Advanced Synthesis of Phenyltetrazole Intermediates for Commercial Angiotensin II Antagonist Production
Advanced Synthesis of Phenyltetrazole Intermediates for Commercial Angiotensin II Antagonist Production
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for critical cardiovascular drug intermediates, particularly those serving the massive market of Angiotensin II receptor blockers (ARBs). Patent CN1832931A introduces a transformative methodology for the preparation of phenyltetrazole derivatives, specifically targeting the ortho-substituted intermediates required for drugs like Losartan and Valsartan. This innovation addresses the longstanding safety and efficiency bottlenecks associated with traditional lithiation chemistry by employing a direct ortho-metallation strategy using magnesium amide bases. The core breakthrough lies in the ability to generate highly reactive organometallic species under significantly milder and safer conditions compared to legacy organolithium protocols. By shifting the paradigm from cryogenic lithiation to reflux-based magnesiation, this technology offers a compelling value proposition for manufacturers aiming to optimize their supply chains for high-volume API production.
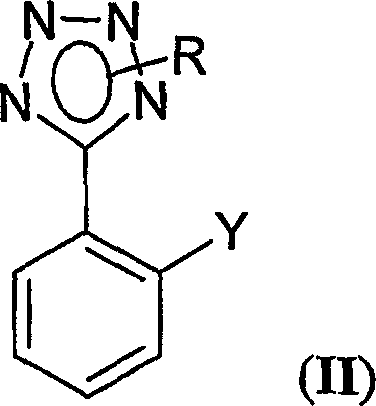
The structural versatility of the compounds generated via this process is evident in Formula (II), where the ortho-position can be functionalized with boron or zinc groups essential for downstream cross-coupling reactions. This specific intermediate architecture is the cornerstone for constructing the biphenyl-tetrazole motif found in nearly all major ARB medications. The patent details how varying the protecting group R and the substituent Y allows for precise tuning of the molecule's reactivity and solubility profiles. For R&D directors evaluating process feasibility, the ability to access these diverse substitution patterns without compromising yield is a critical advantage. Furthermore, the elimination of hazardous reagents simplifies the regulatory landscape for facility approvals, making this route particularly attractive for established production sites looking to modernize their capabilities without extensive infrastructure overhauls.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
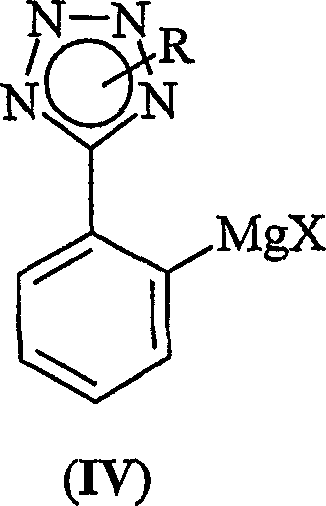
Historically, the synthesis of ortho-functionalized phenyltetrazoles has relied heavily on the use of organolithium reagents, which, while effective, present severe operational challenges on an industrial scale. The primary drawback is the extreme pyrophoric nature of alkyllithium compounds, necessitating specialized handling equipment, inert atmosphere gloveboxes, and rigorous safety protocols that drive up capital expenditure. Moreover, these reactions typically require cryogenic temperatures, often below minus 70 degrees Celsius, to control selectivity and prevent decomposition, leading to substantial energy consumption and increased processing time. An alternative approach involving Grignard reagents was attempted to mitigate these risks, but as illustrated by Formula (IV), the resulting magnesium species often exhibited insufficient reactivity for direct cross-coupling. This lack of reactivity forced manufacturers to introduce additional transmetallation steps, thereby elongating the synthetic sequence and increasing the potential for yield loss and impurity accumulation.
