Advanced Synthesis of Cis-4-O-Protected-2-Cyclopentenol Derivatives for High-Purity Pharmaceutical Intermediates
The pharmaceutical industry continuously demands higher purity and stereochemical precision in the synthesis of complex intermediates, particularly for potent therapeutic classes like immunosuppressants and prostaglandins. Patent CN1179770A discloses a robust and highly stereoselective methodology for the preparation of cis-4-O-protected-2-cyclopentenol derivatives, which serve as critical precursors in these value chains. This technology leverages a sophisticated Lewis acid-mediated reduction strategy combined with enzymatic kinetic resolution to overcome traditional challenges in controlling relative and absolute stereochemistry. By utilizing specific reaction conditions involving lithium halides and aluminum hydrides at controlled low temperatures, the process achieves exceptional cis-selectivity, minimizing the formation of unwanted trans-isomers and 1,2- or 1,4-addition byproducts. For R&D teams and procurement specialists seeking a reliable pharmaceutical intermediate supplier, understanding the mechanistic nuances of this patent provides a pathway to securing high-quality raw materials that meet stringent regulatory specifications for chiral drugs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes to cyclopentenol derivatives often struggle with poor stereocontrol during the reduction of cyclopentenones. Standard hydride reductions without specific modifiers frequently yield mixtures of cis and trans isomers, necessitating difficult and costly purification steps such as repeated chromatography or crystallization to isolate the desired cis-configuration. Furthermore, achieving optical purity typically requires the use of stoichiometric chiral auxiliaries or expensive transition metal catalysts, which introduce issues regarding heavy metal residues and environmental compliance. The lack of selectivity in conventional methods leads to significant material loss, increased waste generation, and unpredictable batch-to-batch consistency, posing substantial risks for commercial scale-up of complex pharmaceutical intermediates. These inefficiencies directly impact the cost of goods sold and can delay project timelines due to the need for extensive process optimization to meet purity thresholds.
The Novel Approach
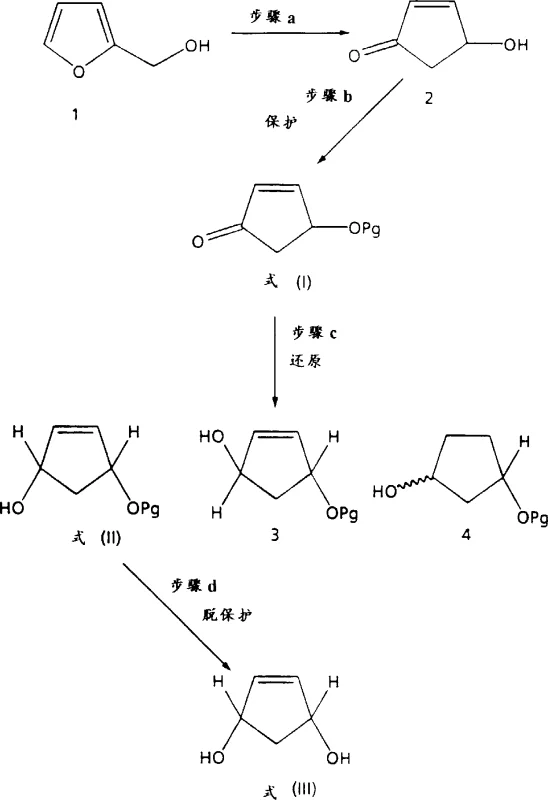
The methodology outlined in CN1179770A introduces a paradigm shift by employing a Lewis acid-modified reduction system that inherently favors the formation of the cis-isomer. By dissolving the 4-O-protected-2-cyclopentenone in anhydrous solvents like toluene or diethyl ether and treating it with lithium iodide or lithium bromide prior to the addition of lithium aluminum hydride, the reaction trajectory is altered to favor hydride delivery from the same face as the oxygen protecting group. This approach allows the reaction to proceed at temperatures ranging from -100°C to 20°C, with optimal results observed around -25°C, yielding cis-isomer ratios as high as 95.3%. Following the chemical reduction, the process utilizes enzymatic enantioselective acylation to resolve the racemic mixture, avoiding the need for chiral HPLC or derivatization with expensive chiral reagents. This dual-strategy ensures both high diastereoselectivity and enantiomeric excess, streamlining the production workflow significantly.
