Revolutionizing Beta-Amino Acid Production: A High-Enantioselective Commercial Strategy
Revolutionizing Beta-Amino Acid Production: A High-Enantioselective Commercial Strategy
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to access complex chiral building blocks, and the technology disclosed in patent CN1168883A represents a significant leap forward in this domain. This patent details a novel, highly efficient, and enantioselective process for the preparation of enantiomerically pure cyclopentane-beta-amino acids of the general formula (I). These compounds are critical scaffolds in the development of advanced therapeutic agents, particularly those requiring rigid conformational constraints to enhance binding affinity and metabolic stability. The breakthrough lies in the strategic combination of asymmetric alcoholysis and Curtius rearrangement, which allows manufacturers to bypass the tedious multi-step sequences traditionally associated with these molecules. By leveraging chiral amine bases and specific allyl alcohol derivatives, the process achieves exceptional stereocontrol right from the initial ring-opening of meso-dicarboxylic acid anhydrides. This technical advancement not only addresses the longstanding challenge of obtaining high optical purity but also fundamentally alters the economic landscape of producing these valuable intermediates.
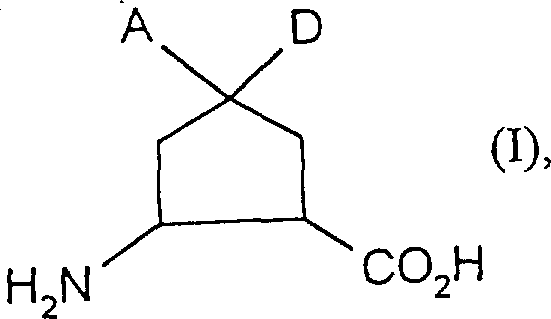
For R&D directors and process chemists, the implications of this methodology are profound, as it offers a robust route to access diverse derivatives where substituents A and D can vary from hydrogen to halogens or alkyl groups. The ability to tune these structural elements while maintaining a consistent, high-yielding core synthesis provides immense flexibility for medicinal chemistry campaigns. Furthermore, the process is designed to be scalable, utilizing inert solvents and standard reagents that are readily available in the global supply chain. As we delve deeper into the mechanistic nuances and commercial advantages, it becomes clear that this patent provides a blueprint for next-generation manufacturing of beta-amino acids, positioning adopters of this technology as leaders in cost-effective and high-quality chemical production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of enantiomerically pure pentamethylene-beta-amino acids was fraught with inefficiencies that hampered both research timelines and commercial viability. Traditional methods, such as those referenced in earlier literature like EP571800 or WO 95/19337, typically required a cumbersome six-step synthesis sequence starting from corresponding dicarboxylic anhydrides. These legacy processes often suffered from mediocre overall yields, frequently ranging between only 28% to 40% of the theoretical value, which translates to significant material loss and increased waste generation. Moreover, achieving high enantiomeric purity often necessitated complex resolution steps or the use of expensive chiral auxiliaries that were difficult to recover and recycle. The reliance on Hofmann rearrangements in some older protocols introduced additional safety hazards and operational complexities due to the handling of hazardous intermediates. Consequently, procurement managers faced inflated costs due to low throughput, while supply chain heads struggled with the unpredictability of long synthesis cycles that were prone to bottlenecks at multiple purification stages.
The Novel Approach
In stark contrast, the novel approach outlined in CN1168883A streamlines the entire production workflow by condensing the synthesis into just three major stages, effectively halving the operational complexity compared to prior art. This method initiates with an asymmetric alcoholysis of meso-dicarboxylic acid anhydrides using allyl alcohols in the presence of enantiomerically pure chiral amine bases, directly yielding enantiomerically pure dicarboxylic acid monoesters. This is followed by a seamless conversion into acid azides and a subsequent Curtius rearrangement to generate isocyanates, which are then trapped with allyl alcohols to form stable urethane intermediates. One of the most transformative aspects of this new route is the physical state of the intermediates; unlike the oils often encountered in traditional synthesis, the key intermediates of general formula (VII) are crystalline solids. This crystallinity allows for simple isolation via filtration and recrystallization, which acts as a powerful purification engine to boost enantiomeric excess to greater than 99%. The result is a process that delivers total recovery of greater than or equal to 45% of the theoretical value, representing a substantial improvement in material efficiency and cost structure.
