Advanced Optical Resolution Technology for High-Purity Chiral Pharmaceutical Intermediates
Advanced Optical Resolution Technology for High-Purity Chiral Pharmaceutical Intermediates
The production of optically active 1,1'-binaphthol (BINOL) and its derivatives represents a critical bottleneck in the supply chain for modern asymmetric synthesis, serving as indispensable chiral ligands and intermediates for high-value pharmaceuticals. Patent CN1215028C introduces a groundbreaking optical resolution method that leverages supramolecular chemistry principles to separate racemic diphenols with exceptional efficiency and economic viability. This technology addresses the longstanding industry challenge of obtaining high-enantiomeric excess (ee) materials without relying on prohibitively expensive chiral auxiliaries or complex chromatographic separations. By utilizing readily available chiral sources such as L-glutamic acid to synthesize novel resolution reagents, this method establishes a robust pathway for the commercial scale-up of complex pharmaceutical intermediates. The core innovation lies in the selective formation of stable molecular crystals between the chiral reagent and one specific enantiomer of the racemate, driven by precise three-dimensional spatial complementarity and weak intermolecular interactions. This approach not only simplifies the operational workflow but also ensures that the resulting chiral materials meet the stringent purity specifications required by global regulatory bodies for API manufacturing.
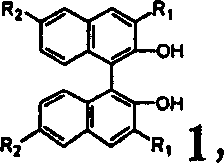
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional strategies for obtaining optically pure binaphthyl compounds have historically been plagued by significant economic and technical inefficiencies that hinder large-scale adoption in cost-sensitive markets. Conventional methods often rely on the direct asymmetric oxidative coupling of naphthol derivatives, which frequently suffers from moderate enantioselectivity and requires expensive transition metal catalysts that pose contamination risks in final drug products. Alternatively, enzymatic resolution techniques, while effective, are constrained by the high cost of biocatalysts, narrow substrate specificity, and the logistical complexities of maintaining enzyme activity under industrial conditions. Furthermore, classical diastereomeric salt formation methods often necessitate the use of stoichiometric amounts of costly chiral resolving agents that are difficult to recover and recycle, leading to substantial waste generation and inflated raw material costs. The reliance on preparative chiral HPLC for purification, although capable of high resolution, is fundamentally unsuitable for ton-scale manufacturing due to prohibitive solvent consumption and low throughput capabilities. These cumulative drawbacks create a fragile supply chain for critical chiral ligands, exposing pharmaceutical manufacturers to volatility in pricing and availability of key starting materials.
The Novel Approach
The methodology disclosed in CN1215028C revolutionizes this landscape by introducing a supramolecular inclusion crystallization technique that combines high selectivity with operational simplicity and economic sustainability. Instead of forming covalent bonds or relying on biological catalysts, this method exploits the subtle differences in spatial fit between a chiral host molecule and the guest enantiomers to drive selective crystallization. The process utilizes pyroglutamic acid derivatives, which are synthesized from abundant natural amino acids, acting as highly efficient resolving agents that can be used in sub-stoichiometric amounts relative to the racemate. This novel approach eliminates the need for complex reaction setups or hazardous reagents, reducing the process to straightforward steps of dissolution, heating, cooling, and filtration that are easily scalable in standard chemical reactors. Crucially, the method demonstrates remarkable versatility, successfully resolving a broad spectrum of substrates including 1,1'-biphenol, 1,1'-binaphthol, and their various halogenated or alkylated derivatives without requiring process re-optimization for each new compound. The ability to recover both enantiomers from the mother liquor and the clathrate crystals further maximizes atom economy, making this a superior choice for cost reduction in chiral ligand manufacturing.
Mechanistic Insights into Supramolecular Inclusion Crystallization
The efficacy of this resolution process is rooted in the precise molecular recognition capabilities of the pyroglutamic acid-derived resolving agents, which act as chiral hosts capable of distinguishing between enantiomeric guests. As illustrated by the general structure of the recommended reagents, the rigid pyrrolidone ring combined with specific amide or ester side chains creates a well-defined chiral pocket that preferentially accommodates one enantiomer of the racemic diphenol. This selectivity is driven by a network of weak non-covalent interactions, including hydrogen bonding between the hydroxyl groups of the binaphthol and the carbonyl or amine functionalities of the reagent, alongside pi-pi stacking and steric repulsion effects. When the racemic mixture interacts with the optically pure reagent in solution, the thermodynamically more stable diastereomeric complex precipitates out as a crystalline solid, effectively removing one enantiomer from the equilibrium. The remaining enantiomer stays in the mother liquor, allowing for its subsequent isolation through concentration and recrystallization, thereby achieving a complete separation of the optical isomers. This mechanism bypasses the kinetic limitations of enzymatic reactions and the equilibrium constraints of traditional salt formation, offering a more direct and controllable path to high optical purity.
