Advanced Synthesis of 2-Cyanoimino-1,3-Thiazolidine: Enhancing Purity and Scalability for Global Supply Chains
The chemical industry constantly seeks robust methodologies for producing high-value heterocyclic intermediates, and patent CN1220687C presents a significant breakthrough in the synthesis of 2-cyanoimino-1,3-thiazolidine. This compound serves as a pivotal building block for various neonicotinoid insecticides and pharmaceutical agents, where impurity profiles can dictate the efficacy and safety of the final active ingredient. The disclosed technology addresses long-standing challenges regarding the stability of the key N-cyanocarbonimidic acid ester intermediate, which has historically plagued manufacturers with low yields and decomposition issues. By introducing a novel stabilization step involving a reducing agent wash, the process ensures the isolation of high-purity crystalline intermediates that are suitable for long-term storage and reliable downstream processing. This advancement not only enhances the chemical integrity of the supply chain but also provides a scalable route that mitigates the risks associated with handling volatile and toxic reagents commonly found in legacy synthetic pathways.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of N-cyano carbamates relied heavily on the direct use of gaseous cyanogen chloride or the reaction of unstable precursors that decomposed rapidly under standard processing conditions. Prior art documents, such as European Patent EP 14,064, describe methods requiring strict airtight systems to handle toxic gases, creating substantial capital expenditure barriers for safe industrial implementation. Furthermore, conventional extraction and concentration techniques often resulted in significant product loss, with reported yields frequently hovering around merely 50% due to the thermal instability of the ester during solvent removal. The hygroscopic nature of these traditional intermediates made them prone to hydrolysis, complicating storage logistics and necessitating immediate consumption which disrupts continuous manufacturing flows. These inherent inefficiencies translated into higher production costs and inconsistent quality, forcing procurement teams to manage complex inventory strategies to avoid material degradation before the final cyclization step could occur.
The Novel Approach
The methodology outlined in CN1220687C fundamentally transforms the production landscape by generating the N-cyanocarbonimidic acid ester in an aqueous environment followed by a critical stabilization wash with a reducing agent solution. This innovative step effectively neutralizes oxidative impurities and prevents the decomposition pathways that typically degrade the ester during isolation, allowing for the recovery of stable, high-purity crystals. The process utilizes readily available raw materials such as sodium cyanide, sodium hydroxide, and methanol, reacting them with chlorine in a controlled manner to form the intermediate in situ without the need for handling gaseous cyanogen chloride directly. Subsequent extraction with organic solvents like chloroform, combined with precise pH control and low-temperature crystallization, ensures that the intermediate retains its structural integrity with purity levels exceeding 99%. This robust approach eliminates the need for specialized gas-handling infrastructure and significantly improves the overall mass balance of the synthesis, offering a commercially viable alternative to older, less efficient technologies.
Mechanistic Insights into Stabilized Cyclization Chemistry
The core of this synthesis lies in the careful management of the N-cyano carbamate intermediate, which acts as the electrophilic partner in the subsequent ring-closing reaction. In the first stage, the reaction of alkali metal cyanide with chlorine in the presence of an alcohol generates the reactive carbamate species, which is immediately protected from oxidative degradation by the introduction of a reducing agent such as sodium bisulfite during the workup phase. This chemical intervention is crucial because it scavenges residual oxidizing agents that would otherwise attack the sensitive cyano-imino functionality, thereby preserving the yield and purity of the isolated solid. The resulting intermediate is then dried thoroughly to remove trace water, as even minor moisture content can trigger hydrolysis back to the starting materials, underscoring the importance of rigorous dehydration protocols using molecular sieves or similar desiccants. By securing a stable, high-purity feedstock, the process sets the stage for a highly efficient second step where the intermediate reacts cleanly with nucleophiles.
In the second stage, the stabilized N-cyanocarbonimidic acid ester undergoes a nucleophilic substitution and cyclization reaction with 2-aminoethanethiol to form the target thiazolidine ring system. The reaction mechanism involves the initial attack of the thiol group on the electrophilic carbon of the carbamate, followed by intramolecular cyclization facilitated by the amine group under controlled alkaline conditions. Precise pH regulation between 10 and 11 is essential during this phase to ensure the amine is sufficiently nucleophilic while preventing the polymerization side reactions that can occur at higher pH levels. The use of 2-aminoethanethiol hydrochloride salt allows for better solubility and handling, with the free base generated in situ by the addition of sodium hydroxide to drive the reaction forward. This controlled environment minimizes the formation of tars and oligomers, resulting in a crude product that requires minimal purification to meet stringent commercial specifications for agrochemical applications.
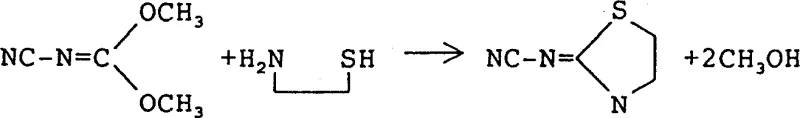
How to Synthesize 2-Cyanoimino-1,3-Thiazolidine Efficiently
The synthesis protocol detailed in the patent provides a clear roadmap for manufacturers aiming to implement this high-efficiency route in their own facilities. It begins with the preparation of the stabilized intermediate in an aqueous phase, followed by extraction and drying, before moving to the final cyclization step in a separate reactor. The detailed standardized synthesis steps below outline the specific molar ratios, temperature controls, and pH adjustments required to replicate the high yields and purity described in the intellectual property documentation. Adhering to these parameters is critical for maintaining the stability of the intermediate and ensuring the success of the final ring-closure reaction.