The Novel Approach
The methodology disclosed in CN1832931A circumvents these issues by utilizing magnesium amide bases, such as diisopropylaminomagnesium, to effect direct ortho-metallation of the tetrazole ring. This approach generates a highly reactive magnesium intermediate, designated as Formula (V), which possesses superior nucleophilicity compared to the sluggish Grignard species of the prior art. Because the reaction can be conducted at reflux temperatures in common ether solvents like tetrahydrofuran, the need for expensive cryogenic cooling is entirely eliminated. This thermal robustness not only enhances safety by removing the risk of thermal runaway associated with exothermic lithiation but also significantly accelerates the reaction kinetics. The resulting intermediate can then be seamlessly converted into the desired zinc or boron derivatives through simple transmetallation, streamlining the overall process flow and reducing the total number of unit operations required to reach the final API intermediate.
Mechanistic Insights into Magnesium Amide Mediated Ortho-Metallation
The mechanistic pathway of this novel synthesis relies on the unique basicity and coordination properties of the magnesium amide complex. Unlike simple Grignard reagents which exist in a Schlenk equilibrium, the magnesium amide base acts as a potent, non-nucleophilic base that selectively deprotonates the ortho-position of the phenyl ring adjacent to the tetrazole moiety. This deprotonation is facilitated by the coordination of the magnesium center to the nitrogen atoms of the tetrazole ring, directing the metalation to the adjacent carbon atom with high regioselectivity. The formation of the carbon-magnesium bond in Formula (V) is the rate-determining step, and the use of refluxing THF ensures that the activation energy barrier is overcome efficiently without degrading the sensitive tetrazole heterocycle. This controlled metalation prevents the formation of poly-metalated byproducts that are common in aggressive lithiation conditions, thereby ensuring a cleaner reaction profile from the outset.
Following the formation of the magnesiated species, the subsequent transmetallation step is crucial for installing the functional handle required for Suzuki or Negishi coupling. When reacting with zinc chloride, the magnesium species undergoes a rapid exchange to form the corresponding organozinc compound, which is stable and highly reactive towards palladium-catalyzed cross-coupling. Alternatively, reaction with borate esters yields the boronic acid or ester derivatives directly. The patent highlights that this transmetallation proceeds with high fidelity, preserving the integrity of the tetrazole protecting group. From an impurity control perspective, this two-step one-pot sequence minimizes the exposure of reactive intermediates to atmospheric moisture or oxygen, as the entire transformation can be managed within a closed reactor system. This containment strategy is vital for maintaining the stringent purity specifications required for pharmaceutical intermediates, effectively suppressing the formation of hydrolysis byproducts or homocoupling impurities.
How to Synthesize Phenyltetrazole Derivatives Efficiently
The practical execution of this synthesis involves charging a reactor with the protected phenyltetrazole precursor and a solution of the magnesium amide base in anhydrous tetrahydrofuran. The mixture is then heated to reflux, typically around 65 to 70 degrees Celsius, and maintained for several hours to ensure complete conversion to the magnesiated intermediate. Monitoring via HPLC or NMR confirms the disappearance of the starting material and the formation of the organometallic species. Once metalation is complete, the electrophile source, such as zinc chloride or trimethyl borate, is added to the reaction mixture, often at a slightly reduced temperature to manage any exotherm. The subsequent stirring period allows for the quantitative transfer of the organic group to the zinc or boron center. Finally, the reaction is quenched with a mild acid or water, and the product is isolated through standard workup procedures involving phase separation and crystallization.
- React the protected phenyltetrazole precursor with a magnesium amide base, such as diisopropylaminomagnesium, in an ether solvent like THF under reflux conditions to generate the ortho-magnesiated intermediate.
- Perform a transmetallation reaction by adding an electrophile source, such as zinc chloride or a borate ester, to the reaction mixture to convert the magnesium species into the desired zinc or boron derivative.
- Quench the reaction mixture with acid or water, separate the organic phase, and purify the resulting phenyltetrazole derivative through crystallization or concentration to obtain the high-purity intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this magnesium-mediated process translates into tangible strategic benefits regarding cost stability and operational reliability. The most significant advantage is the drastic reduction in safety-related overheads; by eliminating the need for organolithium reagents, facilities can avoid the high costs associated with specialized storage, transport, and disposal of pyrophoric materials. This shift lowers the barrier to entry for contract manufacturing organizations (CMOs) and allows for production in a wider range of facilities, thereby diversifying the supply base and reducing dependency on single-source vendors who possess niche cryogenic capabilities. Furthermore, the simplified process flow, which removes the need for intermediate isolation of unstable species, leads to shorter cycle times and higher throughput per reactor volume. These efficiencies collectively contribute to a more resilient supply chain capable of meeting fluctuating market demands for cardiovascular medications without the volatility associated with complex, hazard-prone chemistries.
