Advanced Manufacturing of Alpha-Ketol Unsaturated Fatty Acids for High-Value Applications
The chemical industry continuously seeks robust methodologies for constructing complex carbon skeletons, particularly for bioactive molecules like alpha-ketol unsaturated fatty acids. Patent CN1394198A introduces a transformative approach to synthesizing these valuable intermediates, specifically addressing the historical challenge of installing a double bond at the beta-position of the ketone moiety without compromising yield or purity. Traditional organic synthesis routes often struggled with double bond migration and low selectivity, resulting in complex mixtures that were difficult to purify on a commercial scale. This new method leverages a strategic coupling between mono-substituted acetylenes and epoxy compounds, establishing a reliable foundation for producing high-purity agrochemical intermediate and pharmaceutical building blocks. By fundamentally altering the bond formation strategy, this technology offers a pathway to significantly enhance production efficiency and material throughput for downstream applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art techniques for synthesizing alpha-ketol structures relied heavily on condensation reactions between carbonyl carbons and alpha-carbanions, such as those derived from dithianes or sulfonium salts. For instance, Reaction Formula I utilized samarium iodide to condense acyl chlorides and aldehydes, but this invariably produced complex mixtures where the desired product could not be isolated effectively. Similarly, Reaction Formula II involved lithiating dithiane derivatives, which led to uncontrollable side reactions and poor selectivity. Perhaps most critically, Reaction Formula III, which employed methylthiomethyl p-toluenesulfonates, suffered from extremely low yields, often reported at 20% or less, due to the instability of intermediates and the propensity for double bond transfer. These inefficiencies created significant bottlenecks in cost reduction in fine chemical manufacturing, as extensive purification steps were required to salvage minimal amounts of target material from crude reaction masses.
The Novel Approach
In stark contrast, the methodology disclosed in CN1394198A utilizes a nucleophilic addition of lithiated acetylenes to epoxy compounds, effectively constructing the carbon backbone in a single, high-yielding step. As illustrated in the reaction scheme below, this approach bypasses the unstable intermediates of previous methods, directly forming the propargylic alcohol structure which serves as a stable precursor. The patent data indicates that the yield for the key intermediate compound (4) can reach 85% or greater, representing a dramatic improvement over the sub-20% yields of legacy processes. This streamlined route minimizes waste generation and simplifies the downstream processing requirements, making it an ideal candidate for commercial scale-up of complex fatty acid derivatives. The ability to selectively reduce the triple bond to a cis-double bond further ensures that the final product meets the stringent stereochemical requirements necessary for biological activity.
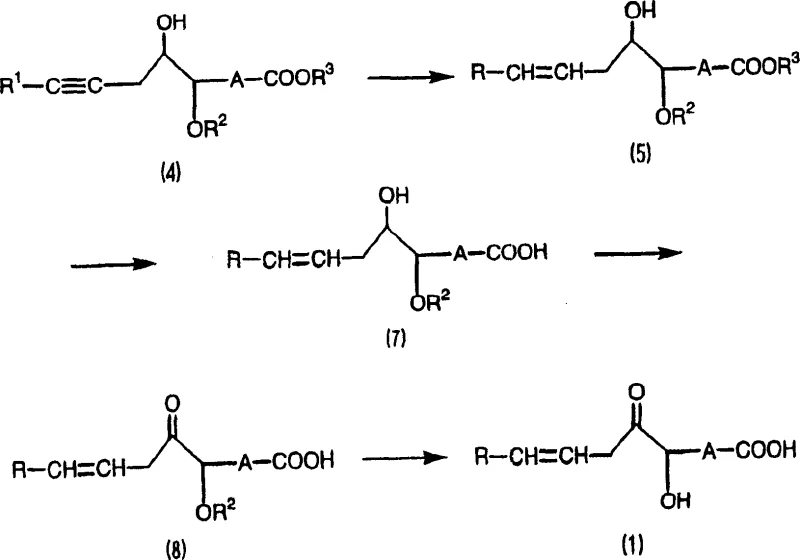
Mechanistic Insights into Acetylene-Epoxide Coupling and Oxidation
The core of this synthetic innovation lies in the controlled nucleophilic attack of a 1-lithiated acetylene species onto the less hindered carbon of an epoxide ring. This reaction is typically conducted at low temperatures, preferably -50°C or lower, using organolithium reagents like n-butyllithium in solvents such as THF or diethyl ether. The presence of Lewis acids like boron trifluoride diethyl etherate can further enhance regioselectivity and reaction rates. Following the coupling, the resulting propargylic alcohol undergoes a selective reduction of the triple bond. The patent specifies the use of nickel acetate-sodium borohydride systems or Lindlar-type catalysts to achieve cis-selectivity, which is crucial for the biological function of the final fatty acid. Subsequent oxidation of the secondary hydroxyl group to a ketone is achieved using mild oxidants like o-iodoxybenzoic acid or Jones reagent, ensuring that the sensitive cis-double bonds remain intact without isomerization.