- Generate N-cyanocarbonimidic acid ester by reacting alkali metal cyanide, hydroxide, and lower alcohol with chlorine in aqueous solution, followed by stabilization with a reducing agent wash.
- Extract the stabilized intermediate using an organic solvent such as chloroform and dry thoroughly to prevent hydrolysis before the next step.
- React the purified intermediate with 2-aminoethanethiol hydrochloride in alkaline solution, adjusting pH and temperature to induce cyclization and crystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this stabilized synthesis route offers profound advantages in terms of cost predictability and logistical flexibility. The ability to isolate and store the N-cyano carbamate intermediate as a stable solid decouples the two main reaction steps, allowing manufacturers to produce the intermediate in large batches during optimal conditions and store it for future use. This decoupling significantly reduces the risk of production stoppages caused by upstream supply fluctuations, as the intermediate can serve as a strategic buffer stock. Furthermore, the elimination of gaseous cyanogen chloride handling reduces the regulatory burden and insurance costs associated with hazardous material management, leading to substantial indirect cost savings. The high purity of the final product also minimizes the need for expensive recrystallization or chromatography steps, streamlining the manufacturing workflow and reducing solvent consumption.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by improving overall yield through the stabilization of the key intermediate, which drastically reduces raw material waste compared to conventional low-yield methods. By avoiding the use of expensive and difficult-to-handle reagents like diphenyl esters or gaseous cyanogen chloride, the raw material bill of materials is simplified and made more economical. The reduction in side reactions and polymerization during the cyclization step means that less energy and solvent are required for purification, directly lowering the variable cost per kilogram of the final API intermediate. Additionally, the ability to recycle filtrates from the crystallization step further enhances the atom economy of the process, contributing to a leaner and more cost-effective production model.
- Enhanced Supply Chain Reliability: The stability of the isolated intermediate allows for extended shelf life, enabling manufacturers to build inventory buffers that protect against supply chain disruptions and demand spikes. Unlike previous methods where the intermediate had to be used immediately due to rapid decomposition, this technology supports a make-to-stock strategy that improves delivery reliability for downstream customers. The use of common industrial chemicals such as sodium cyanide and chlorine ensures that raw material sourcing is not dependent on niche suppliers, reducing the risk of supply bottlenecks. This robustness in the supply chain is critical for meeting the just-in-time delivery requirements of major agrochemical and pharmaceutical clients who prioritize continuity of supply above all else.
- Scalability and Environmental Compliance: The process is designed for easy scale-up from laboratory to commercial production, utilizing standard stainless steel reactors and common unit operations like liquid-liquid extraction and crystallization. The aqueous nature of the initial reaction step simplifies waste treatment, as the effluent contains primarily inorganic salts that can be managed through standard wastewater treatment protocols. By minimizing the generation of hazardous organic by-products and avoiding the release of toxic gases, the technology aligns with increasingly stringent environmental regulations and corporate sustainability goals. This environmental compliance reduces the risk of regulatory fines and shutdowns, ensuring long-term operational viability for manufacturing sites.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. They are derived from the specific advantages and operational parameters detailed in the patent documentation to assist decision-makers in evaluating the feasibility of adoption. Understanding these nuances is essential for R&D and operations teams planning to integrate this route into their existing manufacturing portfolios.
Q: Why is the stability of the N-cyano carbamate intermediate critical for industrial production?
A: Conventional methods often produce unstable intermediates that decompose during concentration, leading to yields as low as 50%. The patented stabilization method using a reducing agent wash ensures the intermediate remains stable for storage and transport, significantly improving overall process efficiency.
Q: How does this process improve safety compared to traditional cyanogen chloride methods?
A: Traditional routes often require handling gaseous cyanogen chloride, which is highly toxic and requires specialized airtight equipment. This method generates the necessary species in situ from safer solid precursors like sodium cyanide and chlorine in solution, drastically reducing occupational hazards.
Q: What purity levels can be achieved with this crystallization technique?
A: By utilizing specific pH controls and low-temperature crystallization after the reducing agent wash, the process consistently achieves purity levels exceeding 99%, minimizing the need for costly downstream purification steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Cyanoimino-1,3-Thiazolidine Supplier
At NINGBO INNO PHARMCHEM, we leverage advanced synthetic methodologies like the one described in CN1220687C to deliver high-performance intermediates that meet the rigorous demands of the global agrochemical and pharmaceutical markets. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that every batch meets stringent purity specifications and consistency standards. We operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify the identity and purity of our products, guaranteeing that our clients receive materials that facilitate smooth downstream processing. Our commitment to quality and technical excellence makes us a preferred partner for companies seeking to optimize their supply chains with reliable, high-quality chemical building blocks.
We invite you to contact our technical procurement team to discuss how our capabilities can support your specific project requirements and volume needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized processes can reduce your total cost of ownership. We encourage potential partners to reach out for specific COA data and route feasibility assessments to validate the suitability of our materials for your applications. Let us collaborate to drive efficiency and innovation in your chemical manufacturing operations.
