights into Supported Noble Metal-Catalyzed Transfer Hydrogenation
The core of this technological breakthrough lies in the sophisticated design of the heterogeneous catalyst system. The patent specifies the use of supported noble metals, including palladium, ruthenium, gold, silver, iridium, platinum, or rhodium, with palladium and ruthenium being particularly preferred for their balance of activity and cost. These active metal components are dispersed on high-surface-area carriers such as SiO2, Al2O3, activated carbon, or porous carbon materials. The mechanism involves the dehydrogenation of benzyl alcohol on the metal surface to generate benzaldehyde and active hydrogen species transiently. These active species immediately reduce the nitro group of the nitrobenzene derivative to a hydroxylamine or amine intermediate, which then condenses with the in situ generated benzaldehyde to form the Schiff base (imine). The presence of an inorganic base, preferably potassium hydroxide, is critical; it facilitates the dehydrogenation step of the alcohol and promotes the condensation equilibrium towards the imine product. This synergistic interaction between the metal active sites, the support structure, and the basic promoter ensures that the reaction proceeds with exceptional chemoselectivity.
Controlling impurity profiles is a paramount concern for R&D directors, particularly the prevention of over-reduction to N-benzylaniline, a common side reaction in reductive aminations. The patented catalyst system achieves remarkable selectivity, often exceeding 95% and reaching up to 98% in optimized examples (e.g., Example 4). This high selectivity is attributed to the specific electronic and geometric properties of the supported metal nanoparticles, which favor the desorption of the imine product before it can undergo further hydrogenation. Furthermore, the use of porous carbon supports, as demonstrated in Example 1, provides a unique microenvironment that may enhance mass transfer and stabilize the intermediate species. The robustness of the catalyst is further evidenced by its ability to maintain high conversion rates (100% for nitrobenzene) across various substrates, including substituted nitrobenzenes like 3,5-dichloronitrobenzene and o-nitrotoluene. This mechanistic precision ensures that the final high-purity N-benzylideneaniline products meet stringent quality specifications required for downstream drug synthesis without requiring complex purification steps.
How to Synthesize N-benzylideneaniline Efficiently
Implementing this advanced synthesis route requires precise adherence to the optimized reaction parameters defined in the patent to ensure maximum yield and safety. The process is designed to be scalable, moving seamlessly from laboratory verification to commercial production. The general procedure involves charging a reactor with the nitrobenzene derivative, a molar excess of benzyl alcohol (typically a ratio of 1:5 to 1:30), and the supported noble metal catalyst. An inorganic base is added to initiate the catalytic cycle, and the system is purged with nitrogen to maintain an inert atmosphere. The mixture is then heated to the optimal temperature window of 160-180°C and stirred for a duration of 16 to 26 hours. Upon completion, the reaction mixture is filtered to recover the solid catalyst for potential recycling, and the filtrate is concentrated. The crude product is subsequently recrystallized using an ethanol-water solution to afford the pure N-benzylideneaniline derivative. For a detailed, step-by-step breakdown of the standard operating procedures and specific catalyst preparation methods, please refer to the technical guide below.
- Prepare a supported noble metal catalyst (e.g., Pd/C or Ru/C) with a metal loading of 0.5-10.0% via impregnation and reduction.
- Mix nitrobenzene compound, benzyl alcohol (molar ratio 1: 5 to 1:30), inorganic base (e.g., KOH), and the catalyst in a reactor under nitrogen.
- Heat the mixture to 160-180°C for 16-26 hours, then filter and recrystallize the product from ethanol-water solution.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this catalytic transfer hydrogenation technology offers profound strategic benefits beyond mere technical superiority. The most immediate impact is seen in raw material sourcing and cost structure. By substituting expensive and volatile anilines and benzaldehydes with stable, commodity-grade nitrobenzenes and benzyl alcohols, manufacturers can achieve substantial cost savings. The elimination of external hydrogen gas removes the need for specialized high-pressure infrastructure and the associated safety compliance costs, further driving down capital expenditure (CAPEX) and operational expenditure (OPEX). Moreover, the process operates under atmospheric pressure (nitrogen blanket), significantly reducing the risk profile of the manufacturing site and ensuring business continuity by mitigating safety-related shutdowns. The high conversion rates and selectivity mean less waste generation and lower downstream purification costs, aligning perfectly with modern green chemistry mandates and environmental regulations.
