Advanced Chemical Synthesis and Purification Strategies for High-Purity β-Nicotinamide Mononucleotide
Advanced Chemical Synthesis and Purification Strategies for High-Purity β-Nicotinamide Mononucleotide
The rapidly expanding market for anti-aging therapeutics has placed β-nicotinamide mononucleotide (NMN) at the forefront of biochemical research, driven by its critical role as a precursor to nicotinamide adenine dinucleotide (NAD+). As detailed in patent CN111253448A, recent technological breakthroughs have addressed the longstanding challenges of low yield and high production costs associated with traditional synthesis methods. This patent discloses a sophisticated preparation and purification method that fundamentally optimizes the chemical pathway by introducing a critical purification step for the furanose substrate intermediate prior to phosphorylation. By strategically isolating the target intermediate from complex reaction mixtures using specific phase extraction techniques, manufacturers can achieve significantly higher purity levels and improved overall process efficiency. This technical advancement represents a pivotal shift for reliable pharmaceutical intermediate suppliers aiming to meet the rigorous quality standards demanded by the global nutraceutical and pharmaceutical industries.
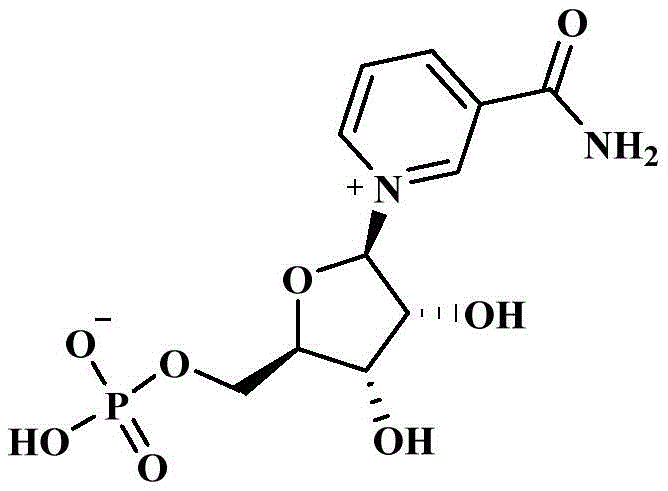
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of β-nicotinamide mononucleotide has been hindered by significant technical bottlenecks inherent in both biological and early chemical synthesis routes. In biological synthesis, the reliance on nicotinamide phosphoribosyl transferase (NAMPT) introduces severe supply chain vulnerabilities due to the enzyme's high cost and limited availability, often resulting in prohibitively low yields that restrict commercial viability. Conversely, traditional chemical synthesis pathways, while avoiding enzymatic constraints, typically suffer from multi-step functional group conversions that generate a plethora of byproducts and impurities. These impurities, including unreacted tetraacetyl ribose and various ester derivatives, complicate the final purification process, often necessitating complex resin column elution procedures that degrade overall recovery rates. Furthermore, the lack of effective intermediate purification means that impurities carry through to the phosphorylation stage, competing for reagents and drastically reducing the selectivity of the final reaction, which ultimately inflates the cost of goods sold for high-purity NMN.
The Novel Approach
The innovative methodology presented in the patent data overcomes these deficiencies by decoupling the purification of the furanose substrate from the final phosphorylation reaction, thereby creating a cleaner reaction environment for the critical bond-forming step. Instead of subjecting a crude mixture directly to phosphorylation conditions, this novel approach employs a dual-extraction strategy that leverages the differential solubility properties of the target intermediate versus its contaminants. By first utilizing a water-oil phase extraction to remove polar impurities like ammonia and methanol, and subsequently employing a phosphorylation auxiliary agent phase to selectively extract the target furanose compound, the process ensures that only high-purity substrate enters the final reaction vessel. This strategic intervention not only simplifies the downstream processing requirements but also dramatically enhances the reaction kinetics during phosphorylation, as the phosphorus oxychloride reagent is no longer consumed by side reactions with extraneous nucleophiles. The result is a streamlined, robust process capable of delivering superior product quality with reduced operational complexity.
