Advanced Stereoselective Synthesis of 16-Phenoxy Prostaglandin Trienoic Acid Derivatives for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust synthetic pathways for complex bioactive molecules, particularly within the realm of eicosanoids where stereochemical integrity dictates biological efficacy. Patent CN85101840A presents a groundbreaking methodology for the manufacture of 16-phenoxy and 16-substituted phenoxy-prostaglandin trienoic acid derivatives, addressing long-standing challenges in stereoisomer resolution and molecular stability. This technology enables the production of specific stereoisomers, such as the R-allene configuration, which exhibits superior biological properties including potent gastric secretion inhibition and enhanced chemical stability compared to traditional oily prostaglandin mixtures. By leveraging a novel sequence involving stereospecific homologation and rearrangement reactions, this process allows for the selective manipulation of hydroxyl groups at C-9, C-11, and C-15 positions without inducing molecular degradation. For R&D directors and procurement specialists, understanding the nuances of this patented route is essential for securing a reliable prostaglandin intermediate supplier capable of delivering high-purity materials that meet stringent regulatory specifications for gastrointestinal therapeutics.
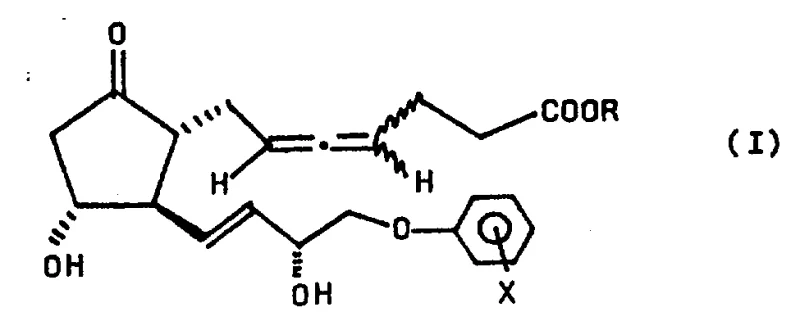
The limitations of conventional methods for synthesizing prostaglandin analogues often revolve around the difficulty in isolating single stereoisomers from complex reaction mixtures, leading to products that are viscous oils with poor stability profiles. Traditional approaches frequently result in racemic mixtures or diastereomers that require extensive and yield-limiting chromatographic separations late in the synthesis, complicating the supply chain and inflating manufacturing costs. Furthermore, standard protection strategies often lack orthogonality, meaning that removing a protecting group at one position might inadvertently affect others or cause elimination rearrangements to inactive 'B'-type prostaglandins. The novel approach detailed in this patent overcomes these hurdles by introducing a strategic sequence where a propargyl alcohol intermediate is resolved early in the synthesis. This early resolution ensures that subsequent steps, including the critical conversion to an allene compound, proceed with high stereochemical fidelity. The ability to produce a crystalline solid rather than an oil represents a paradigm shift in process chemistry, drastically simplifying downstream processing and quality control measures for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Stereospecific Claisen Rearrangement and Orthogonal Protection
The core mechanistic innovation lies in the transformation of a stereochemically pure propargyl alcohol into a corresponding allene compound via a stereospecific homologation and rearrangement reaction. This is achieved utilizing a trialkyl orthoacetate reagent in the presence of a catalytic amount of low molecular weight alkanoic acid, facilitating a Claisen rearrangement that preserves the stereochemical information established in the previous step. The reaction conditions are meticulously controlled, typically involving heating in a preheated oil bath to maintain temperatures between 100-130°C while distilling off byproducts to drive the equilibrium forward. This specific rearrangement is crucial because it locks the stereochemistry of the upper side chain, ensuring that the resulting allene moiety possesses the desired R or S configuration without racemization. Concurrently, the synthesis employs a sophisticated orthogonal protection strategy where C-11 and C-15 hydroxyls are masked with base-stable, acid-labile ether groups like tetrahydropyranyl, while the C-9 hydroxyl is protected with a base-labile silyl ether. This differentiation allows chemists to selectively deprotect and oxidize the C-9 position using strong bases or oxidants without compromising the integrity of the lower chain protecting groups, a feat that is mechanically impossible with non-orthogonal systems.
