Advanced Catalytic Oxidation for Scalable Production of Key Agrochemical Intermediates
The pharmaceutical and agrochemical industries are constantly seeking robust synthetic pathways that balance high purity with operational safety, a challenge vividly addressed in patent CN1186310C. This pivotal intellectual property discloses a highly efficient process for preparing 3,3-dimethyl-2-formylcyclopropanecarboxylic ester, a critical building block in the synthesis of potent pyrethroid insecticides. The innovation lies in replacing hazardous traditional oxidants with a benign hydrogen peroxide system mediated by transition metal catalysts such as tungsten or molybdenum oxides. For R&D directors and process chemists, this represents a significant leap forward in green chemistry application, offering a route that minimizes toxic waste streams while maintaining rigorous control over the impurity profile. By leveraging this technology, manufacturers can achieve a reliable agrochemical intermediate supplier status, ensuring that the supply chain remains uninterrupted by regulatory crackdowns on hazardous reagents. The versatility of the method allows for the use of various ester groups (R), ranging from simple methyl and ethyl groups to complex aralkyl substituents, providing immense flexibility for downstream derivatization.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of formyl-functionalized cyclopropanes relied heavily on ozonolysis or oxidation with periodic acid, both of which present formidable challenges for commercial scale-up. Ozonolysis, while effective chemically, requires specialized infrastructure to generate and handle ozone, a gas that is not only highly toxic but also poses a significant explosion risk if not managed with extreme precision. Furthermore, the quenching of ozonides often generates unstable intermediates that can lead to runaway reactions if thermal control is lost. Alternatively, methods utilizing periodic acid suffer from severe economic drawbacks due to the high cost of the reagent and the generation of stoichiometric amounts of iodine-containing waste, which complicates environmental compliance and wastewater treatment. These legacy processes create bottlenecks in cost reduction in agrochemical intermediates manufacturing, forcing producers to absorb high operational expenditures that erode profit margins. Consequently, there has been a persistent industry demand for a safer, more economical alternative that does not compromise on yield or product quality.
The Novel Approach
The methodology described in the patent introduces a transformative catalytic cycle that utilizes hydrogen peroxide as the terminal oxidant, activated by a suite of tungsten, molybdenum, or rhenium-based catalysts. This approach fundamentally alters the reaction landscape by operating under mild conditions, typically between 0°C and 80°C, which drastically reduces energy consumption compared to high-thermal processes. The reaction proceeds by selectively cleaving the isobutenyl side chain of the chrysanthemic acid ester precursor to generate the desired aldehyde functionality without disturbing the sensitive cyclopropane ring. As illustrated below, the transformation converts the starting material directly into the target aldehyde with high atom economy.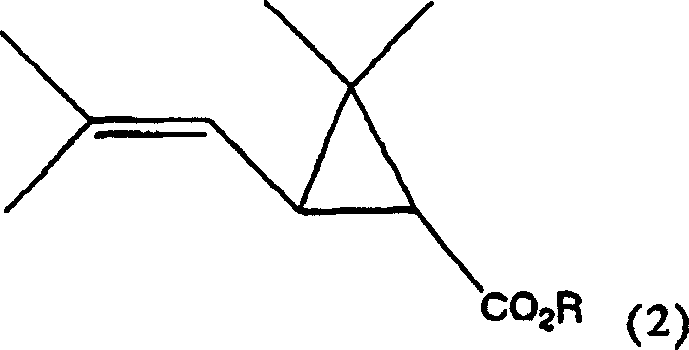
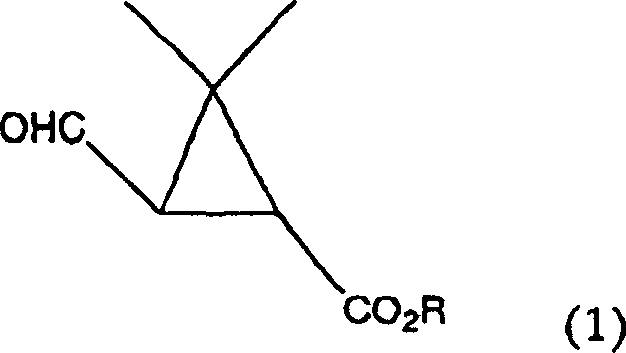 This visual representation underscores the structural fidelity maintained during the oxidation, where the gem-dimethyl cyclopropane core remains intact. The use of commercially available hydrogen peroxide solutions further simplifies logistics, eliminating the need for on-site generation of dangerous gases and allowing for the commercial scale-up of complex agrochemical intermediates with greater confidence and safety.
