Advanced Ferriporphyrin Catalysis for Efficient Trifluoroethylation of Aromatic Amines
Advanced Ferriporphyrin Catalysis for Efficient Trifluoroethylation of Aromatic Amines
The pharmaceutical and agrochemical industries are constantly seeking robust methodologies to introduce fluorinated motifs into complex molecular scaffolds, driven by the unique metabolic stability and lipophilicity these groups confer. Patent CN108997144B introduces a groundbreaking approach for the synthesis of N-trifluoroethylated aromatic amines utilizing a ferriporphyrin catalyst system. This technology represents a significant departure from traditional high-energy processes, enabling the transformation to proceed under mild, room temperature conditions with exceptional efficiency. By leveraging the unique redox properties of iron porphyrins, the method achieves high yields ranging from 45% to 93% across a diverse array of substrates. This innovation not only streamlines the synthetic route but also aligns with modern green chemistry principles by minimizing energy consumption and hazardous waste generation.
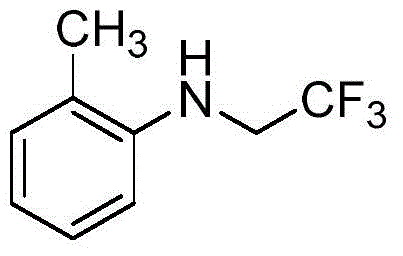
The versatility of this catalytic system is evident in its tolerance for various functional groups, allowing for the direct modification of aniline derivatives without extensive protecting group strategies. The process utilizes readily available starting materials such as trifluoroethylamine hydrochloride and sodium nitrite, which are converted in situ to the active trifluorodiazoethane species. This eliminates the logistical challenges associated with handling unstable diazo compounds directly. Furthermore, the reaction operates in a biphasic water-organic solvent system, facilitating easier workup procedures compared to strictly anhydrous conditions required by many competing methodologies. For procurement teams, this translates to a reliable supply chain for high-purity trifluoroethylated aromatic amines with reduced dependency on exotic reagents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of N-trifluoroethylated aromatic amines has relied heavily on cross-coupling reactions involving aryl halides and 2,2,2-trifluoroethylamine, often necessitating the use of expensive palladium catalysts. These traditional routes frequently demand harsh reaction conditions, with temperatures soaring as high as 250°C to overcome kinetic barriers, which poses significant safety risks and energy costs. Additionally, alternative methods utilizing N-H insertion with pre-formed trifluorodiazonium salts require the isolation of these highly unstable and potentially explosive intermediates. Such isolation steps not only complicate the operational workflow but also introduce severe safety hazards that are difficult to mitigate on a commercial scale. The reliance on noble metals further exacerbates cost issues, as residual palladium must be rigorously removed to meet stringent pharmaceutical purity specifications, adding multiple purification stages to the manufacturing process.
The Novel Approach
In stark contrast, the ferriporphyrin-catalyzed method described in the patent offers a streamlined one-pot solution that operates efficiently at room temperature, drastically reducing the thermal energy input required for production. By generating the reactive trifluorodiazoethane species in situ from stable amine salts and nitrites, the process completely bypasses the need for isolating hazardous intermediates, thereby enhancing overall plant safety. The use of earth-abundant iron porphyrin catalysts replaces costly noble metals, offering a sustainable alternative that does not compromise on catalytic activity or substrate scope. This approach allows for the direct functionalization of a wide variety of aromatic amines, including those with electron-donating substituents which typically enhance reaction yields. The simplicity of the operation, combined with the mild conditions, makes this technology particularly attractive for the commercial scale-up of complex fluorinated intermediates.