Impurity control is inherently built into this mechanism by avoiding the harsh conditions that promote double bond migration. In conventional methods, the basic conditions required for enolate formation often triggered unwanted shifts in the double bond position, leading to regioisomers that were difficult to separate. By fixing the double bond geometry early through the selective hydrogenation of the alkyne, this new process locks in the desired Z-configuration before the final oxidation step. Furthermore, the use of robust protecting groups such as tert-butyldimethylsilyl (TBDMS) for hydroxyls and methyl esters for carboxyls ensures that functional groups remain inert during the lithiation and reduction phases. This orthogonal protection strategy allows for precise deprotection sequences at the end of the synthesis, delivering high-purity alpha-ketol unsaturated fatty acid with minimal chromatographic purification, thereby reducing lead time for high-purity intermediates in industrial settings.
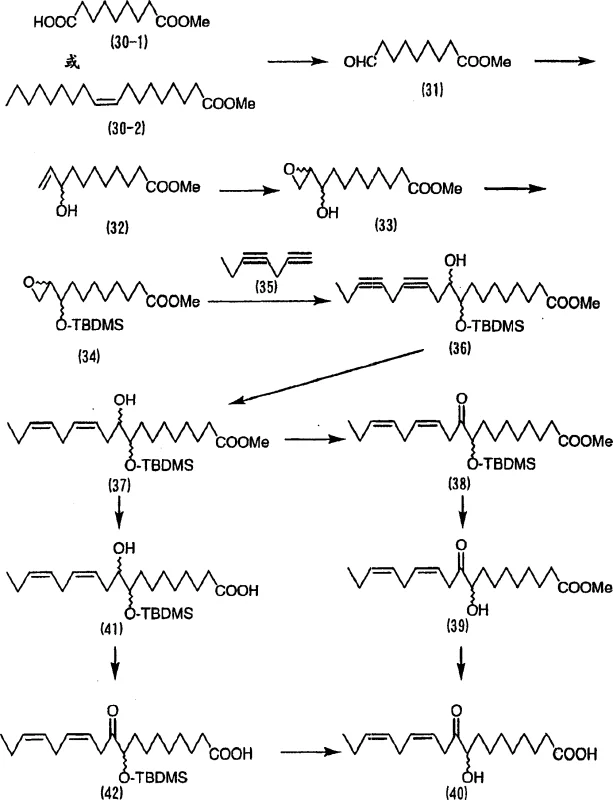
How to Synthesize Alpha-Ketol Unsaturated Fatty Acid Efficiently
The practical implementation of this synthesis involves a sequence of well-defined unit operations that are amenable to standard chemical manufacturing equipment. The process begins with the preparation of the epoxy starting material, which can be derived from readily available unsaturated fatty acid esters via epoxidation and protection steps. Once the epoxy component is ready, it is coupled with the appropriate acetylene derivative under strictly anhydrous conditions to prevent quenching of the lithiated species. The detailed standardized synthesis steps see the guide below for specific molar ratios and temperature profiles that maximize yield.