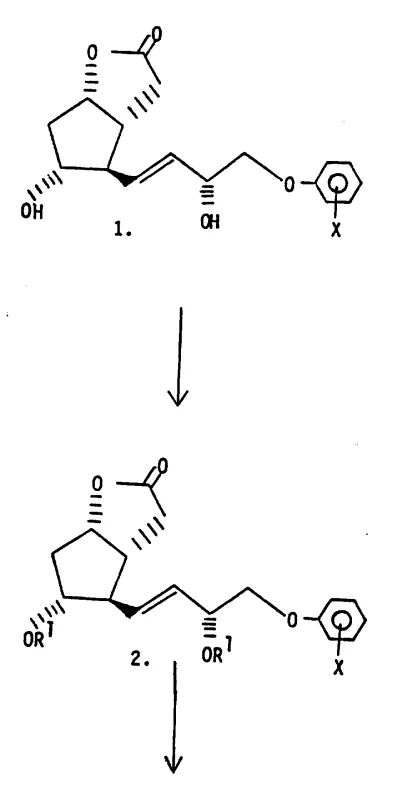
Impurity control is intrinsically built into this mechanistic design through the physical state of the intermediates and the specificity of the reagents used. The patent highlights that the specific R-allene stereoisomer spontaneously crystallizes upon cooling, a phenomenon that serves as a self-purification mechanism, excluding oily impurities and diastereomers from the crystal lattice. This crystallization behavior is distinct from the corresponding S-isomer or racemic mixtures which remain oils or waxy solids, providing a clear physical handle for purification that does not rely solely on expensive chromatography. Furthermore, the use of bulky silyl ether protecting groups at the C-9 position facilitates the initial chromatographic separation of propargyl alcohol epimers, which is a prerequisite for the high purity of the final allene product. By preventing the formation of elimination byproducts through the careful selection of acid-labile protecting groups that can be removed under mild conditions, the process minimizes the generation of degradation products that typically plague prostaglandin synthesis. This rigorous control over reaction pathways ensures that the final active pharmaceutical ingredient precursors meet the high purity standards required for clinical applications.
How to Synthesize 16-Phenoxy Prostaglandin Intermediates Efficiently
The synthesis of these high-value intermediates requires precise execution of multi-step organic transformations, beginning with the preparation of the appropriate phenoxylactone starting material and proceeding through a series of protection, rearrangement, and homologation steps. The detailed standardized synthesis protocols involve specific molar ratios of reagents, such as using a slight molar excess of base for lactone hydrolysis and controlling the temperature strictly during the Claisen rearrangement to prevent thermal degradation. Operators must adhere to inert atmosphere conditions, typically using dry nitrogen, to protect sensitive intermediates like metal acetylides and aldehydes from oxidation or moisture-induced decomposition. The following guide outlines the critical operational parameters necessary to replicate the high yields and stereochemical purity described in the patent documentation, serving as a foundational reference for process development teams aiming to implement this technology.
- Initiate the synthesis by selectively protecting the C-11 and C-15 hydroxyl groups of the phenoxylactone starting material with base-stable, acid-labile ether groups, followed by lactone ring opening and conversion to a protected aldehyde.
- React the aldehyde with a metal acetylene compound to form propargyl alcohol diastereomers, separate them via chromatography, and subject the pure isomer to a stereospecific Claisen rearrangement using trialkyl orthoacetate to generate the allene moiety.
- Perform chain homologation to extend the side chain, followed by selective oxidation at C-9 and final acidic hydrolysis to remove protecting groups without degrading the sensitive allene structure.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers substantial benefits for procurement managers and supply chain heads focused on cost reduction in pharmaceutical manufacturing and supply continuity. The ability to isolate the final product as a crystalline solid rather than an oil eliminates the need for complex lyophilization or specialized low-temperature storage logistics, thereby reducing warehousing costs and minimizing the risk of product degradation during transit. This physical stability translates directly into enhanced supply chain reliability, as the material can be stockpiled for longer durations without significant loss of potency, ensuring consistent availability for downstream drug formulation. Moreover, the stereospecific nature of the synthesis reduces the overall volume of waste solvents and silica gel associated with separating complex mixtures of isomers, aligning with modern environmental compliance standards and reducing the burden on waste treatment facilities. The streamlined process flow, characterized by fewer purification bottlenecks, allows for more predictable production schedules and shorter lead times for high-purity pharmaceutical intermediates, enabling manufacturers to respond more agilely to market demand fluctuations.