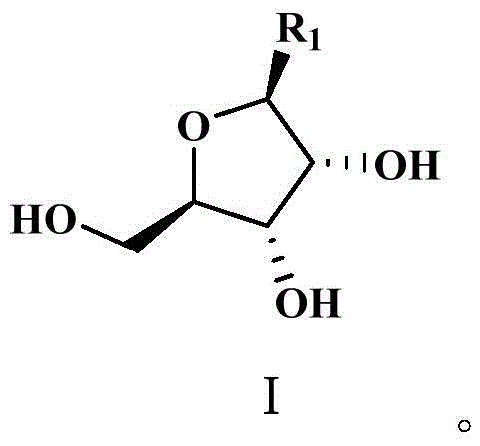
Mechanistic Insights into Phase-Transfer Purification and Phosphorylation
The core of this technological advancement lies in the precise manipulation of solubility parameters to achieve high-fidelity separation of the furanose intermediate, represented structurally in Formula I. The mechanism begins with a liquid-liquid extraction where the crude intermediate mixture is partitioned between an aqueous phase and an organic oil phase, such as dichloromethane or ethyl acetate. In this equilibrium, the target compound, which possesses specific lipophilic characteristics due to its nicotinamide or ester substituents, preferentially migrates into the organic phase, while highly polar impurities and residual catalysts remain in the aqueous layer. This initial separation is critical for removing bulk contaminants that could otherwise interfere with subsequent reaction steps. Following this, the organic phase containing the enriched intermediate is subjected to a second extraction using a phosphorylation auxiliary agent, typically a phosphate ester like triethyl phosphate. This step exploits the specific affinity of the furanose hydroxyl groups for the phosphate medium, effectively pulling the target molecule into a phase that is chemically compatible with the upcoming phosphorylation reagents, thus creating a seamless transition between purification and synthesis.
Furthermore, the control of impurity profiles is meticulously managed through the selection of extraction solvents and the optimization of mixing times, which are specified in the range of 10 to 30 minutes to ensure thermodynamic equilibrium without degrading the sensitive glycosidic bonds. By repeating the extraction with the phosphorylation auxiliary agent two to four times, the process maximizes the recovery of the valuable intermediate while leaving behind non-polar organic impurities that do not share the same affinity for the phosphate phase. This high degree of selectivity ensures that when the purified intermediate is finally reacted with phosphorus oxychloride at low temperatures ranging from -20°C to -5°C, the reaction proceeds with exceptional specificity. The absence of competing nucleophiles allows for the efficient formation of the phosphate ester bond at the 5'-position of the ribose ring, minimizing the formation of pyrophosphate byproducts or cyclic phosphate impurities that are common in less controlled environments.
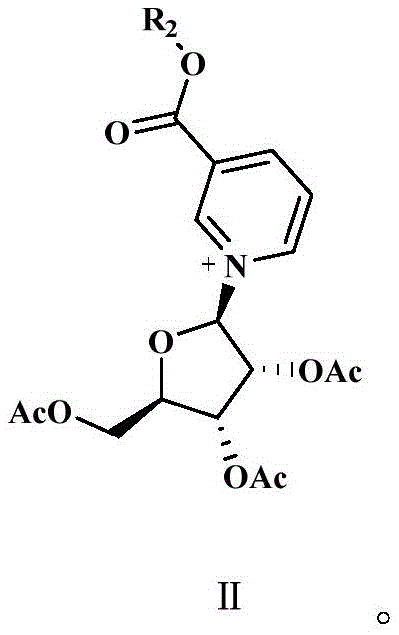
How to Synthesize β-Nicotinamide Mononucleotide Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for transitioning from raw nicotinic acid esters and tetraacetyl ribose to the final high-purity nucleotide. The process begins with the condensation of the nicotinic acid derivative with the sugar moiety, followed by a crucial deprotection or ammonolysis step to generate the crude furanose intermediate. However, the defining feature of this method is the immediate intervention of the purification sequence described previously, which acts as a gatekeeper before the final phosphorylation. Operators must strictly adhere to the temperature controls during the phosphorylation step, maintaining the reaction mixture between -15°C and -10°C to prevent thermal degradation of the labile nucleotide structure. Detailed standardized synthetic steps for implementing this purification-enhanced route are provided below to ensure reproducibility and safety in a GMP-compliant environment.