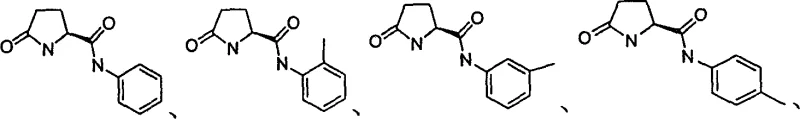
Impurity control in this system is inherently managed by the crystallization dynamics, as the lattice energy of the inclusion crystal favors the incorporation of the matched enantiomer while excluding mismatched species and chemical impurities. The high degree of stereoselectivity ensures that the initial precipitate possesses a significant enantiomeric excess, typically ranging from 70% to 80% ee, which can be further upgraded to greater than 99% ee through a single recrystallization step. This self-purifying nature of the crystallization process minimizes the burden on downstream purification units, reducing the need for extensive chromatographic polishing that often leads to product loss. Furthermore, the robustness of the supramolecular interaction means that minor variations in reaction conditions, such as temperature fluctuations or solvent composition, do not drastically compromise the optical purity of the final product. The method's tolerance to various substituents on the binaphthyl backbone, including halogens and alkoxy groups, indicates that the recognition mechanism is adaptable to electronic and steric changes, ensuring consistent quality across a diverse portfolio of chiral intermediates. This reliability is paramount for pharmaceutical applications where batch-to-batch consistency is a strict regulatory requirement.
How to Synthesize Optically Active Binaphthol Efficiently
Implementing this resolution technology in a production environment involves a streamlined sequence of unit operations that leverage standard chemical engineering equipment, ensuring a smooth transition from laboratory bench to pilot plant. The process begins with the preparation of the chiral resolving agent, typically achieved by condensing L-glutamic acid with an appropriate amine or alcohol under reflux conditions to form the active pyroglutamic derivative. Once the reagent is prepared, it is mixed with the racemic diphenol substrate in a selected organic solvent system, such as a mixture of tetrahydrofuran and ethanol, at a controlled molar ratio that optimizes crystal yield without excessive reagent usage. The mixture is heated to reflux to ensure complete dissolution and molecular interaction, followed by controlled cooling to induce the nucleation and growth of the diastereomeric inclusion crystals. Detailed standardized synthesis steps for specific derivatives and optimization parameters are provided in the guide below to assist technical teams in replicating these results.
- Prepare the optically pure resolution reagent (e.g., L-glutamic acid derivative) and dissolve the racemic diphenol in a suitable organic solvent.
- Mix the resolution reagent with the racemate at a molar ratio of 0.5-3: 1 and heat to reflux to form molecular crystals selectively.
- Filter the precipitated clathrate crystals, treat with organic solvent to release the optically active product, and recover the reagent from the mother liquor.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, this optical resolution method offers compelling advantages that directly address the pain points of cost volatility and supply continuity in the fine chemicals sector. By shifting away from proprietary enzymatic processes or scarce natural chiral pools, manufacturers can secure a more resilient supply chain for critical chiral building blocks that are less susceptible to market fluctuations. The simplicity of the operation, involving basic filtration and recrystallization rather than specialized bioreactors or high-pressure chromatography columns, significantly lowers the barrier to entry for contract manufacturing organizations looking to expand their chiral capabilities. This accessibility fosters a more competitive supplier landscape, driving down costs and ensuring that pharmaceutical companies have multiple qualified sources for these essential intermediates. Furthermore, the high recovery rate of the resolving agent means that the effective cost per kilogram of the resolved product is drastically reduced over time, as the initial investment in the chiral source is amortized over many production cycles. These factors combine to create a sustainable economic model that supports long-term partnerships between chemical suppliers and pharmaceutical innovators.
- Cost Reduction in Manufacturing: The economic viability of this process is primarily driven by the exceptional recoverability of the chiral resolution reagent, which can be reclaimed from the mother liquor or the dissociated complex with efficiency rates exceeding 90%. This high recovery capability transforms the resolving agent from a consumable expense into a reusable asset, substantially lowering the variable cost associated with each batch of production. Additionally, the method operates effectively with a molar ratio of reagent to substrate as low as 0.5:1, meaning that less chiral material is required to resolve a given amount of racemate compared to traditional stoichiometric methods. The elimination of expensive transition metal catalysts and the reduction in solvent usage due to the efficiency of crystallization further contribute to significant operational expenditure savings. Consequently, the overall cost structure for producing high-purity chiral ligands becomes much more favorable, enabling competitive pricing for downstream API synthesis.