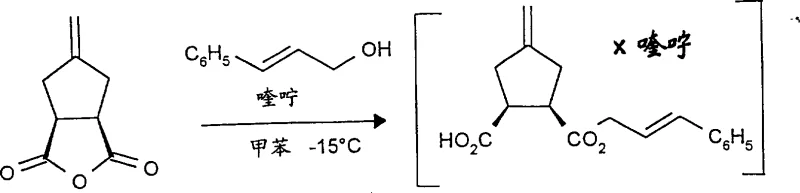
Mechanistic Insights into Asymmetric Alcoholysis and Curtius Rearrangement
The core of this technological breakthrough relies on a sophisticated interplay between stereoselective ring-opening and rearrangement chemistry. The process begins with the reaction of a meso-dicarboxylic acid anhydride with an allyl alcohol, such as cinnamyl alcohol, mediated by a chiral amine base like quinine or quinidine. This asymmetric alcoholysis is the enantio-determining step, where the chiral base directs the nucleophilic attack of the alcohol onto one of the carbonyl carbons of the anhydride, breaking the symmetry of the meso compound. The reaction proceeds through an enantiomerically pure salt intermediate, which ensures that the resulting dicarboxylic acid monoester possesses high optical purity, typically exceeding 85% ee initially and reaching up to 98% ee after simple processing. The choice of solvent plays a critical role here, with inert organic solvents like toluene, ether, or tetrahydrofuran providing the optimal environment for this stereochemical differentiation. This step effectively sets the chiral foundation for the entire molecule, eliminating the need for downstream resolution techniques that often discard half of the produced material.
Following the establishment of chirality, the monoester undergoes a Curtius rearrangement, a classic yet highly effective transformation for converting carboxylic acids into amines with retention of configuration. In this specific protocol, the carboxylic acid group is activated, often using diphenylphosphoryl azide or via an acyl azide intermediate formed with sodium azide, and then heated to induce the migration of the alkyl group with the expulsion of nitrogen gas. This generates a highly reactive isocyanate intermediate of general formula (VI). Crucially, the patent highlights that during the formation or subsequent handling of these isocyanates, a crystallization phenomenon occurs that further purifies the material, concentrating the enantiomeric excess to above 99%. The isocyanate is then immediately reacted with another equivalent of allyl alcohol to form a stable urethane derivative, protecting the newly formed amine functionality. Finally, the urethane and ester protecting groups are cleaved simultaneously using palladium catalysts and nucleophilic scavengers, releasing the free cyclopentane-beta-amino acid. This mechanistic elegance ensures that the stereochemical integrity established in the first step is preserved throughout the entire sequence, delivering a product of exceptional quality.
How to Synthesize Cyclopentane-Beta-Amino Acids Efficiently
The synthesis of these high-value intermediates requires precise control over reaction conditions to maximize yield and optical purity. The process is designed to be operationally straightforward, beginning with the dissolution of the meso-anhydride in an inert solvent like toluene under a nitrogen atmosphere at low temperatures, typically around -15°C. The addition of the chiral base and allyl alcohol must be carefully managed to maintain the thermal profile necessary for high enantioselectivity. Following the isolation of the monoester, the Curtius rearrangement is conducted at elevated temperatures, generally between 60°C and 110°C, ensuring complete conversion to the isocyanate before trapping with alcohol. The final deprotection step utilizes palladium catalysis in solvents such as ethyl acetate or ethanol, often with morpholine as a scavenger, to cleanly reveal the target amino acid. For detailed standard operating procedures and specific molar ratios, please refer to the technical guide below.
- Perform asymmetric alcoholysis of meso-dicarboxylic acid anhydrides using allyl alcohols and a chiral amine base to form enantiomerically pure dicarboxylic acid monoesters.
- Convert the monoesters into acid azides and subsequently rearrange them into isocyanates via the Curtius rearrangement protocol.
