Advanced Photocatalytic Trifluoromethylation for High-Value Peptide Intermediates
Introduction to Next-Generation Peptide Functionalization
The pharmaceutical industry is constantly seeking more efficient pathways to modify bioactive molecules, particularly peptides, which play crucial roles in hormonal regulation and cellular function. Patent CN112062704B introduces a groundbreaking preparation method for trifluoromethylated polypeptide compounds, addressing the urgent need for mild and selective functionalization techniques. This technology leverages visible light photocatalysis to drive the direct introduction of trifluoromethyl groups onto the indole ring of tryptophan residues within polypeptide chains. By utilizing a mixed solvent system of DMF and water, the process adheres to modern green chemistry standards, eliminating the need for toxic organic solvents often associated with traditional fluorination methods. The innovation lies in its ability to achieve high site selectivity under room temperature conditions, preserving the delicate stereochemistry and integrity of the peptide backbone while enhancing metabolic stability through fluorination.

This patent represents a significant leap forward for manufacturers aiming to produce high-purity pharmaceutical intermediates with reduced environmental impact. The method avoids the cumbersome steps of introducing and removing protecting groups, which traditionally plague peptide synthesis. Instead, it employs a hypervalent iodine reagent, specifically 1-(trifluoromethyl)-1,2-benziodoxol-3(1H)-one, activated by common photocatalysts such as fluorescein or eosin Y. This approach not only simplifies the operational workflow but also drastically improves the overall atom economy of the synthesis. For R&D teams focused on developing novel peptide therapeutics, this technology offers a reliable route to access fluorinated analogs that may exhibit superior pharmacokinetic profiles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the trifluoromethylation of aromatic systems within complex molecules like peptides has been fraught with challenges regarding chemoselectivity and functional group tolerance. Traditional strategies often necessitate the use of N-protected anilines or similar precursors to attach the CF3 group, requiring additional synthetic operations that increase both cost and waste. The subsequent removal of these protecting groups typically demands strongly acidic or basic reaction conditions, which can be detrimental to sensitive peptide bonds and side-chain functionalities. Furthermore, the incompatibility of many important functional groups with these harsh conditions limits the scope of substrates that can be effectively modified. This lack of compatibility often leads to lower overall yields and the formation of difficult-to-remove impurities, complicating the purification process and reducing the viability of the route for commercial scale-up of complex peptide intermediates.
The Novel Approach
In stark contrast, the method disclosed in CN112062704B utilizes a direct C-H trifluoromethylation strategy driven by visible light irradiation. This novel approach bypasses the need for pre-functionalization or protection of the nitrogen atoms, allowing for a one-step conversion of the starting polypeptide into the desired trifluoromethylated product. The reaction proceeds smoothly at room temperature in a benign DMF and water mixture, showcasing exceptional compatibility with various amino acid residues. By employing a photocatalyst, the system generates reactive radical species under mild conditions, ensuring high site selectivity for the C2 or C3 position of the indole ring depending on the substrate structure. This streamlined process not only reduces the number of synthetic steps but also minimizes the generation of hazardous waste, making it an ideal candidate for cost reduction in API manufacturing where efficiency and sustainability are paramount.
Mechanistic Insights into Photoredox-Catalyzed C-H Activation
The core of this technology relies on a photoredox catalytic cycle that activates the hypervalent iodine reagent to generate a trifluoromethyl radical. Upon irradiation with blue LED light, the photocatalyst, such as tris(2,2'-bipyridyl)ruthenium dichloride or organic dyes like fluorescein, transitions to an excited state. This excited species then engages in a single-electron transfer (SET) process with the 1-(trifluoromethyl)-1,2-benziodoxol-3(1H)-one reagent. This interaction facilitates the homolytic cleavage of the carbon-iodine bond, releasing the highly reactive trifluoromethyl radical while regenerating the ground state of the catalyst. The generated radical subsequently attacks the electron-rich indole moiety of the tryptophan residue within the polypeptide chain, forming a new carbon-carbon bond. This mechanism is particularly advantageous because it operates under neutral conditions, avoiding the protonation or deprotonation events that could lead to racemization or peptide degradation.