This visual representation underscores the structural fidelity maintained during the oxidation, where the gem-dimethyl cyclopropane core remains intact. The use of commercially available hydrogen peroxide solutions further simplifies logistics, eliminating the need for on-site generation of dangerous gases and allowing for the commercial scale-up of complex agrochemical intermediates with greater confidence and safety.
Mechanistic Insights into Tungsten-Catalyzed Oxidative Cleavage
The core of this technological advancement lies in the activation of hydrogen peroxide by the transition metal catalyst to form a reactive peroxo-species capable of attacking the electron-rich double bond of the isobutenyl group. In the presence of tungstic acid or tungstates, the peroxide coordinates to the metal center, increasing its electrophilicity and facilitating the formation of an epoxide or glycol intermediate on the side chain. Subsequent rearrangement and cleavage steps, likely driven by the acidic nature of the reaction medium or added mineral acids, lead to the scission of the carbon-carbon double bond and the formation of the formyl group. This mechanism is highly selective, avoiding over-oxidation to the carboxylic acid, which is a common side reaction in less controlled oxidative environments. The ability to tune the catalyst loading, typically ranging from 0.001 to 0.95 moles per mole of substrate, allows process chemists to optimize the reaction kinetics for specific batch sizes. Furthermore, the inclusion of phase transfer catalysts like quaternary ammonium salts can enhance the interaction between the aqueous peroxide and the organic substrate, ensuring homogeneous reaction progress even in biphasic systems.
Impurity control is another critical aspect where this mechanistic understanding provides a competitive edge. Traditional methods often struggle with the formation of ring-opened byproducts or over-oxidized acids due to the harsh conditions required. In contrast, the tungsten-catalyzed system operates with sufficient gentleness to preserve the strained cyclopropane ring, which is susceptible to acid-catalyzed ring opening under vigorous conditions. The patent data indicates that the stereochemistry of the starting material—whether cis, trans, (+), or (-)—is preserved in the final product, meaning that optical purity is not compromised during the transformation. This is vital for producing high-purity agrochemical intermediates where biological activity is often stereodependent. By carefully managing the water content through the use of dehydrating agents like anhydrous magnesium sulfate or azeotropic distillation, the reaction equilibrium can be shifted towards the desired aldehyde, minimizing the formation of hydrate impurities. This level of control ensures that the final API intermediate meets the stringent purity specifications required by global regulatory bodies.
How to Synthesize 3,3-Dimethyl-2-Formylcyclopropanecarboxylate Efficiently
Implementing this synthesis route requires a systematic approach to reagent preparation and reaction monitoring to ensure optimal yields and safety. The process begins with the activation of the catalyst, often involving the pre-mixing of the tungsten source with hydrogen peroxide to generate the active peroxo-complex before the introduction of the organic substrate. Solvent selection plays a pivotal role, with tert-butanol and other water-miscible or immiscible organic solvents serving as effective media to dissolve both the oxidant and the lipophilic chrysanthemic ester. Detailed standardized synthesis steps are provided in the guide below to assist technical teams in replicating these results.
- Prepare the catalytic system by mixing a tungsten-based catalyst (such as tungstic acid or sodium tungstate) with hydrogen peroxide in a suitable solvent like tert-butanol.
- Introduce the chrysanthemic acid ester substrate (Formula 2) into the reaction mixture under controlled temperature conditions, typically between 0°C and 80°C.
