Scalable Green Synthesis of 2,4-Diamino-5-Nitroso-6-Hydroxypyrimidine for Guanine Manufacturing
Scalable Green Synthesis of 2,4-Diamino-5-Nitroso-6-Hydroxypyrimidine for Guanine Manufacturing
The pharmaceutical industry is constantly seeking more sustainable and cost-effective pathways for producing critical antiviral intermediates, and patent CN108558776B presents a groundbreaking methodology for the preparation of 2,4-diamino-5-nitroso-6-hydroxypyrimidine. This specific intermediate serves as a pivotal precursor in the synthesis of guanine, which is essential for manufacturing lovir-class antiviral drugs such as acyclovir and ganciclovir, as well as folic acid derivatives. The core innovation lies in the strategic replacement of concentrated sulfuric acid with dilute formic acid during the nitrosation phase, a shift that fundamentally alters the waste profile and economic viability of the process. By integrating solvent recovery systems for methanol and enabling the mechanical application of nitrosation mother liquor, this technology addresses the chronic issues of high-salt wastewater discharge that have plagued conventional production methods. Furthermore, the process facilitates the recovery of sodium nitrate as a high-purity byproduct, transforming a potential waste liability into a revenue-generating asset for manufacturers. This comprehensive approach not only aligns with stringent environmental regulations but also offers a robust framework for scaling up production without compromising on yield or purity specifications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of nitrosopyrimidine intermediates has relied heavily on the use of excessive amounts of concentrated sulfuric acid and sodium nitrite to drive the nitrosation reaction to completion. This traditional approach generates vast quantities of high-salt and high-acid wastewater, creating a significant environmental burden and escalating disposal costs for manufacturing facilities. A critical flaw in these legacy processes is the inability to recycle the reaction mother liquor effectively, primarily because the accumulation of sodium sulfate and the dilution of sulfuric acid render the solution unusable for subsequent batches. Consequently, valuable reagents such as excess sodium nitrite and sulfuric acid are wasted in every cycle, leading to inflated raw material costs and inefficient resource utilization. Additionally, the corrosive nature of concentrated sulfuric acid necessitates specialized equipment and rigorous safety protocols, further increasing the capital expenditure required for plant maintenance and operation. The resulting waste streams often require complex neutralization and treatment procedures before discharge, adding another layer of operational complexity and expense to the overall manufacturing workflow.
The Novel Approach
In stark contrast to the wasteful conventional techniques, the novel methodology outlined in the patent introduces a streamlined process that utilizes dilute formic acid as the acidifying agent for the nitrosation reaction. This substitution allows for the effective recycling of the nitrosation mother liquor, as the formic acid system does not suffer from the same salt accumulation issues that plague sulfuric acid-based routes. The process is designed to recover anhydrous methanol and sodium nitrate with high efficiency, ensuring that solvents and reagents are continuously cycled back into the production line rather than being discarded. By employing dilute formic acid, the reaction conditions become more manageable, facilitating easier separation of the product and significantly reducing the volume of wastewater generated. This approach not only simplifies the downstream processing steps but also enhances the overall atom economy of the synthesis, making it a superior choice for modern, eco-conscious chemical manufacturing. The integration of these recovery loops creates a closed-loop system that minimizes fresh reagent input while maximizing output quality and consistency.
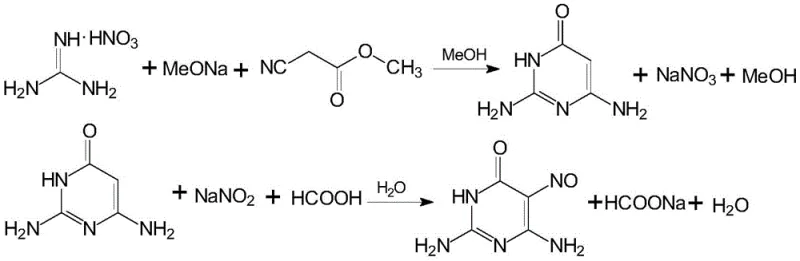
Mechanistic Insights into Formic Acid-Mediated Nitrosation and Cyclization
The mechanistic foundation of this improved synthesis begins with the cyclization of methyl cyanoacetate and a guanidine salt within a sodium methoxide methanol solution, a reaction that proceeds efficiently under reflux conditions to form the pyrimidine ring structure. Following cyclization, the addition of recovered anhydrous methanol induces the precipitation of sodium nitrate, a byproduct that can be filtered off with a purity exceeding 99.0%, effectively removing it from the reaction matrix before the nitrosation step. The subsequent nitrosation reaction is conducted in a dilute formic acid solution, where the pH is carefully controlled between 1 and 4 to optimize the generation of the active nitrosating species while minimizing side reactions. This specific pH range is crucial for maintaining the stability of the intermediate and ensuring high selectivity for the 5-nitroso position on the pyrimidine ring. The use of formic acid, a weaker acid compared to sulfuric acid, provides a milder reaction environment that reduces the risk of over-nitrosation or degradation of the sensitive amino groups present on the molecule. Furthermore, the presence of recycled nitrosation mother liquor in the reaction mixture helps to buffer the system and maintain consistent reaction kinetics across multiple batches.
