Optimizing PDE IV Inhibitor Intermediate Production via Advanced Lithium-Mediated Hydrolysis
Introduction to Advanced PDE IV Inhibitor Intermediate Synthesis
The development of potent phosphodiesterase type IV (PDE IV) inhibitors represents a critical frontier in the treatment of bronchial asthma and other inflammatory respiratory conditions. Patent CN1192025C discloses a sophisticated methodology for the preparation of substituted 4-phenyl-4-cyanocyclohexanecarboxylic acids, which serve as pivotal intermediates in the manufacture of these therapeutic agents. The core innovation lies in a novel hydrolysis protocol that utilizes Group I(a) or II(a) metal halides within an aprotic dipolar amide solvent system. This approach addresses longstanding challenges in impurity management and process safety, specifically regarding the handling of cyanide byproducts generated during nitrile hydrolysis. By leveraging the differential solubility properties of lithium salts in mixed solvent systems, this technology enables the direct isolation of high-purity carboxylate intermediates. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediates supplier, understanding the mechanistic advantages of this route is essential for evaluating long-term supply chain stability and cost-efficiency in anti-asthma drug manufacturing.
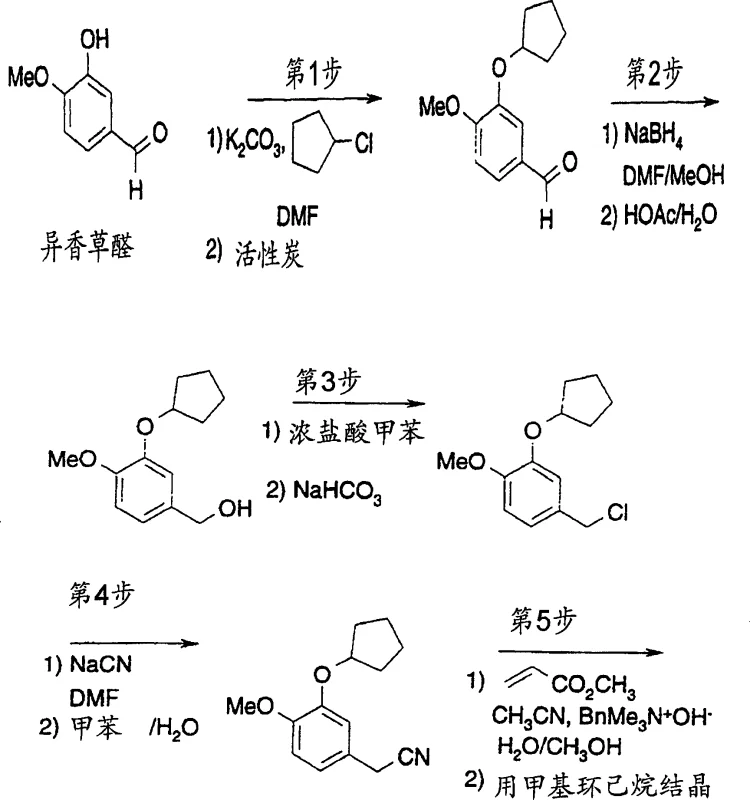
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for generating 4-substituted-4-(3,4-disubstituted phenyl)cyclohexanecarboxylic acids often rely on harsh acidic or basic hydrolysis conditions that can compromise the structural integrity of sensitive functional groups. Conventional methods frequently struggle with the efficient removal of hydrocyanic acid (HCN), a toxic byproduct formed when acylnitriles are hydrolyzed to carboxylic acids. In standard protocols, the separation of the desired acid from cyanide salts often necessitates complex work-up procedures, including multiple extraction steps or oxidative treatments to neutralize residual cyanide, which significantly increases waste generation and operational complexity. Furthermore, the use of non-selective hydrolysis conditions can lead to the formation of regio-isomeric impurities or degradation products that are difficult to remove, thereby impacting the overall purity profile required for pharmaceutical grade materials. These inefficiencies translate into higher production costs and extended lead times, creating bottlenecks for commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
The methodology described in CN1192025C introduces a paradigm shift by employing a metal halide-mediated ring-opening and hydrolysis sequence in a carefully controlled solvent environment. Instead of isolating the free acid directly, the process forms an insoluble carboxylate salt, specifically a lithium salt, which precipitates out of the reaction mixture. This precipitation event is the key differentiator; it allows for the physical separation of the product from soluble impurities, most notably the metal cyanide salts which remain dissolved in the mother liquor. The use of aprotic dipolar amide solvents like DMF, combined with controlled amounts of water, facilitates the hydrolysis while maintaining the solubility differential required for purification. This streamlined approach eliminates the need for extensive downstream purification steps, offering a more robust and scalable pathway for cost reduction in pharmaceutical intermediates manufacturing. The result is a process that not only enhances safety by sequestering cyanide in the waste stream but also improves the overall yield and purity of the final active intermediate.