- Maintain stirring for the required duration (e.g., 2 to 24 hours) to ensure complete conversion, followed by workup involving extraction and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this catalytic oxidation process translates into tangible strategic benefits that extend beyond simple chemical yield. The shift away from ozone and periodic acid removes significant capital and operational barriers, allowing for more flexible manufacturing scheduling and reduced dependency on specialized hazardous material vendors. By utilizing hydrogen peroxide, a commodity chemical with a stable and robust global supply chain, manufacturers can mitigate the risks associated with supply disruptions of exotic reagents. This stability is crucial for maintaining continuous production lines and meeting the just-in-time delivery expectations of downstream agrochemical formulators. Furthermore, the simplified waste profile reduces the burden on environmental health and safety departments, leading to lower disposal costs and a smaller environmental footprint.
- Cost Reduction in Manufacturing: The elimination of expensive stoichiometric oxidants like periodic acid results in substantial cost savings on raw material procurement, as hydrogen peroxide is significantly cheaper on a molar basis. Additionally, the removal of the need for ozone generation equipment reduces capital expenditure (CAPEX) and maintenance costs associated with corrosion-resistant reactors and gas handling systems. The catalytic nature of the tungsten species means that small amounts of metal can drive the conversion of large quantities of substrate, further driving down the cost per kilogram of the final product. These factors combined create a much more favorable economic model for the production of high-volume agrochemical intermediates.
- Enhanced Supply Chain Reliability: Sourcing hydrogen peroxide and tungsten salts is far less complex than securing supplies of ozone or high-purity periodic acid, which may be subject to stricter transport regulations. The use of common solvents like tert-butanol or ethyl acetate ensures that the entire bill of materials consists of readily available industrial chemicals. This accessibility reduces lead time for high-purity agrochemical intermediates, enabling faster response to market demand fluctuations. Suppliers can maintain higher inventory levels of raw materials without the safety concerns associated with storing explosive gases, thereby enhancing overall supply chain resilience against external shocks.
- Scalability and Environmental Compliance: The reaction conditions described, operating at moderate temperatures and atmospheric pressure, are inherently easier to scale from pilot plant to multi-ton production than high-pressure ozonolysis. The aqueous nature of the oxidant and the potential for catalyst recycling or easy separation simplify the downstream processing and wastewater treatment protocols. This alignment with green chemistry principles facilitates smoother regulatory approvals and reduces the risk of production shutdowns due to environmental non-compliance. The process demonstrates excellent scalability, making it suitable for both niche custom synthesis and large-scale commodity production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this tungsten-catalyzed oxidation technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this route into their existing manufacturing portfolios.
Q: Why is the tungsten-catalyzed method preferred over ozonolysis for this intermediate?
A: The tungsten-catalyzed method eliminates the severe safety hazards associated with handling toxic and explosive ozone gas, while also avoiding the high costs of periodic acid reagents used in older methods.
Q: Does this oxidative process affect the stereochemistry of the cyclopropane ring?
A: No, the process is stereospecific regarding the cyclopropane ring; it can utilize cis/trans or (+)/(-) mixtures of chrysanthemic esters without altering the stereochemical integrity of the final aldehyde product.
Q: What are the primary cost drivers eliminated by this new catalytic route?
A: The primary cost drivers eliminated are the capital expenditure for specialized ozone generation equipment and the recurring operational expense of purchasing expensive stoichiometric oxidants like periodic acid.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,3-Dimethyl-2-Formylcyclopropanecarboxylate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to stay competitive in the global agrochemical market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch. By leveraging the catalytic efficiencies described in patent CN1186310C, we can offer our partners a supply solution that balances cost-effectiveness with uncompromising quality standards.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project requirements. Whether you need a Customized Cost-Saving Analysis to evaluate the economic impact of switching to this greener process or require specific COA data to validate our quality capabilities, we are ready to assist. Contact us today to request route feasibility assessments and discover how our expertise in complex intermediate synthesis can drive value for your organization.
