Scalable Synthesis of Chiral 3-(2-Nitroethyl) Tetrahydrofuran Compounds for Advanced Drug Discovery
Introduction to Next-Generation Chiral Tetrahydrofuran Scaffolds
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for higher purity and more sustainable synthetic routes. A pivotal advancement in this domain is documented in Chinese patent CN103755667A, which discloses a highly efficient preparation method for chiral 3-(2-nitroethyl) tetrahydrofuran compounds. These specific molecular scaffolds are not merely academic curiosities; they serve as critical building blocks in the total synthesis of complex natural products and potent medicinal agents. The tetrahydrofuran ring system is ubiquitous in bioactive molecules, ranging from the deoxyribose units in DNA to clinically vital antiviral and anticancer nucleoside drugs. Consequently, the ability to access these structures with high stereochemical control is paramount for modern drug discovery programs aiming to develop next-generation therapeutics.
The innovation presented in this patent addresses a long-standing challenge in organic synthesis: the difficulty of activating the 3-position of the tetrahydrofuran ring for stereoselective functionalization. While 2-substituted and 2,5-disubstituted analogues are well-documented, the 3-position has historically been recalcitrant to catalytic activation. The disclosed technology overcomes this barrier through a novel organocatalytic strategy that delivers products with exceptional enantiomeric purity. As illustrated in the general structural representation below, the versatility of the R1 group allows for the generation of a diverse array of derivatives suitable for various pharmacological applications.
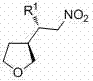
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this breakthrough, the synthesis of chiral 3-substituted tetrahydrofurans was fraught with significant inefficiencies that hindered large-scale production. A notable precedent in the literature, reported by Masafumi Hirano and colleagues in 2013, utilized a chiral ruthenium catalyst to effect an ene dimerization reaction. While chemically feasible, this transition metal-catalyzed approach suffered from severe practical drawbacks that rendered it suboptimal for industrial application. The most glaring issue was the excessively long reaction time, which extended up to seven days to reach completion, thereby tying up reactor capacity and increasing energy consumption disproportionately. Furthermore, the stereochemical outcome was disappointing, with enantiomeric excess (ee) values plateauing at merely 80%, necessitating costly and yield-eroding recrystallization or chromatographic purification steps to meet the stringent purity standards required for active pharmaceutical ingredients (APIs).
The Novel Approach
In stark contrast, the methodology outlined in patent CN103755667A represents a paradigm shift towards efficiency and precision. By employing a proline-derived chiral pyrrolidine as an organocatalyst, the inventors have achieved a dramatic reduction in reaction time, typically completing the transformation within 1 to 48 hours, with optimized conditions yielding results in just 12 hours. More impressively, the enantioselectivity is vastly superior, with ee values reaching as high as 99%, effectively eliminating the need for extensive chiral resolution downstream. This leap in performance is exemplified by specific embodiments where yields exceed 95% with near-perfect optical purity. For instance, the synthesis of the phenyl-substituted derivative demonstrates the robustness of this new protocol, delivering high-quality material suitable for immediate use in subsequent synthetic transformations without further enrichment.
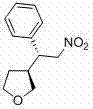
Mechanistic Insights into Proline-Derived Organocatalysis
The success of this synthetic route lies in the sophisticated design of the catalytic cycle, which leverages the unique properties of secondary amine organocatalysis. The core of the transformation is an asymmetric Michael addition reaction between 2-hydroxytetrahydrofuran and a nitroalkene. The catalyst, a chiral pyrrolidine derivative featuring a bulky silyl-protected hydroxyl group and an aryl moiety, acts as a Lewis base to activate the nucleophile while simultaneously organizing the transition state through hydrogen bonding interactions. This dual activation mode ensures that the attack on the nitroalkene occurs from a specific facial direction, thereby establishing the critical stereocenter at the 3-position of the tetrahydrofuran ring with high fidelity. The structural intricacies of this catalyst, particularly the steric bulk provided by the silyl group, are essential for shielding one face of the reactive intermediate, thus enforcing the observed high enantioselectivity.
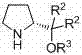
Following the initial carbon-carbon bond formation, the process incorporates a strategic reductive deoxygenation step to finalize the scaffold. The intermediate, which retains a hydroxyl group at the 2-position, is subjected to treatment with triethylsilane and boron trifluoride etherate. This Lewis acid-mediated reduction selectively removes the oxygen functionality without compromising the newly formed stereocenter or the sensitive nitro group on the side chain. This two-step sequence—organocatalytic addition followed by chemoselective reduction—is elegantly simple yet powerful. It avoids the use of harsh reducing agents that might otherwise lead to racemization or decomposition of the fragile heterocyclic core. The result is a clean conversion to the target 3-(2-nitroethyl) tetrahydrofuran, maintaining the integrity of the chiral information installed in the first step.
How to Synthesize Chiral 3-(2-Nitroethyl) Tetrahydrofuran Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to reaction parameters, although the procedure is designed to be operationally straightforward. The process begins with the dissolution of the nitroalkene substrate and 2-hydroxytetrahydrofuran in a suitable solvent such as chloroform or toluene. The addition of the chiral catalyst and a carboxylic acid additive, such as acetic acid or benzoic acid, initiates the Michael addition. Temperature control is flexible, ranging from 0°C to 50°C, allowing operators to balance reaction rate against selectivity based on specific substrate requirements. Once the intermediate is formed and isolated via standard aqueous workup, the second stage involves mixing the residue with triethylsilane in dichloromethane and slowly introducing boron trifluoride etherate. The detailed standardized operating procedures for scaling this reaction are provided in the technical guide below.