Mechanistic Insights into Lithium Bromide-Mediated Hydrolysis
The chemical elegance of this process resides in the specific interaction between the metal halide catalyst, the solvent system, and the substrate. The reaction initiates with the ring-opening of a cyanoepoxide precursor using a Group I(a) metal halide, such as lithium bromide, in the presence of water. This step generates an acylnitrile intermediate which is subsequently hydrolyzed to the corresponding carboxylic acid functionality. Crucially, the reaction is conducted in a solvent system comprising DMF and water, where the solubility characteristics of lithium salts are exploited to drive purification. Upon the addition of a strong base, typically lithium hydroxide, the carboxylic acid is converted into its lithium carboxylate form. Due to the specific solvation properties in the DMF/water mixture, the lithium carboxylate exhibits low solubility and precipitates as a solid. Conversely, the lithium cyanide generated from the hydrolysis of the nitrile group remains highly soluble in this medium. This differential solubility is the mechanistic cornerstone that allows for the effective removal of cyanide contaminants without requiring additional chemical scavengers or oxidation steps.
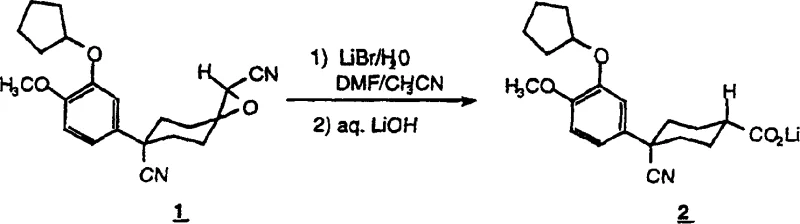
Furthermore, the choice of lithium as the cation is not arbitrary but is driven by thermodynamic and kinetic factors inherent to the solvent system. Lithium cyanide demonstrates significantly higher solubility in DMF compared to sodium or potassium cyanide, ensuring that the toxic byproduct stays in solution during the filtration of the product salt. This mechanistic feature provides a built-in safety valve for the process, minimizing the risk of HCN release during work-up. The reaction temperature, maintained above 60°C, ensures sufficient kinetic energy for the hydrolysis to proceed to completion while avoiding thermal degradation of the sensitive cyclohexane ring system. For technical teams evaluating process feasibility, this mechanism offers a clear advantage in terms of impurity control, as the crystallization of the lithium salt acts as a powerful purification step that inherently rejects soluble ionic impurities. The subsequent acidification of the isolated lithium salt to obtain the free acid is a straightforward operation that preserves the high purity achieved in the precipitation step.
How to Synthesize 4-cyano-4-(3-cyclopentyloxy-4-methoxyphenyl)cyclohexanecarboxylic Acid Efficiently
The practical execution of this synthesis requires precise control over solvent ratios and reagent addition sequences to maximize yield and purity. The process begins with the preparation of a reaction mixture containing the cyanoepoxide precursor, lithium bromide, and a solvent blend of DMF, acetonitrile, and water. Heating this mixture under inert atmosphere facilitates the initial ring-opening and hydrolysis transformations. Once the reaction is deemed complete via analytical monitoring, a stoichiometric excess of aqueous lithium hydroxide is introduced to induce the precipitation of the target lithium carboxylate. The detailed standardized synthesis steps for this procedure are outlined below.