A particularly ingenious aspect of this mechanism is the integration of waste streams from downstream guanine production back into this upstream intermediate synthesis. The ring-closure reaction for converting triaminopyrimidine to guanine typically generates large volumes of dilute formic acid filtrate, which is often too weak to be reused in the guanine step itself. However, this patent identifies that such dilute formic acid solutions, with concentrations ranging from 35% to 65%, are perfectly suited for the nitrosation of the diamino-hydroxypyrimidine intermediate. This cross-process recycling eliminates the need for fresh concentrated acid and solves the disposal problem for the guanine unit's waste stream. Additionally, the washing liquids from the filtration of the nitrosopyrimidine product are collected and concentrated via MVR (Mechanical Vapor Recompression) to recover sodium formate, which can then be reused in the guanine ring-closure step. This intricate web of material flows demonstrates a deep understanding of process chemistry, turning potential waste liabilities into valuable feedstocks and creating a highly integrated and efficient manufacturing ecosystem.
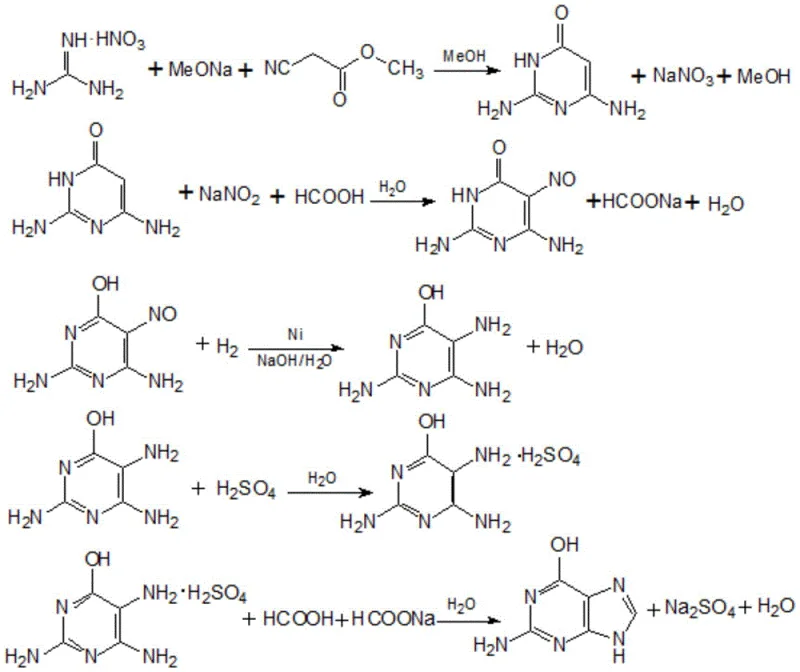
How to Synthesize 2,4-Diamino-5-Nitroso-6-Hydroxypyrimidine Efficiently
The execution of this synthesis requires precise control over reaction parameters and a disciplined approach to solvent management to fully realize the benefits of the patented process. Operators must ensure that the cyclization reaction reaches completion before proceeding to the crystallization of sodium nitrate, as residual starting materials can interfere with the purity of the final nitrosated product. The nitrosation step demands careful monitoring of temperature, ideally maintained between 55°C and 65°C, and pH levels to prevent the formation of unwanted byproducts. Detailed standardized operating procedures are essential for managing the complex recycling loops of methanol, formic acid, and sodium formate to ensure batch-to-batch consistency. For a comprehensive guide on the specific operational parameters and step-by-step instructions, please refer to the technical protocol below.
- Perform cyclization of methyl cyanoacetate and guanidine salt in sodium methoxide methanol solution under reflux.
- Dilute with recovered anhydrous methanol to precipitate and filter high-purity sodium nitrate byproduct.