Mechanistic Insights into Lewis Acid-Mediated Stereoselective Reduction
The core innovation lies in the interaction between the Lewis acid and the substrate's carbonyl group. When lithium iodide is introduced to the reaction mixture containing the 4-O-protected-2-cyclopentenone, it coordinates with the carbonyl oxygen, creating a rigid chelated intermediate. This coordination locks the conformation of the five-membered ring and directs the approaching hydride species from the lithium aluminum hydride to attack from the less hindered face, which coincides with the orientation of the protecting group at the 4-position. This mechanistic pathway effectively suppresses the formation of the trans-alcohol and prevents over-reduction or conjugate addition side reactions that are common in unmodified reductions. The choice of solvent plays a pivotal role, with mixtures of toluene and tert-butyl methyl ether providing the ideal polarity and solubility profile to maintain the active catalytic species while ensuring efficient heat transfer during the exothermic reduction phase.
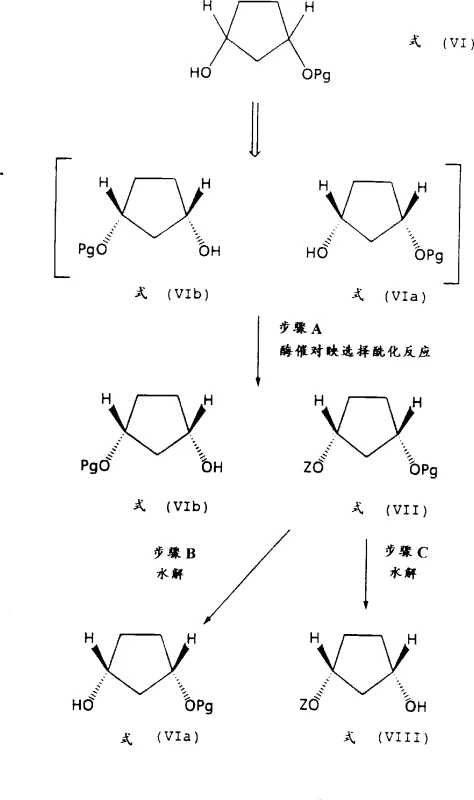
Following the chemical synthesis of the racemic cis-alcohol, the patent details a biocatalytic resolution step that exploits the subtle steric differences between enantiomers. Enzymes such as pancreatin or Candida antarctica lipase B are employed in the presence of vinyl acetate as an acyl donor. These biocatalysts exhibit high specificity, acylating only one enantiomer of the cis-alcohol to form an ester, while leaving the other enantiomer untouched. This kinetic resolution allows for the physical separation of the two enantiomers via standard chromatography or distillation, as the ester and the free alcohol possess distinct physical properties. The unreacted alcohol can be recovered with high enantiomeric excess, often exceeding 98% ee, while the ester can be hydrolyzed to yield the opposite enantiomer. This biological step complements the chemical reduction by providing the absolute stereochemical control necessary for active pharmaceutical ingredient (API) synthesis without the burden of toxic metal catalysts.
How to Synthesize Cis-4-O-Protected-2-Cyclopentenol Efficiently
The synthesis protocol described in the patent offers a scalable route that balances reaction speed with stereochemical fidelity. The process begins with the protection of 4-hydroxy-2-cyclopentenone using silyl chlorides or tetrahydropyranyl reagents to form the stable ketone precursor. Subsequent reduction under the optimized Lewis acid conditions yields the racemic cis-alcohol, which is then subjected to enzymatic resolution. The detailed standardized synthesis steps below outline the precise reagent ratios, temperature profiles, and workup procedures required to replicate the high yields and selectivity reported in the experimental examples, ensuring reproducibility for process chemists aiming to implement this technology.
- Dissolve the 4-O-protected-2-cyclopentenone precursor in an anhydrous organic solvent such as toluene or diethyl ether under an inert atmosphere.