- React a mono-substituted acetylene with an epoxy compound using an organolithium base to form the propargylic alcohol intermediate.
- Selectively reduce the triple bond to a cis-double bond using a nickel or palladium catalyst system.
- Oxidize the secondary hydroxyl group to a ketone and remove protecting groups to yield the final alpha-ketol unsaturated fatty acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement professionals and supply chain managers, the adoption of this synthetic route offers tangible benefits regarding cost stability and material availability. By eliminating the need for exotic reagents like samarium iodide or complex dithiane precursors, the raw material costs are significantly reduced, relying instead on commodity chemicals like acetylenes and simple epoxides. The high yield of the key coupling step means that less starting material is required to produce the same amount of final product, directly impacting the cost of goods sold. Additionally, the simplified purification profile reduces the consumption of solvents and silica gel, contributing to substantial cost savings in waste disposal and environmental compliance. This efficiency translates into a more predictable pricing structure for buyers seeking a reliable agrochemical intermediate supplier.
- Cost Reduction in Manufacturing: The elimination of low-yielding steps and complex purification protocols drastically lowers the operational expenditure associated with production. Since the intermediate yields exceed 85%, the overall material throughput is maximized, meaning fewer batches are needed to meet demand. The avoidance of expensive transition metals and specialized ligands further decreases the direct material costs, allowing for competitive pricing in the global market without sacrificing margin.
- Enhanced Supply Chain Reliability: The starting materials for this process, such as mono-substituted acetylenes and fatty acid derivatives, are widely available from multiple global sources, reducing the risk of supply disruption. The robustness of the reaction conditions allows for flexible manufacturing schedules, as the process is less sensitive to minor variations in temperature or reagent quality compared to the finicky lithiation of dithianes. This reliability ensures consistent delivery timelines, which is critical for downstream formulators who depend on steady inputs for their own production lines.
- Scalability and Environmental Compliance: The chemistry described is inherently scalable, having been demonstrated in multi-gram to kilogram scales within the patent examples without loss of efficiency. The reduced use of heavy metals and toxic sulfur-containing byproducts simplifies the environmental permitting process and lowers the burden on wastewater treatment facilities. This aligns with modern green chemistry principles, making the facility more sustainable and reducing the regulatory risks associated with hazardous waste generation.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this patented technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on stereochemical control and reagent selection. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What is the primary advantage of the acetylene-epoxide coupling method over conventional dithiane lithiation?
A: The acetylene-epoxide coupling method avoids the formation of complex mixtures and double bond migration products common in dithiane lithiation, achieving intermediate yields of 85% or higher compared to less than 20% in older methods.
Q: Can this synthesis method produce optically active fatty acids?
A: Yes, by using optically active epoxy compounds with defined R or S configurations at the allylic position, the method preserves stereochemistry to produce optically active alpha-ketol unsaturated fatty acids.
Q: What protecting groups are recommended for the hydroxyl and carboxyl functionalities?
A: Ether-type protecting groups such as TBDMS, MOM, or MEM are preferred for hydroxyl protection, while methyl, ethyl, or benzyl groups are suitable for carboxyl protection to ensure stability during the lithiation and reduction steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Ketol Unsaturated Fatty Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient synthetic routes in maintaining a competitive edge in the fine chemical sector. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patent CN1394198A are fully realized in practical manufacturing environments. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of alpha-ketol unsaturated fatty acid meets the exacting standards required for agrochemical and pharmaceutical applications. Our commitment to quality assurance means that clients receive materials with verified stereochemistry and minimal impurity profiles, ready for immediate use in formulation or further synthesis.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis method can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this high-yield protocol. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your specific volume requirements and quality targets.