- Cost Reduction in Manufacturing: The economic impact of replacing cryogenic lithiation with reflux-based magnesiation is profound, primarily driven by the elimination of energy-intensive cooling systems and the reduction in reactor occupancy time. Without the need to maintain temperatures below minus 70 degrees Celsius, utility costs for refrigeration are significantly curtailed, directly improving the gross margin of the manufacturing process. Additionally, the higher yields reported in the patent examples imply less raw material waste and lower solvent consumption per kilogram of product, further driving down the variable cost of goods sold. The use of commercially available and relatively inexpensive magnesium amide bases, as opposed to custom-synthesized organolithiums, also stabilizes the raw material cost structure against market fluctuations.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the logistical complexities of handling hazardous reagents, which are subject to strict transportation regulations and limited carrier availability. By transitioning to a chemistry that utilizes standard, non-hazardous reagents and solvents, the supply chain becomes inherently more robust and less prone to disruptions caused by regulatory compliance issues or shipping delays. The scalability of the process is another key factor; since the reaction does not rely on heat transfer limitations imposed by cryogenic jackets, scaling from pilot plant to commercial production is straightforward and predictable. This predictability allows supply chain planners to forecast lead times with greater accuracy, ensuring that downstream API synthesis schedules are met consistently.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process aligns well with modern green chemistry principles by reducing the generation of hazardous waste streams. The absence of lithium salts in the waste effluent simplifies wastewater treatment protocols and reduces the environmental footprint of the manufacturing site. Moreover, the improved atom economy and reduced solvent usage contribute to a lower overall E-factor, a key metric for sustainable chemical manufacturing. As regulatory bodies increasingly scrutinize the environmental impact of pharmaceutical production, adopting this cleaner technology positions manufacturers favorably for future audits and inspections, mitigating the risk of production halts due to non-compliance.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this phenyltetrazole synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer and for commercial teams negotiating supply agreements.
Q: Why is the magnesium amide method superior to organolithium reagents for tetrazole synthesis?
A: Organolithium reagents require extreme cryogenic conditions and pose significant safety risks due to pyrophoricity. The magnesium amide method described in CN1832931A operates at reflux temperatures in standard solvents, drastically reducing operational hazards and energy costs associated with cooling.
Q: What represents the key impurity challenge in conventional Grignard approaches?
A: Conventional Grignard reagents often form species with low reactivity (Formula IV) that necessitate additional transmetallation steps, increasing the risk of side reactions and impurity generation. The novel direct metallation approach minimizes these steps, leading to a cleaner impurity profile.
Q: Can this process be scaled for industrial production of Losartan intermediates?
A: Yes, the process utilizes common solvents like THF and toluene and avoids hazardous cryogenic operations, making it highly suitable for large-scale commercial manufacturing. The use of stable magnesium amide bases ensures consistent batch-to-batch reproducibility essential for GMP production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phenyltetrazole Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global supply of life-saving cardiovascular medications. Our technical team has extensively analyzed the process described in CN1832931A and possesses the expertise to implement this advanced magnesium-mediated route at commercial scale. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with unwavering consistency. Our state-of-the-art facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of phenyltetrazole derivative meets the exacting standards required for GMP API synthesis. We are committed to delivering not just a product, but a partnership built on technical excellence and supply chain security.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project requirements. By leveraging our process optimization capabilities, we can provide a Customized Cost-Saving Analysis that quantifies the potential economic benefits for your organization. We encourage you to request specific COA data and route feasibility assessments to validate the superiority of this method for your portfolio. Let us collaborate to secure a sustainable and cost-effective supply of these critical intermediates for the global market.
