Advanced Polyol-Carrier Strategy for High-Purity Gemfibrozil Manufacturing and Scale-Up
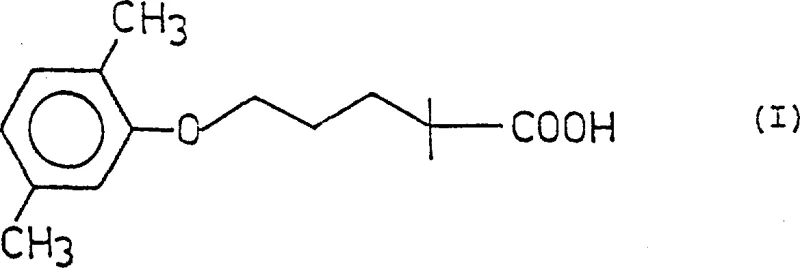
The pharmaceutical industry continuously seeks robust synthetic pathways for lipid-regulating agents, particularly for high-volume drugs like Gemfibrozil. Patent CN1032134C introduces a transformative approach to the preparation of 2,2-dimethyl-5-(2,5-dimethylphenoxy)pentanoic acid, addressing critical bottlenecks in yield and purity that have plagued conventional manufacturing. This innovation centers on a unique polyol-carrier strategy that fundamentally alters the reaction kinetics and workup procedures. By binding isobutyric acid moieties to a multifunctional alcohol backbone, the process eliminates the formation of troublesome azeotropes and accelerates key coupling steps. For R&D directors and procurement specialists, this represents a significant opportunity to optimize cost reduction in pharmaceutical intermediate manufacturing while ensuring supply chain reliability through simplified processing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Gemfibrozil has relied on two primary routes, both of which suffer from significant industrial drawbacks. Route A, involving the coupling of 2,5-dimethylphenol with a haloalkyl chain followed by reaction with isobutyric acid derivatives, typically yields poor overall conversion rates, often hovering between 39% and 46%. Furthermore, the intermediates generated in this pathway, particularly haloalkyl aryl ethers, are difficult to purify and often require recycling of unreacted materials. Route B, which alkylates isobutyric acid esters first, faces even more severe challenges regarding raw material handling. The use of isobutyl isobutyrate is problematic because it forms stable azeotropes with common organic solvents and water, making anhydrous conditions difficult to maintain without energy-intensive distillation processes using agents like phosphorus pentoxide. These complexities lead to substantial material loss and complicate the scale-up of complex pharmaceutical intermediates.
The Novel Approach
The methodology disclosed in CN1032134C circumvents these issues by employing a polyol carrier, such as 1,3-propanediol or ethylene glycol, to anchor the isobutyric acid units. This structural modification creates a polyester intermediate that possesses distinct physical properties compared to simple fatty esters. Crucially, these polyol-based intermediates do not form azeotropes with water or standard organic solvents, allowing for straightforward distillation and drying. This simplification translates directly into operational efficiency, as the need for specialized drying agents and complex solvent recovery systems is eliminated. Additionally, the steric and electronic environment provided by the polyol backbone appears to enhance the reactivity of the alpha-carbon, facilitating faster alkylation and subsequent etherification steps under milder conditions than previously possible.
Mechanistic Insights into Polyol-Carrier Mediated Synthesis
The core of this technological advancement lies in the strategic use of a multifunctional alcohol to act as a temporary scaffold. The process begins with the esterification of a polyol (Formula VIII) with isobutyric acid to generate a polyester (Formula IX). This step is critical because it converts a volatile, azeotrope-forming acid derivative into a stable, high-boiling intermediate. Following this, the alpha-carbon of the isobutyryl group is deprotonated using a strong base, such as lithium diisopropylamide, and alkylated with a dihalopropane. The resulting halogenated polyester (Formula X) serves as the key electrophile for the subsequent coupling reaction. The presence of the polyol chain likely influences the solubility and aggregation state of the enolate intermediate, contributing to the observed acceleration in reaction rates.
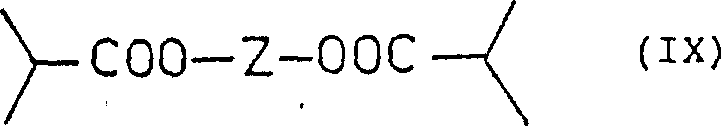
Following the alkylation, the terminal halogen atom is displaced by 2,5-dimethylphenol or its ester derivative. This nucleophilic substitution proceeds with remarkable speed; the patent data indicates completion in as little as 10 minutes at moderate temperatures, a stark contrast to the 6 to 13 hours required in traditional protocols. The final step involves hydrolysis, which cleaves the ester bonds linking the drug molecule to the polyol carrier. This releases the free acid (Gemfibrozil) and regenerates the polyol, which can potentially be recovered and reused. The mechanism ensures that impurities associated with incomplete reactions or side products are minimized, leading to a crude product that is sufficiently pure for direct recrystallization to meet stringent pharmacopoeial standards.