- Cost Reduction in Manufacturing: The economic argument for this process is compelling due to the fundamental shift in feedstock economics. Nitrobenzene derivatives are generally more abundant and less costly than their corresponding anilines, which often require energy-intensive reduction steps to produce. Additionally, benzyl alcohol is a stable, easily handled liquid compared to benzaldehyde, which is prone to oxidation and requires careful storage. By combining the reduction and condensation steps into a single pot, the process eliminates unit operations, reduces solvent consumption, and minimizes labor hours. The ability to recycle the supported noble metal catalyst adds another layer of value, as the precious metal content can be recovered or the catalyst reused, effectively amortizing the catalyst cost over multiple batches and significantly lowering the variable cost per kilogram of the final API intermediate.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available, bulk chemical feedstocks. Unlike specialized reagents that may suffer from supply bottlenecks, nitrobenzenes and benzyl alcohols are produced on a massive global scale, ensuring consistent availability and price stability. The simplified process flow, which does not rely on external hydrogen supply chains or cryogenic storage, reduces dependency on third-party utility providers. This autonomy allows for more flexible production scheduling and faster response times to market demand fluctuations. Furthermore, the robustness of the catalyst system means that production runs are less susceptible to batch failures caused by catalyst deactivation or sensitivity to moisture/oxygen, guaranteeing a steady flow of commercial scale-up of complex pharmaceutical intermediates to downstream customers.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this technology is inherently superior. The absence of high-pressure hydrogen eliminates the risk of catastrophic explosions, a critical factor for insurance and regulatory compliance. The reaction generates water as the primary byproduct of the hydrogen transfer, minimizing the load on wastewater treatment facilities compared to processes generating heavy metal salts or acidic waste. The high atom economy of the transfer hydrogenation ensures that the majority of the reactant mass ends up in the desired product, reducing the carbon footprint of the manufacturing process. This alignment with sustainability goals not only future-proofs the supply chain against tightening environmental regulations but also enhances the brand reputation of the end-product manufacturers who prioritize green sourcing in their vendor selection criteria.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of integrating this synthesis route into their existing portfolios, we have compiled answers to common inquiries regarding the catalyst performance, substrate scope, and operational parameters. These insights are derived directly from the experimental data and claims within patent CN103214392A, providing a transparent view of the technology's capabilities. Understanding these nuances is vital for making informed decisions about process adoption and supplier qualification.
Q: What is the primary advantage of using benzyl alcohol over benzaldehyde in this synthesis?
A: Benzyl alcohol serves as both a reactant and a hydrogen donor, eliminating the need for hazardous external hydrogen gas and reducing raw material costs compared to unstable anilines and expensive benzaldehydes.
Q: Can this catalytic system prevent over-reduction to N-benzylaniline?
A: Yes, the optimized supported noble metal catalysts (such as Pd/C or Ru/C) exhibit high chemoselectivity, achieving over 95% selectivity for the imine product while minimizing further reduction to the amine.
Q: Is the catalyst reusable in this process?
A: The patent indicates that the prepared noble metal catalysts possess high stability and can be recycled, contributing to a more sustainable and cost-effective manufacturing process.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-benzylideneaniline Supplier
The technical potential of the catalytic transfer hydrogenation method described in CN103214392A represents a significant opportunity for optimizing the production of key pharmaceutical and agrochemical intermediates. At NINGBO INNO PHARMCHEM, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this innovative chemistry to life. Our state-of-the-art facilities are equipped to handle supported noble metal catalysis with the utmost precision, ensuring stringent purity specifications and rigorous QC labs verify every batch against global pharmacopeia standards. We understand that transitioning to a new synthetic route requires a partner who can navigate the complexities of process validation and regulatory filing with expertise and reliability.
We invite you to collaborate with us to leverage this advanced technology for your specific supply chain needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this greener, safer route can improve your margins. Please contact our technical procurement team today to request specific COA data for our N-benzylideneaniline portfolio and discuss route feasibility assessments for your custom projects. Let us help you secure a sustainable, cost-effective, and high-quality supply of critical intermediates for your global operations.