- Enhanced Supply Chain Reliability: The reliance on L-glutamic acid, a commodity chemical produced on a massive scale for the food and feed industries, ensures that the raw materials for the resolving agent are abundant and price-stable. Unlike specialized enzymes or exotic chiral amines that may have limited suppliers and long lead times, the precursors for this technology are readily available from multiple global sources, mitigating the risk of supply disruptions. The robustness of the chemical synthesis of the resolving agent itself allows for rapid scale-up in response to surges in demand, ensuring that production capacity can be flexibly adjusted without lengthy qualification periods. This stability is crucial for pharmaceutical supply chains that require guaranteed continuity of supply to meet clinical trial timelines and commercial launch schedules. By adopting this method, procurement teams can diversify their supplier base and reduce dependency on single-source providers of critical chiral intermediates.
- Scalability and Environmental Compliance: The process is inherently scalable because it relies on fundamental unit operations such as crystallization and filtration that are well-understood and easily modeled from gram to ton scales. The patent data confirms that the yield and optical purity remain consistent regardless of the batch size, indicating that there are no hidden scale-up effects that would compromise product quality during commercialization. From an environmental standpoint, the method generates minimal waste, particularly since the resolving agent is recycled and the solvents used are common organic liquids that can be distilled and reused. The absence of heavy metals or toxic biological waste streams simplifies effluent treatment and helps manufacturing sites maintain compliance with increasingly stringent environmental regulations. This green chemistry profile not only reduces disposal costs but also aligns with the sustainability goals of modern pharmaceutical companies, enhancing the corporate social responsibility profile of the supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this supramolecular resolution technology, based on the detailed experimental data provided in the patent documentation. Understanding these aspects is vital for R&D directors and process engineers evaluating the feasibility of integrating this method into their existing workflows. The answers reflect the specific performance metrics and operational parameters observed during the development of this invention, ensuring accuracy and relevance for decision-makers.
Q: What is the primary advantage of this resolution method over enzymatic hydrolysis?
A: This supramolecular inclusion method utilizes inexpensive, naturally derived chiral sources like L-glutamic acid, avoiding the high cost and complexity associated with enzyme catalysts while achieving comparable or superior optical purity through simple crystallization.
Q: Can the chiral resolution reagent be reused in this process?
A: Yes, the patent explicitly states that the resolution reagent can be recovered from the mother liquor or the dissociated clathrate with a recovery rate exceeding 90%, allowing for multiple reuse cycles without significant loss of resolution efficiency.
Q: What types of solvents are compatible with this resolution technique?
A: The process is highly versatile regarding solvent choice, supporting common organic solvents such as dichloromethane, chloroform, benzene, toluene, tetrahydrofuran, methanol, ethanol, and mixtures thereof, facilitating easy adaptation to existing manufacturing infrastructure.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,1'-Binaphthol Supplier
As the demand for high-performance chiral ligands continues to grow in the pharmaceutical and agrochemical sectors, partnering with a manufacturer that possesses deep expertise in advanced resolution technologies is essential for maintaining a competitive edge. NINGBO INNO PHARMCHEM stands at the forefront of this field, leveraging cutting-edge methodologies like the supramolecular inclusion crystallization described in CN1215028C to deliver superior quality intermediates. Our facility is equipped with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements without compromising on the stringent purity specifications demanded by global regulators. Our rigorous QC labs employ state-of-the-art analytical techniques to verify enantiomeric excess and chemical purity, guaranteeing that every batch of 1,1'-binaphthol or its derivatives performs consistently in your asymmetric catalytic reactions.
We invite you to engage with our technical procurement team to discuss how our optimized resolution processes can drive value for your specific projects. By requesting a Customized Cost-Saving Analysis, you can gain insights into how switching to our supply chain can reduce your overall raw material costs and improve process efficiency. We encourage potential partners to contact us directly to obtain specific COA data for our current inventory and to schedule a consultation for route feasibility assessments tailored to your unique synthetic targets. Let us be your trusted partner in navigating the complexities of chiral synthesis and securing a reliable supply of critical pharmaceutical intermediates.