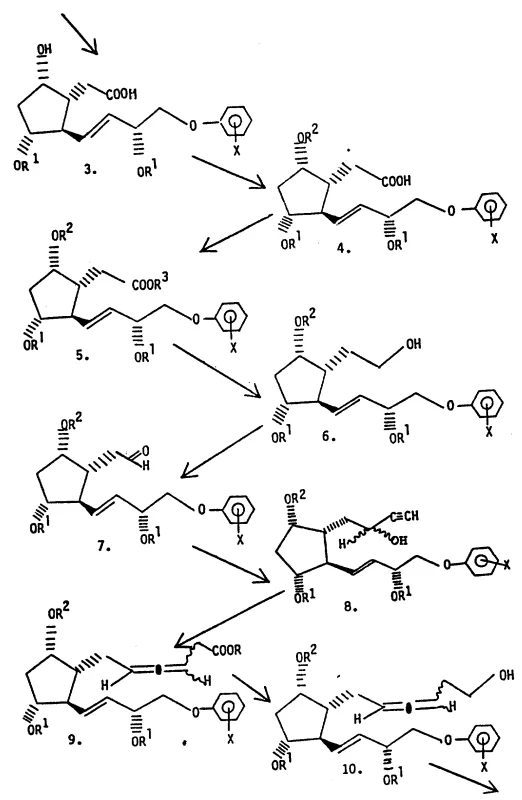
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts in favor of organocatalytic rearrangement steps and the reduction in chromatographic load due to early stereochemical resolution significantly lowers the cost of goods sold. By avoiding the need for expensive chiral columns or repeated recrystallizations of oily mixtures, the process achieves substantial cost savings in raw materials and consumables. The high yield of the crystalline product further amplifies these economic benefits by maximizing the output per batch, effectively distributing fixed operational costs over a larger quantity of saleable material.
- Enhanced Supply Chain Reliability: The robust nature of the intermediates, particularly the stability of the protected species and the final crystalline acid, ensures that the supply chain is less vulnerable to disruptions caused by material instability. The use of commercially available starting materials and standard reagents like trialkyl orthoacetates and silyl chlorides means that sourcing risks are minimized, as there is no dependence on exotic or single-source catalysts. This accessibility of raw materials guarantees a steady flow of production, mitigating the risk of stockouts that could delay the manufacturing of finished dosage forms.
- Scalability and Environmental Compliance: The reaction conditions described, such as the use of common solvents like dichloromethane and toluene under reflux, are readily scalable from laboratory to pilot and commercial plant scales without requiring specialized high-pressure or cryogenic equipment. The process design inherently minimizes the generation of hazardous byproducts, and the ability to recover and recycle solvents from the distillation steps during the rearrangement reaction supports sustainable manufacturing practices. This scalability ensures that the technology can meet global demand volumes while adhering to increasingly strict environmental regulations regarding solvent emissions and chemical waste disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route, providing clarity on its practical application and advantages over legacy methods. These insights are derived directly from the experimental data and claims within the patent specification, offering a transparent view of the technology's capabilities for potential partners and licensees. Understanding these details is crucial for making informed decisions about integrating this process into existing manufacturing portfolios.
Q: What is the primary advantage of the crystalline R-allene stereoisomer described in this patent?
A: Unlike typical prostaglandins which are viscous oils, the specific R-allene stereoisomer (Formula I') spontaneously crystallizes upon cooling. This solid state significantly facilitates purification, handling, and chemical analysis, while improving long-term stability and shelf life compared to oily counterparts.
Q: How does the orthogonal protection strategy prevent molecular degradation during synthesis?
A: The process utilizes base-stable, acid-labile groups for C-11 and C-15, and a base-labile group for C-9. This allows for the selective removal of the C-9 protecting group for oxidation under basic conditions without affecting the lower chain, followed by mild acidic deprotection of the remaining groups, preventing elimination rearrangements to 'B'-type prostaglandins.
Q: Why is the Claisen rearrangement critical for stereochemical purity in this route?
A: The conversion of the separated propargyl alcohol epimer to the allene compound via a trialkyl orthoacetate-mediated Claisen rearrangement is stereospecific. This ensures that the stereochemistry established during the chromatographic separation of the propargyl alcohol is faithfully transferred to the final allene product, avoiding the formation of complex diastereomeric mixtures.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 16-Phenoxy Prostaglandin Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of precision and reliability in the production of complex pharmaceutical intermediates like those described in patent CN85101840A. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the intricate stereochemical requirements of prostaglandin synthesis are met with unwavering consistency. We operate stringent purity specifications and maintain rigorous QC labs equipped with advanced analytical instrumentation to verify the crystalline nature and stereochemical integrity of every batch, guaranteeing that our clients receive materials that are ready for immediate formulation. Our commitment to quality extends beyond mere compliance; we actively optimize these processes to enhance yield and reduce environmental impact, positioning us as a strategic partner for long-term growth in the gastrointestinal therapeutic sector.
We invite you to engage with our technical procurement team to discuss how our manufacturing capabilities can support your specific project needs and timeline. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the economic advantages of switching to our supply chain for these critical intermediates. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to validate our technical expertise and secure a stable supply of high-quality 16-phenoxy prostaglandin derivatives for your drug development programs.