- Perform a Michael addition reaction between nitroalkenes and 2-hydroxytetrahydrofuran using a proline-derived chiral pyrrolidine catalyst in chloroform at 0-50°C.
- Purify the resulting intermediate via acid wash and solvent extraction to remove unreacted starting materials and catalyst residues.
- Execute a reductive deoxygenation using triethylsilane and boron trifluoride etherate at room temperature to yield the final chiral product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from transition metal catalysis to this organocatalytic protocol offers tangible economic and logistical benefits. The elimination of precious metals like ruthenium not only removes a significant line item from the raw material budget but also simplifies the regulatory burden associated with heavy metal residuals in final drug substances. This shift translates directly into cost reduction in pharmaceutical intermediate manufacturing, as the expensive scavenging resins and specialized filtration equipment required to meet ppm-level metal specifications are no longer necessary. Furthermore, the use of commodity chemicals such as proline derivatives and silanes ensures a stable and resilient supply chain, mitigating the risks associated with geopolitical fluctuations in the availability of rare earth or platinum group metals.
- Cost Reduction in Manufacturing: The economic argument for adopting this technology is compelling when analyzed through the lens of process mass intensity and operational expenditure. By drastically shortening the reaction time from a week-long ordeal to a matter of hours, manufacturers can achieve significantly higher throughput using existing reactor infrastructure. This acceleration reduces utility costs related to heating, cooling, and stirring over extended periods. Additionally, the high yields and exceptional enantiomeric purity minimize the loss of valuable starting materials during purification, ensuring that a greater proportion of input mass is converted into saleable product. The avoidance of cryogenic conditions, which are often required for high-selectivity metal catalysis, further lowers energy consumption and capital investment in specialized refrigeration equipment.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the reliance on single-source suppliers for complex ligands or exotic catalysts. This novel method utilizes catalysts derived from proline, an abundant and inexpensive amino acid, which can be sourced from multiple global vendors. This diversification of the supply base reduces the risk of production stoppages due to vendor shortages. Moreover, the robustness of the reaction conditions means that the process is less sensitive to minor variations in raw material quality or environmental factors, leading to more consistent batch-to-batch performance. This reliability is crucial for maintaining just-in-time inventory levels and meeting the rigorous delivery schedules demanded by downstream API manufacturers.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process aligns perfectly with the principles of green chemistry. The absence of toxic heavy metals simplifies waste stream management and reduces the cost of hazardous waste disposal. The solvents employed, such as chloroform and dichloromethane, are standard industrial solvents with well-established recovery and recycling protocols, facilitating a circular approach to solvent usage. The mild reaction temperatures and atmospheric pressure operations enhance plant safety by minimizing the risk of thermal runaways or high-pressure incidents. These factors collectively make the commercial scale-up of complex pharmaceutical intermediates smoother and more compliant with increasingly stringent global environmental regulations.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating this technology for their specific projects, we have compiled answers to common inquiries regarding the scope and limitations of the patent. These insights are derived directly from the experimental data and comparative analysis provided in the intellectual property documentation. Understanding these nuances is essential for project managers assessing the feasibility of integrating this route into their existing development pipelines. The following responses address key concerns regarding substrate compatibility, purification strategies, and potential scale-up challenges.
Q: What are the primary advantages of this organocatalytic method over traditional transition metal catalysis?
A: This method eliminates the need for expensive and toxic transition metals like Ruthenium, significantly reducing reaction time from seven days to under 20 hours while improving enantiomeric excess from 80% to over 99%.
Q: What is the scope of substituents compatible with this synthetic route?
A: The process demonstrates excellent tolerance for various R1 groups including aryl, substituted aryl (nitro, halogen, alkoxy), aromatic heterocycles, and alkyl chains, allowing for diverse library synthesis.
Q: How does this technology impact the cost of goods for antiviral drug intermediates?
A: By utilizing readily available organocatalysts and mild reaction conditions without cryogenic requirements, the process drastically simplifies purification and lowers overall manufacturing costs compared to legacy methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral 3-(2-Nitroethyl) Tetrahydrofuran Supplier
The technical potential of the synthesis described in CN103755667A is immense, offering a clear pathway to high-value chiral building blocks that are currently difficult to source. At NINGBO INNO PHARMCHEM, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this laboratory innovation to the global market. Our facility is equipped with rigorous QC labs and advanced analytical instrumentation capable of verifying stringent purity specifications, ensuring that every batch of chiral tetrahydrofuran intermediate meets the exacting standards of the pharmaceutical industry. We understand that consistency is key in drug development, and our process engineering team is dedicated to optimizing every unit operation to maximize yield and optical purity.
We invite R&D directors and procurement specialists to collaborate with us to evaluate the feasibility of this route for your specific target molecules. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this organocatalytic method. We encourage you to contact us today to obtain specific COA data from our pilot batches and to discuss route feasibility assessments tailored to your project timelines. Let us help you secure a reliable supply of high-purity intermediates while driving down your overall cost of goods.