- React the isocyanates with allyl alcohols to form urethane intermediates, followed by catalytic cleavage to yield the final amino acid.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthesis route offers compelling advantages that directly address the pain points of modern chemical procurement and supply chain management. The reduction of the synthesis from six steps to three stages fundamentally alters the cost basis of production by minimizing labor, energy consumption, and solvent usage. Fewer unit operations mean less equipment occupancy time and a smaller physical footprint required for manufacturing, which translates to lower overhead costs per kilogram of product. Furthermore, the high overall yield of greater than 45% theoretical value significantly reduces the amount of raw materials required to produce a fixed quantity of the final API intermediate, providing a direct buffer against volatility in raw material pricing. For procurement managers, this efficiency means a more predictable and stable cost structure, allowing for better long-term budgeting and contract negotiations with downstream pharmaceutical clients.
- Cost Reduction in Manufacturing: The elimination of multiple isolation and purification steps inherent in the older six-stage processes leads to substantial cost savings in terms of consumables and waste disposal. By avoiding the use of complex resolution agents and reducing the number of solvent swaps, the process minimizes the environmental burden and the associated costs of waste treatment. The ability to isolate intermediates as crystalline solids via simple filtration rather than energy-intensive chromatography or distillation further drives down operational expenditures. Additionally, the recovery of the chiral amine base, which can be regenerated using mineral acids, adds another layer of economic efficiency by reducing the net consumption of expensive chiral reagents.
- Enhanced Supply Chain Reliability: The robustness of this chemical route enhances supply chain reliability by reducing the risk of batch failures associated with complex multi-step syntheses. The crystalline nature of the intermediates ensures consistent quality and ease of handling, reducing the likelihood of degradation or contamination during storage and transport. This stability allows for the strategic stocking of key intermediates, enabling manufacturers to respond more rapidly to fluctuations in market demand without compromising on lead times. The use of commercially available and stable reagents like cinnamyl alcohol and common chiral alkaloids ensures that the supply of starting materials is secure and not subject to the bottlenecks often seen with exotic or custom-synthesized reagents.
- Scalability and Environmental Compliance: This process is inherently scalable, having been demonstrated to work effectively with standard industrial solvents like toluene and ethyl acetate which are well-understood in large-scale reactor systems. The high atom economy of the Curtius rearrangement and the efficient use of reagents contribute to a greener manufacturing profile, aligning with increasingly stringent global environmental regulations. The reduction in waste generation and the potential for solvent recycling make this route attractive for companies aiming to improve their sustainability metrics. Scalability is further supported by the fact that the reaction conditions, such as temperature and pressure, are within standard operating ranges for existing chemical infrastructure, facilitating a smooth transition from pilot plant to commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this technology into their existing manufacturing portfolios. The answers provided are derived directly from the experimental data and claims within the patent documentation, ensuring accuracy and relevance for technical decision-makers.
Q: What is the primary advantage of this new synthesis method over conventional routes?
A: The primary advantage is the significant reduction in synthetic steps from six stages down to three major stages, which drastically improves overall yield to over 45% of theoretical value while maintaining enantiomeric excess greater than 99%.
Q: How is high optical purity achieved in this process?
A: High optical purity is achieved through asymmetric alcoholysis using chiral amine bases like quinine or quinidine, followed by a crystallization step during the formation of the isocyanate intermediate which further concentrates the enantiomeric excess to above 99%.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the process is highly suitable for scale-up because the key intermediates, particularly the general formula (VII) compounds, are crystalline solids that can be easily isolated by filtration, simplifying purification and handling compared to oil-based intermediates in older methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cyclopentane-Beta-Amino Acids Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthesis technology described in CN1168883A for the production of high-purity pharmaceutical intermediates. As a leading CDMO partner, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in a practical, industrial setting. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including the critical enantiomeric excess values that define the quality of these beta-amino acids. We are committed to delivering products that meet the exacting standards of the global pharmaceutical industry, leveraging our expertise in asymmetric synthesis and rearrangement chemistry to provide a secure and high-quality supply.
We invite potential partners to engage with our technical procurement team to discuss how this advanced manufacturing route can optimize your supply chain and reduce costs. By requesting a Customized Cost-Saving Analysis, you can gain specific insights into how adopting this 3-stage process can impact your bottom line compared to traditional methods. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project needs, ensuring that your development timelines are met with the highest quality intermediates available in the market.