- Mix Group I(a) or II(a) metal halide (e.g., LiBr) with an aprotic dipolar amide solvent (e.g., DMF), water, and the cyanoepoxide precursor.
- Heat the mixture to at least 60°C under inert atmosphere to effect ring-opening and hydrolysis to the acylnitrile and subsequently the acid.
- Add a strong base (e.g., LiOH) to precipitate the carboxylate salt while keeping cyanide salts in solution, then isolate and acidify.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented hydrolysis technology offers substantial benefits for supply chain resilience and operational expenditure. The primary advantage stems from the simplification of the purification workflow, which directly correlates to reduced processing time and lower utility consumption. By eliminating the need for complex cyanide destruction steps or multiple recrystallizations to meet purity specifications, manufacturers can achieve significant cost savings in pharmaceutical intermediates manufacturing. The robustness of the precipitation-based purification also means that the process is less sensitive to minor variations in raw material quality, enhancing the reliability of supply for downstream API production. This stability is crucial for maintaining continuous manufacturing operations and meeting strict delivery schedules for global pharmaceutical clients.
- Cost Reduction in Manufacturing: The elimination of expensive heavy metal catalysts and the reduction in solvent usage for purification steps contribute to a leaner cost structure. The ability to recover and recycle the DMF solvent further amplifies these economic benefits, as the mother liquor containing the soluble cyanide salts can be treated separately without contaminating the main product stream. This efficient use of resources ensures that the production of high-purity pharmaceutical intermediates remains economically viable even at large scales.
- Enhanced Supply Chain Reliability: The use of readily available commodity chemicals such as lithium bromide and lithium hydroxide mitigates the risk of supply disruptions associated with specialized reagents. The process operates under relatively mild conditions compared to high-pressure hydrogenation or cryogenic reactions, reducing the dependency on specialized equipment and lowering the barrier for contract manufacturing organizations to adopt the technology. This accessibility ensures a broader base of potential suppliers, strengthening the overall supply chain security.
- Scalability and Environmental Compliance: The inherent safety features of the process, particularly the containment of cyanide in the aqueous waste stream, simplify environmental compliance and waste treatment protocols. The solid-liquid separation allows for easier handling of intermediates on a multi-ton scale, facilitating seamless technology transfer from pilot plant to commercial production. This scalability is essential for meeting the growing demand for PDE IV inhibitors in the global respiratory therapy market.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis route. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for process evaluation.
Q: How does this method handle hazardous HCN byproducts?
A: The process utilizes a unique precipitation mechanism where the carboxylate salt precipitates out of the aqueous aprotic solvent system, while the generated metal cyanide salts remain highly soluble in the mother liquor, allowing for safe and efficient physical separation without additional oxidation steps.
Q: What are the preferred solvents for this hydrolysis reaction?
A: The patent specifies the use of proton-dipolar amide-based solvents such as dimethylformamide (DMF), dimethylacetamide, or N-methylpyrrolidone, often in combination with water-miscible co-solvents like acetonitrile to optimize solubility and reaction kinetics.
Q: Why is lithium bromide preferred over other metal halides?
A: Lithium bromide is particularly effective because the resulting lithium cyanide salt exhibits superior solubility in the reaction medium compared to sodium or potassium analogs, ensuring minimal contamination of the final precipitated carboxylate product.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-cyano-4-(3-cyclopentyloxy-4-methoxyphenyl)cyclohexanecarboxylic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of next-generation respiratory therapeutics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to market supply is seamless and efficient. We are committed to delivering stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 4-cyano-4-(3-cyclopentyloxy-4-methoxyphenyl)cyclohexanecarboxylic acid meets the exacting standards required for clinical and commercial applications. Our facility is equipped to handle the specific solvent systems and safety protocols necessary for this lithium-mediated chemistry, providing a secure and compliant manufacturing environment.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic advantages specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and innovation in your drug development programs.