- Conduct nitrosation in dilute formic acid solution using recycled mother liquor, followed by crystallization and drying.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this technology translates into tangible improvements in cost structure and supply reliability that go beyond simple yield metrics. The ability to recover and sell high-purity sodium nitrate as a byproduct creates a new revenue stream that directly offsets the cost of raw materials, effectively lowering the net production cost of the intermediate. Moreover, the elimination of high-salt wastewater treatment reduces the operational expenditure associated with environmental compliance and waste disposal, which are often hidden but significant costs in fine chemical manufacturing. The process's reliance on recycled solvents and reagents diminishes the volatility associated with fluctuating market prices for fresh chemicals, providing a more stable and predictable cost base for long-term planning. This stability is crucial for maintaining competitive pricing in the global market for antiviral intermediates, where margin pressure is often intense.
- Cost Reduction in Manufacturing: The substitution of concentrated sulfuric acid with recovered dilute formic acid eliminates the need for purchasing large quantities of fresh strong acid, leading to substantial savings in reagent costs. Additionally, the recovery of sodium nitrate with chemical-grade purity allows manufacturers to sell this byproduct, turning a waste disposal cost into a profit center that improves the overall gross margin of the production line. The recycling of methanol and the reuse of nitrosation mother liquor further reduce the consumption of fresh solvents, compounding the cost savings over time. By minimizing the generation of hazardous waste, the facility also avoids the high fees associated with third-party waste treatment services, resulting in a leaner and more economically efficient operation.
- Enhanced Supply Chain Reliability: The closed-loop nature of this process reduces dependency on external suppliers for certain reagents, as a significant portion of the acid and solvent requirements are met through internal recycling. This self-sufficiency mitigates the risk of supply disruptions caused by logistics issues or market shortages, ensuring a more continuous and reliable production schedule. The simplified waste profile means that production is less likely to be halted by environmental regulatory inspections or waste tank capacity limits, which are common bottlenecks in traditional chemical plants. Consequently, manufacturers can offer more reliable lead times to their customers, strengthening business relationships and enhancing their reputation as a dependable partner in the pharmaceutical supply chain.
- Scalability and Environmental Compliance: The process is inherently designed for scale, utilizing standard unit operations like distillation, filtration, and crystallization that are easily adaptable from pilot to commercial scale without complex engineering changes. The significant reduction in wastewater volume and toxicity simplifies the permitting process for new facilities and ensures ongoing compliance with increasingly strict environmental regulations globally. The use of MVR technology for concentrating sodium formate demonstrates a commitment to energy efficiency, further aligning the process with modern sustainability goals. This environmental friendliness not only future-proofs the manufacturing asset but also appeals to downstream pharmaceutical clients who are increasingly auditing their suppliers for green chemistry credentials.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this advanced synthesis route. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation to ensure accuracy and relevance for industry professionals. Understanding these details is crucial for evaluating the feasibility of adopting this technology in existing or new manufacturing setups. We encourage stakeholders to review these points carefully to appreciate the full scope of the innovation.
Q: How does this process improve upon traditional sulfuric acid methods?
A: Traditional methods generate high-salt, high-acid wastewater that is difficult to treat. This novel approach uses dilute formic acid, allowing for the recovery of sodium nitrate as a saleable byproduct and enabling the recycling of nitrosation mother liquor, significantly reducing environmental burden.
Q: What is the source of the dilute formic acid used in the reaction?
A: The process utilizes dilute formic acid recovered from the subsequent guanine ring-closure reaction. Instead of discarding low-concentration formic acid filtrates, they are repurposed for the nitrosation step, creating a closed-loop system that minimizes reagent consumption.
Q: Is the sodium nitrate byproduct suitable for commercial sale?
A: Yes, the optimized crystallization conditions using recovered anhydrous methanol yield sodium nitrate with purity levels meeting chemical grade standards (≥99.0%), allowing it to be sold directly rather than treated as waste.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,4-Diamino-5-Nitroso-6-Hydroxypyrimidine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and sustainable intermediate synthesis in the broader context of antiviral drug manufacturing. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and robust. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs that employ advanced analytical techniques to verify every batch. Our infrastructure is designed to handle complex recycling loops and green chemistry protocols, allowing us to maximize the economic and environmental benefits of technologies like the one described in CN108558776B.
We invite you to engage with our technical procurement team to discuss how we can tailor our production capabilities to meet your specific volume and quality requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized processes can reduce your total cost of ownership. We are ready to provide specific COA data and route feasibility assessments to support your R&D and supply chain planning efforts. Partner with us to secure a reliable, high-quality supply of this essential intermediate for your guanine and antiviral drug production needs.