Mechanistic Insights into Ferriporphyrin-Catalyzed Trifluoroethylation
The core of this technological advancement lies in the unique ability of the ferriporphyrin catalyst to mediate the transfer of the trifluoroethyl group under mild conditions. The reaction initiates with the diazotization of trifluoroethylamine hydrochloride by sodium nitrite in an acidic aqueous-organic medium, generating the reactive trifluorodiazoethane species in situ. The iron center within the porphyrin macrocycle then interacts with this diazo compound, likely forming a reactive metal-carbene or radical intermediate that facilitates the N-H insertion into the aromatic amine. This mechanism avoids the high activation energy barriers associated with thermal decomposition of diazo compounds, allowing the reaction to proceed smoothly at ambient temperatures. The specific electronic environment provided by the porphyrin ligand stabilizes the iron center, preventing catalyst deactivation and ensuring sustained turnover throughout the reaction cycle.
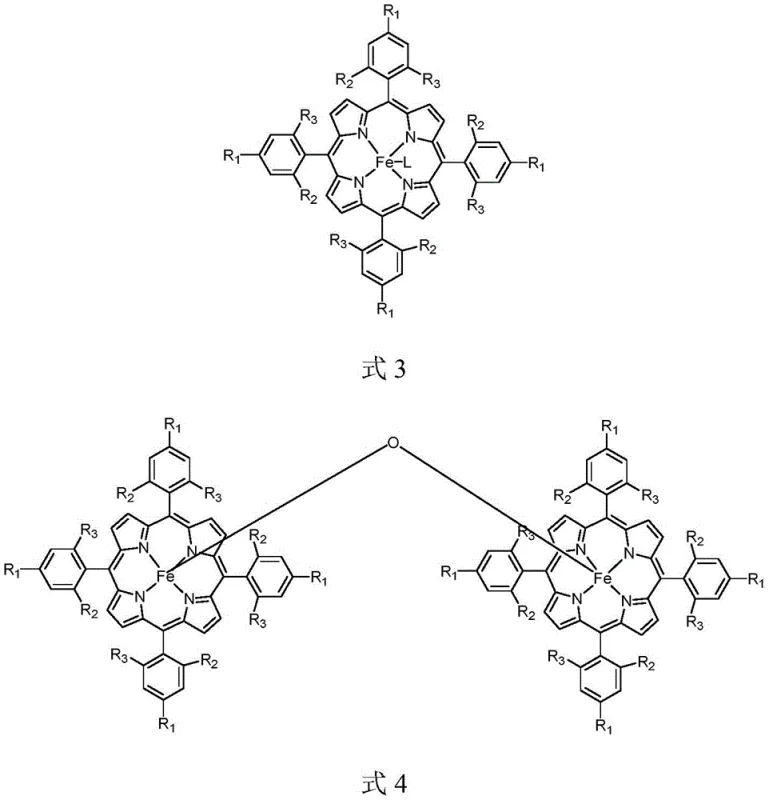
Impurity control is inherently managed by the selectivity of the iron porphyrin catalyst, which favors the desired N-trifluoroethylation over potential side reactions such as C-H insertion or dimerization of the diazo species. The biphasic nature of the solvent system, typically comprising water and dichloromethane, helps to partition inorganic byproducts like sodium salts into the aqueous phase, simplifying the downstream purification process. Furthermore, the mild acidic conditions prevent the protonation of the aromatic amine substrate to an extent that would inhibit nucleophilic attack, maintaining a balance that maximizes product formation. The catalyst loading is remarkably low, often in the range of 0.3% to 1.5% molar ratio relative to the substrate, which minimizes the introduction of metal contaminants into the final product. This high level of chemoselectivity ensures that sensitive functional groups on the aromatic ring, such as halogens or alkoxy groups, remain intact, preserving the structural integrity required for downstream drug synthesis.
How to Synthesize N-Trifluoroethylated Aromatic Amines Efficiently
The synthesis protocol outlined in the patent provides a robust framework for producing high-purity trifluoroethylated anilines suitable for pharmaceutical applications. The procedure begins with the preparation of the diazotization mixture, followed by the sequential addition of the amine substrate and the iron catalyst, all within a single reaction vessel. This one-pot strategy significantly reduces solvent usage and processing time compared to multi-step batch processes. Detailed standardized synthesis steps, including precise stoichiometric ratios and workup procedures, are provided in the guide below to ensure reproducibility and safety during implementation.