- Treat the solution with a suitable Lewis acid like lithium iodide and a reducing agent such as lithium aluminum hydride at temperatures between -100°C and 20°C to achieve high cis-selectivity.
- Perform enzymatic enantioselective acylation using lipases or pancreatin with vinyl acetate to resolve the racemic cis-alcohol into pure enantiomers.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route offers tangible benefits related to cost stability and supply security. The reliance on readily available reagents such as lithium aluminum hydride, lithium iodide, and commercially sourced enzymes reduces dependency on exotic or single-source catalysts that often suffer from supply volatility. The high intrinsic selectivity of the chemical reduction step minimizes the load on downstream purification units, leading to significant savings in solvent consumption and chromatography media. Furthermore, the enzymatic resolution step operates under mild conditions, reducing energy consumption compared to high-temperature or high-pressure chiral hydrogenation processes. These factors collectively contribute to a more resilient supply chain capable of meeting the rigorous quality standards of the global pharmaceutical market.
- Cost Reduction in Manufacturing: The elimination of expensive chiral transition metal catalysts and the reduction in purification steps directly lower the variable costs associated with production. By achieving high cis-selectivity chemically, the process avoids the yield losses typically associated with separating difficult diastereomeric mixtures, thereby improving the overall mass balance and reducing the cost per kilogram of the final intermediate. The use of vinyl acetate as an acylating agent in the resolution step is also economically favorable due to its low cost and the irreversibility of the reaction driven by the formation of acetaldehyde byproduct.
- Enhanced Supply Chain Reliability: The reagents utilized in this process, including common Lewis acids and bulk enzymes, are sourced from a broad base of global suppliers, mitigating the risk of shortages. The robustness of the reaction conditions, which tolerate a range of temperatures and solvent mixtures, ensures that manufacturing can continue even if specific grades of solvents are temporarily unavailable. This flexibility allows for greater agility in sourcing strategies and reduces the likelihood of production stoppages due to raw material constraints, ensuring consistent delivery schedules for downstream API manufacturers.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing unit operations such as filtration, extraction, and distillation that are easily transferred from pilot plant to commercial scale. The avoidance of heavy metals in the resolution step simplifies waste treatment and reduces the environmental footprint of the manufacturing site. Additionally, the high selectivity reduces the generation of hazardous chemical waste associated with failed batches or extensive purification, aligning with modern green chemistry principles and facilitating easier regulatory approval for new drug applications.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis pathway. These insights are derived directly from the experimental data and claims within the patent documentation, providing clarity on reaction parameters and expected outcomes for technical teams evaluating this technology for integration into their existing manufacturing portfolios.
Q: What is the primary advantage of using lithium iodide in this reduction process?
A: Lithium iodide acts as a critical Lewis acid modifier that coordinates with the carbonyl oxygen, directing the hydride attack from the same face as the protecting group to ensure high cis-stereoselectivity, often exceeding 95% cis-isomer formation.
Q: How is optical purity achieved in the production of these intermediates?
A: Optical purity is achieved through enzymatic kinetic resolution, where specific enzymes like pancreatin or Candida antarctica lipase B selectively acylate one enantiomer over the other, allowing for easy separation and achieving ee values greater than 98%.
Q: What are the downstream applications of these cyclopentenol derivatives?
A: These derivatives serve as versatile chiral building blocks for the synthesis of cyclopentyl and cyclopentenyl purine analogs used as immunosuppressants, as well as various prostaglandin analogs essential for cardiovascular and inflammatory disease treatments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cis-4-O-Protected-2-Cyclopentenol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity intermediates play in the development of life-saving medications. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated stereochemical control described in CN1179770A is maintained at every stage of manufacturing. We operate stringent purity specifications and utilize rigorous QC labs equipped with advanced chiral HPLC and NMR capabilities to verify the identity and optical purity of every batch, guaranteeing that our clients receive materials that meet the highest international standards for pharmaceutical synthesis.
We invite you to collaborate with us to optimize your supply chain for cyclopentenol derivatives and related prostaglandin precursors. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us demonstrate how our manufacturing excellence can support your next breakthrough in immunosuppressant or cardiovascular therapy development.
