Advanced One-Step Synthesis of Aryl Tin Compounds for Scalable Pharmaceutical Intermediate Production
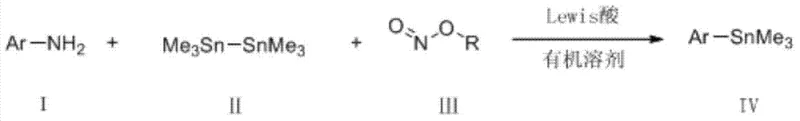
The chemical manufacturing landscape is constantly evolving, driven by the need for more efficient, cost-effective, and scalable synthetic routes for critical intermediates. A significant breakthrough in this domain is detailed in patent CN103665026A, which discloses a novel preparation method for aryl tin compounds. This technology represents a paradigm shift from traditional multi-step or harsh condition processes to a streamlined, one-step conversion of aromatic amines into valuable aryl tin species. By leveraging a Lewis acid-promoted system involving distannanes and alkyl nitrites, this method achieves high reaction rates under remarkably mild conditions. For R&D directors and process chemists, this offers a robust alternative for constructing complex molecular architectures, particularly for applications in pharmaceutical intermediates and fine chemical synthesis where purity and functional group tolerance are paramount.
The significance of this patent extends beyond mere academic interest; it addresses critical pain points in industrial organic synthesis. The ability to directly transform aromatic amines, which are often cheaper and more diverse than their halide counterparts, into organotin reagents opens new avenues for retrosynthetic analysis. This is particularly relevant for the production of high-purity pharmaceutical intermediates, where the presence of residual heavy metals from traditional catalysts can be a disqualifying factor. The method described in CN103665026A eliminates the need for transition metal catalysts like palladium, thereby simplifying downstream purification and ensuring a cleaner impurity profile for the final active pharmaceutical ingredients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of aryl tin compounds has relied heavily on two primary strategies, both of which present substantial logistical and economic challenges for large-scale manufacturing. The first conventional approach involves the generation of aryl Grignard or aryl lithium reagents from aromatic halides, followed by quenching with trialkyltin chlorides. This pathway is fraught with difficulties, including the requirement for strictly anhydrous and oxygen-free conditions to prevent the decomposition of highly reactive organometallic intermediates. Furthermore, the use of stoichiometric amounts of metal reagents generates significant quantities of salt waste, complicating environmental compliance and waste disposal protocols. The second common method utilizes palladium-catalyzed cross-coupling between aromatic halides and distannanes. While effective, this route necessitates the use of expensive noble metal catalysts, which not only increases raw material costs but also introduces the risk of toxic metal contamination in the final product, necessitating costly and time-consuming metal scavenging steps.
The Novel Approach
In stark contrast to these legacy methods, the technology outlined in CN103665026A introduces a direct, one-step conversion of aromatic amines to aryl tin compounds that circumvents the aforementioned limitations. By utilizing a Lewis acid to promote the reaction between an aromatic amine, a distannane, and an alkyl nitrite in an organic solvent, this method operates under mild temperatures ranging from 0°C to 30°C. Crucially, the reaction does not demand strict inert atmosphere conditions and proceeds smoothly in air, drastically reducing the complexity of reactor setup and operational overhead. This approach not only broadens the scope of accessible substrates to include a wide variety of functionalized aromatic amines but also significantly lowers the barrier to entry for producing these valuable building blocks, making it an ideal candidate for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Lewis Acid-Promoted Stannylation
The core innovation of this methodology lies in the synergistic interaction between the Lewis acid, the alkyl nitrite, and the distannane reagent. While the exact mechanistic pathway may involve complex radical or cationic intermediates, the overarching principle involves the in situ generation of a reactive diazonium-like species from the aromatic amine and alkyl nitrite, which is subsequently intercepted by the distannane. The Lewis acid plays a pivotal role in accelerating this transformation, likely by activating the nitrite ester or stabilizing the transition state, thereby allowing the reaction to proceed rapidly at near-ambient temperatures. This catalytic promotion is essential for achieving high yields across a diverse range of substrates, as evidenced by the patent data which shows successful conversion of electron-deficient and electron-rich systems alike. The absence of transition metals ensures that the reaction mechanism is distinct from standard Pd-catalyzed cycles, avoiding issues related to catalyst deactivation or ligand optimization.
From an impurity control perspective, this mechanism offers distinct advantages for the production of high-purity OLED material or pharmaceutical precursors. Since the reaction does not involve halide exchange or metal-halogen insertion, the formation of homocoupling byproducts (biaryls) typically seen in Grignard chemistry is minimized. Furthermore, the tolerance for sensitive functional groups such as esters, aldehydes, and ketones suggests that the reactive intermediates are sufficiently short-lived or selective to avoid attacking these electrophilic sites. This selectivity is critical for maintaining the structural integrity of complex molecules during the stannylation step, ensuring that the resulting aryl tin compounds are suitable for subsequent downstream transformations like Stille couplings without requiring extensive protective group strategies.