- Prepare an acidic solution system containing trifluoroethylamine hydrochloride and sodium nitrite to generate the diazonium species in situ.
- Add the aromatic primary amine substrate and the ferriporphyrin catalyst (e.g., TPPFeCl) to the reaction mixture at room temperature.
- Stir the reaction for approximately 12 hours, then purify the crude product via column chromatography using petroleum ether and acetone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this ferriporphyrin-catalyzed technology offers tangible benefits in terms of cost structure and operational reliability. The elimination of noble metal catalysts removes a major variable cost driver and mitigates the supply risk associated with fluctuating prices of precious metals like palladium. Moreover, the mild reaction conditions reduce the burden on utility systems, as there is no need for high-temperature heating or cryogenic cooling, leading to substantial cost savings in energy consumption. The one-pot nature of the reaction simplifies the manufacturing workflow, reducing the number of unit operations and the associated labor and equipment maintenance costs. These factors collectively contribute to a more resilient and cost-effective supply chain for critical fluorinated intermediates.
- Cost Reduction in Manufacturing: The replacement of expensive palladium catalysts with earth-abundant iron porphyrins results in a drastic reduction in raw material costs, while the low catalyst loading further enhances economic efficiency. The avoidance of high-temperature conditions lowers energy expenditures, and the simplified workup reduces solvent consumption and waste disposal fees. By eliminating the need for isolating unstable intermediates, the process also saves on specialized containment equipment and safety monitoring costs. These cumulative savings allow for a more competitive pricing structure for the final trifluoroethylated products without compromising on quality.
- Enhanced Supply Chain Reliability: The use of widely available starting materials such as trifluoroethylamine hydrochloride and sodium nitrite ensures a stable supply base that is less susceptible to geopolitical disruptions. The robustness of the iron catalyst means that production can be scaled up with confidence, as the reagents are not subject to the same supply constraints as rare earth metals. Additionally, the mild operating conditions reduce the risk of unplanned shutdowns due to equipment failure or safety incidents, ensuring consistent delivery schedules. This reliability is crucial for maintaining continuous production lines for active pharmaceutical ingredients that depend on these key intermediates.
- Scalability and Environmental Compliance: The green chemistry profile of this method, characterized by lower energy usage and reduced hazardous waste, aligns perfectly with increasingly stringent environmental regulations. The aqueous-organic solvent system facilitates easier recycling of solvents and treatment of effluent, minimizing the environmental footprint of the manufacturing process. The absence of heavy metal residues simplifies the regulatory approval process for new drug applications, as the impurity profile is cleaner and easier to characterize. This scalability and compliance make the technology an ideal candidate for long-term commercial production of high-value fluorinated chemicals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ferriporphyrin-catalyzed trifluoroethylation technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation. Understanding these aspects is essential for R&D teams evaluating the feasibility of integrating this method into their existing synthetic routes.
Q: What are the advantages of using ferriporphyrin over palladium catalysts for trifluoroethylation?
A: Ferriporphyrin catalysts operate under mild room temperature conditions and avoid the use of expensive noble metals like palladium, significantly reducing raw material costs and simplifying heavy metal removal processes.
Q: Does this method require isolation of unstable intermediates?
A: No, the process is a one-pot reaction where the trifluorodiazoethane intermediate is generated in situ, eliminating the need for hazardous isolation steps and improving operational safety.
Q: What is the substrate scope for this trifluoroethylation reaction?
A: The method supports a wide range of aromatic primary amines, including those with electron-donating groups like methoxy and methyl, as well as halogen-substituted anilines, yielding products with 45-93% efficiency.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N-Trifluoroethylated Aromatic Amine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the ferriporphyrin system in driving innovation within the pharmaceutical sector. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory breakthroughs are seamlessly translated into industrial reality. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of trifluoroethylated intermediate meets the exacting standards required for global drug development. Our commitment to technical excellence allows us to offer customized solutions that optimize both yield and cost for our partners.
We invite you to collaborate with us to leverage this cutting-edge synthesis method for your next project. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can enhance your supply chain efficiency and product quality.