Impurity control is inherently managed by the high selectivity of the radical addition process. Since the reaction targets specific electron-rich positions on the indole ring, side reactions at other parts of the peptide backbone are minimized. The use of water as a co-solvent further aids in suppressing non-specific radical pathways that might occur in purely organic media. Additionally, the mild reaction temperature prevents thermal decomposition of sensitive intermediates. Post-reaction workup involves standard extraction and silica gel chromatography, which effectively separates the target trifluoromethylated product from any unreacted starting material or minor byproducts. This robustness ensures that the final product meets stringent purity specifications required for pharmaceutical applications, providing a reliable supply chain for high-value intermediates.
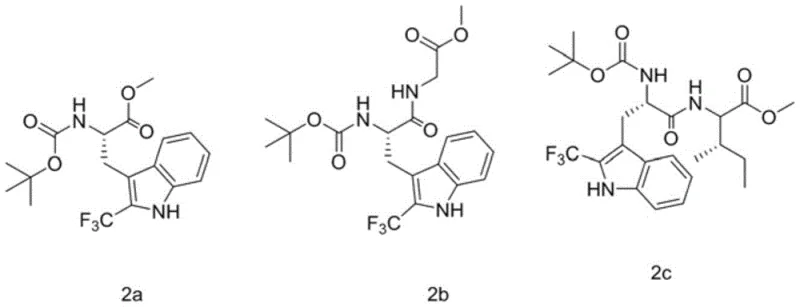
How to Synthesize Trifluoromethylated Polypeptides Efficiently
The synthesis protocol outlined in the patent is designed for operational simplicity and scalability, making it accessible for both laboratory research and industrial production. The process begins by dissolving the polypeptide substrate in a mixture of DMF and water, followed by the addition of the trifluoromethylating reagent and a catalytic amount of the photocatalyst. The reaction vessel is then placed under a blue LED light source at room temperature, where it is stirred for a period ranging from 3 to 8 hours depending on the specific substrate. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and optimal yield.
- Prepare a reaction mixture containing the polypeptide substrate, 1-(trifluoromethyl)-1,2-benziodoxol-3(1H)-one, and a photocatalyst like fluorescein in a DMF/water solvent system.
- Irradiate the mixture with a blue LED light source at room temperature for 3 to 8 hours to drive the radical trifluoromethylation reaction.
- Perform standard aqueous workup involving extraction with ethyl acetate and brine, followed by silica gel column chromatography to isolate the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this photocatalytic technology offers substantial strategic benefits beyond mere technical feasibility. The elimination of protection and deprotection steps translates directly into a simplified manufacturing workflow, which reduces the consumption of raw materials and solvents. This streamlining effect leads to significant cost savings by shortening the overall production timeline and minimizing the labor hours required for multi-step synthesis. Furthermore, the use of commercially available and inexpensive reagents like fluorescein and hypervalent iodine compounds ensures a stable and predictable supply chain, mitigating the risks associated with sourcing exotic or proprietary catalysts.
- Cost Reduction in Manufacturing: The direct C-H activation strategy removes the need for expensive protecting group chemistry, which traditionally accounts for a large portion of synthesis costs in peptide manufacturing. By consolidating multiple steps into a single transformation, the process drastically reduces solvent usage and waste disposal fees. The mild conditions also lower energy consumption compared to high-temperature or high-pressure alternatives, contributing to a leaner and more cost-effective production model that enhances overall profit margins.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as DMF, water, and blue LED lamps ensures that the supply chain is resilient against disruptions. Unlike processes dependent on scarce transition metals or specialized ligands, this method utilizes robust organic photocatalysts that are easy to source in bulk quantities. This accessibility guarantees consistent production schedules and reduces lead times for high-purity peptide derivatives, allowing manufacturers to respond quickly to market demands without being bottlenecked by material availability.