How to Synthesize Gemfibrozil Efficiently
Implementing this synthesis requires precise control over stoichiometry and reaction conditions to maximize the benefits of the polyol carrier system. The process generally involves three main stages: the formation of the polyester carrier, the introduction of the alkyl chain, and the final coupling and hydrolysis. Operators must ensure that the initial esterification is driven to completion, often utilizing azeotropic removal of water during this specific step to favor product formation. Subsequent alkylation demands strict temperature control to manage the exothermic nature of the base addition. The detailed standardized synthesis steps are outlined below to guide technical teams in replicating these high-efficiency results.
- Esterify a polyol carrier (e.g., 1,3-propanediol) with isobutyric acid to form a non-azeotropic polyester intermediate.
- Perform alpha-alkylation on the polyester using 1,3-dihalopropane and a strong base to introduce the propyl chain.
- React the halogenated intermediate with 2,5-dimethylphenol or its ester, followed by hydrolysis to release the final acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this polyol-carrier technology offers tangible benefits that extend beyond mere chemical yield. The elimination of azeotropic distillation requirements significantly reduces energy consumption and solvent usage, directly impacting the cost of goods sold. Furthermore, the dramatic reduction in reaction times—from over half a day to mere minutes for key steps—enhances plant throughput and asset utilization. This efficiency allows manufacturers to respond more agilely to market demand fluctuations without compromising on quality. The ability to produce high-purity pharmaceutical intermediates that meet USP specifications without extensive downstream purification also reduces waste generation and environmental compliance costs.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the removal of complex purification steps associated with azeotropic mixtures. Traditional methods require expensive drying agents like phosphorus pentoxide and energy-intensive distillation columns to separate isobutyl isobutyrate from solvents. By switching to polyol esters that do not form azeotropes, the process simplifies solvent recovery and raw material drying. This reduction in unit operations lowers both capital expenditure for equipment and operational expenditure for energy and consumables, resulting in substantial cost savings over the lifecycle of the product.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the complexity of sourcing and handling sensitive reagents. The new method utilizes readily available polyols and avoids the strict anhydrous constraints that plague the traditional isobutyl ester route. Since the intermediates are more stable and easier to handle, the risk of batch failure due to moisture contamination is significantly mitigated. This robustness ensures a more predictable production schedule, reducing lead times for high-purity pharmaceutical intermediates and providing a buffer against supply chain disruptions caused by processing delays.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety and environmental risks, particularly when dealing with explosive mixtures like sodium hydride in DMSO or prolonged heating at high temperatures. The polyol-carrier method operates under milder conditions and shorter durations, inherently reducing the thermal load and safety hazards associated with large-scale reactors. Additionally, the improved selectivity and yield mean less chemical waste is generated per kilogram of product. This aligns with modern green chemistry principles, facilitating easier regulatory approval and reducing the burden on waste treatment facilities, thereby supporting sustainable commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this method outperforms legacy technologies in terms of efficiency and product quality.
Q: How does the polyol carrier method improve purification compared to traditional isobutyl esters?
A: Traditional isobutyl isobutyrate forms difficult-to-separate azeotropes with solvents. The polyol-based intermediates described in CN1032134C do not form azeotropes, allowing for simple distillation and drying, which significantly streamlines the purification workflow.
Q: What are the reaction time advantages of this novel synthetic route?
A: The patent data indicates that the etherification step using the polyol intermediate can be completed in approximately 10 minutes under mild conditions, whereas conventional methods often require 6 to 13 hours at elevated temperatures, drastically improving throughput.
Q: Does this process meet pharmacopoeial purity standards?
A: Yes, the method is specifically designed to overcome the purity limitations of prior art. By avoiding complex side reactions and enabling effective decolorization and recrystallization, the final product consistently meets United States Pharmacopoeia (USP) specifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Gemfibrozil Supplier
At NINGBO INNO PHARMCHEM, we understand that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust infrastructure. Our team specializes in adapting complex synthetic pathways, such as the polyol-carrier strategy for Gemfibrozil, to fit large-scale production environments. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in practice. Our stringent purity specifications and rigorous QC labs guarantee that every batch meets the highest international standards, providing peace of mind for your regulatory filings.
We invite you to collaborate with us to evaluate the feasibility of this advanced manufacturing route for your specific supply chain needs. Our technical team is prepared to conduct a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating exactly how this process can improve your margins. Please contact our technical procurement team today to request specific COA data and comprehensive route feasibility assessments, and let us help you secure a competitive advantage in the cardiovascular therapeutics market.