How to Synthesize Aryl Tin Compounds Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to reagent ratios and temperature control, although the overall procedure is notably forgiving compared to traditional organometallic methods. The standard protocol involves dissolving the aromatic amine substrate and a Lewis acid promoter, such as p-toluenesulfonic acid, in a suitable organic solvent like 1,2-dichloroethane. Subsequently, the alkyl nitrite and hexamethyldistannane are added to the mixture, and the reaction is allowed to stir for a period typically ranging from 1 to 4 hours. The detailed standardized synthesis steps see the guide below.
- Dissolve the aromatic amine substrate and a Lewis acid promoter (such as p-toluenesulfonic acid) in an organic solvent like 1,2-dichloroethane.
- Add alkyl nitrite (e.g., tert-butyl nitrite) and hexamethyldistannane to the reaction mixture under mild temperature conditions ranging from 0°C to 30°C.
- Stir the reaction for 1 to 4 hours in air, then concentrate and purify the resulting aryl tin compound via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this Lewis acid-promoted stannylation technology offers compelling economic and logistical benefits that extend far beyond the laboratory bench. The primary advantage stems from the utilization of aromatic amines as starting materials, which are frequently more abundant and less expensive than the corresponding aromatic iodides or bromides required for palladium-catalyzed methods. This shift in raw material sourcing can lead to substantial cost savings in the overall bill of materials, particularly for large-volume production runs. Additionally, the elimination of expensive palladium catalysts removes a significant variable cost component and mitigates the supply chain risks associated with the fluctuating prices of precious metals. The simplified operational requirements further contribute to efficiency gains, as the process does not necessitate specialized equipment for handling pyrophoric reagents or maintaining rigorous inert atmospheres.
- Cost Reduction in Manufacturing: The economic impact of this method is driven by the removal of high-cost inputs and the simplification of the process workflow. By avoiding the use of noble metal catalysts, manufacturers eliminate the need for expensive metal scavenging resins and the associated validation testing to ensure residual metal levels meet regulatory standards. Furthermore, the ability to run the reaction in air reduces the consumption of inert gases like nitrogen or argon and lowers the energy costs associated with maintaining strict glovebox or Schlenk line conditions. These factors combine to create a leaner manufacturing process with a significantly reduced cost of goods sold, enhancing the competitiveness of the final chemical products in the global market.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness of the reaction conditions and the availability of starting materials. Aromatic amines are commodity chemicals produced on a massive scale for the dye and polymer industries, ensuring a stable and diversified supply base that is less susceptible to shortages compared to specialized halide intermediates. The tolerance of the reaction to ambient conditions means that production schedules are less vulnerable to disruptions caused by equipment failure in inert gas systems or delays in obtaining ultra-dry solvents. This reliability allows for more predictable lead times and inventory planning, enabling companies to respond more agilely to market demands for critical pharmaceutical intermediates and fine chemicals.
- Scalability and Environmental Compliance: Scaling this process from gram to tonnage quantities is facilitated by the mild thermal profile and the absence of hazardous exotherms typical of Grignard reagent formation. The reaction operates safely between 0°C and 30°C, which can be easily managed with standard cooling systems in large reactors, removing the need for cryogenic infrastructure. From an environmental standpoint, the reduction in heavy metal waste and the avoidance of stoichiometric metal salts align with green chemistry principles, simplifying waste treatment and disposal. This compliance with stringent environmental regulations reduces the administrative burden and potential liabilities associated with hazardous waste management, making the process more sustainable and socially responsible for long-term commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this aryl tin synthesis technology. These answers are derived directly from the experimental data and specifications provided in the patent literature, ensuring accuracy and relevance for process development teams. Understanding these nuances is essential for evaluating the feasibility of integrating this method into existing production workflows and for optimizing reaction parameters for specific substrates.
Q: Does this stannylation method require strict anhydrous or oxygen-free conditions?
A: No, unlike traditional Grignard-based methods, this Lewis acid-promoted protocol proceeds smoothly in air without the need for strict anhydrous or oxygen-free environments, significantly simplifying operational requirements.
Q: What types of functional groups are tolerated during this aryl tin synthesis?
A: The method exhibits excellent functional group tolerance, successfully accommodating esters, cyano groups, aldehydes, ketones, nitro groups, halogens, and trifluoromethyl groups on the aromatic ring.
Q: Why is using aromatic amines advantageous over aromatic halides for this transformation?
A: Aromatic amines are often more cost-effective and readily available starting materials compared to aromatic halides, and this method avoids the use of expensive palladium catalysts typically required for halide stannylation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aryl Tin Compounds Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced synthetic methodologies like the Lewis acid-promoted stannylation described in CN103665026A. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory discoveries are seamlessly translated into robust industrial processes. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs, guaranteeing that every batch of aryl tin compounds meets the exacting standards required for pharmaceutical and electronic material applications. We leverage our deep technical expertise to optimize reaction conditions, maximize yields, and minimize impurities, delivering value that goes beyond simple manufacturing.
We invite you to collaborate with us to explore how this efficient synthesis route can enhance your supply chain and reduce your overall production costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific project needs. By partnering with us, you gain access to specific COA data and route feasibility assessments that empower you to make informed decisions about your chemical sourcing strategy. Contact us today to discuss your requirements and discover how NINGBO INNO PHARMCHEM can serve as your trusted partner in bringing high-quality aryl tin intermediates to market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