- Scalability and Environmental Compliance: The aqueous solvent system aligns perfectly with increasingly strict environmental regulations regarding volatile organic compound (VOC) emissions. The ability to run the reaction in water-rich mixtures simplifies wastewater treatment and reduces the environmental footprint of the facility. Moreover, the scalability of photochemical reactions has improved significantly with modern flow chemistry technologies, allowing this batch process to be easily adapted for continuous manufacturing, thereby supporting the commercial scale-up of complex polymer additives or pharmaceutical intermediates with minimal risk.
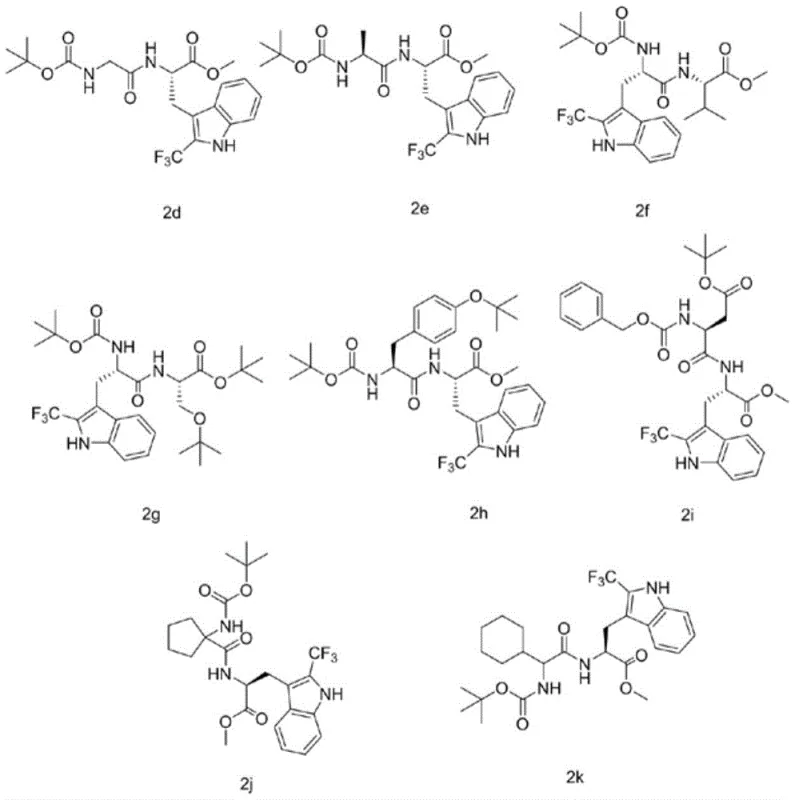
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this trifluoromethylation technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on yield expectations, substrate scope, and purification methods. Understanding these details is crucial for evaluating the feasibility of integrating this method into existing production pipelines.
Q: What are the advantages of this photocatalytic method over traditional trifluoromethylation?
A: Unlike traditional methods requiring N-protection and harsh acidic or basic conditions for deprotection, this patent describes a direct C-H activation route. It operates under mild room temperature conditions using visible light, significantly improving functional group compatibility and atom economy.
Q: Is this process suitable for large-scale manufacturing of peptide intermediates?
A: Yes, the process utilizes commercially available raw materials and a simple DMF/water solvent system, which aligns with green chemistry principles. The mild conditions and lack of complex protection/deprotection steps make it highly scalable for industrial production.
Q: What kind of yields can be expected for these trifluoromethylated derivatives?
A: The patent data demonstrates robust performance across various substrates. For instance, specific examples like compound 2a achieved yields of 89%, while others such as 2h reached 82%, indicating high efficiency and reproducibility for diverse peptide sequences.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trifluoromethylated Polypeptide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of photocatalytic C-H activation in the synthesis of advanced peptide therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN112062704B can be successfully translated into robust industrial processes. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of trifluoromethylated intermediates meets the highest quality standards required by global regulatory bodies.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific molecule. We are ready to provide specific COA data and route feasibility assessments to help you accelerate your development timeline and secure a competitive advantage in the marketplace.