- Extract the raw furanose substrate intermediate using a water-oil phase system to isolate the target compound into the oil phase.
- Perform a secondary extraction on the oil phase using a phosphorylation auxiliary agent (e.g., triethyl phosphate) to further purify the intermediate.
- React the purified intermediate with phosphorus oxychloride at low temperatures (-10°C to -15°C) to achieve final phosphorylation and form β-nicotinamide mononucleotide.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this refined synthesis pathway offers substantial strategic benefits that extend beyond mere technical performance metrics. The elimination of expensive enzymatic catalysts removes a significant variable cost driver and supply risk, replacing it with commodity chemicals that are readily available in the global market. Moreover, the ability to recycle solvents such as dichloromethane and ethyl acetate, as well as the phosphorylation auxiliary agents, contributes to a drastic reduction in raw material consumption and waste disposal costs. This closed-loop solvent management system not only aligns with increasingly stringent environmental regulations but also insulates the manufacturing process from volatility in solvent pricing, ensuring more predictable long-term production economics for buyers seeking cost reduction in pharmaceutical intermediate manufacturing.
- Cost Reduction in Manufacturing: The process achieves significant economic efficiency by eliminating the need for costly chromatographic purification columns typically required to separate NMN from closely related impurities in crude reaction mixes. By shifting the purification burden to the liquid-liquid extraction phase, which utilizes inexpensive solvents and standard separation equipment, the capital expenditure and operational overhead associated with downstream processing are substantially lowered. Additionally, the increased selectivity of the phosphorylation reaction means that less phosphorus oxychloride is wasted on side reactions, further optimizing the stoichiometric efficiency of the most expensive reagent in the sequence.
- Enhanced Supply Chain Reliability: Unlike enzymatic routes that depend on the fermentation and purification of specific transferase enzymes, this chemical synthesis method relies entirely on stable, shelf-stable small molecule reagents. This fundamental shift ensures that production schedules are not vulnerable to biological batch failures or enzyme supply shortages, providing a consistent and reliable output of high-purity NMN. The robustness of the extraction protocol also allows for flexible scaling, meaning that suppliers can rapidly adjust production volumes to meet surging market demand without the lead time constraints associated with bioreactor capacity expansion.
- Scalability and Environmental Compliance: The simplicity of the unit operations involved—primarily mixing, settling, and phase separation—makes this process inherently scalable from pilot plant to multi-ton commercial production. The use of recyclable solvents and the avoidance of heavy metal catalysts simplify the waste stream treatment, reducing the environmental footprint of the manufacturing facility. This compliance-friendly profile facilitates smoother regulatory approvals and reduces the liability associated with hazardous waste disposal, making it an attractive option for sustainable chemical manufacturing initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel purification technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for evaluating the feasibility of this route for your specific application needs. Understanding these nuances is essential for R&D teams planning to integrate this intermediate into their own formulation or synthesis workflows.
Q: How does the pre-phosphorylation purification step impact the final yield of NMN?
A: By removing impurities such as unreacted raw materials and side products before the critical phosphorylation step, the process prevents competitive side reactions. Patent data indicates this method increases total yield from approximately 61% in conventional methods to over 73%.
Q: What specific solvents are utilized in the purification of the furanose intermediate?
A: The process utilizes a biphasic extraction system involving water and organic solvents such as dichloromethane, ethyl acetate, or toluene, followed by extraction with phosphorylation auxiliaries like trimethyl or triethyl phosphate.
Q: Is this chemical synthesis method scalable for industrial production compared to enzymatic routes?
A: Yes, unlike enzymatic synthesis which relies on expensive and limited nicotinamide phosphoribosyl transferase, this chemical route uses readily available raw materials and standard extraction equipment, making it highly suitable for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable β-Nicotinamide Mononucleotide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of purity and consistency in the production of bioactive nucleotides like β-nicotinamide mononucleotide. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated purification protocols described in patent CN111253448A can be seamlessly translated into industrial reality. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced HPLC and NMR capabilities to verify that every batch meets the highest international standards for pharmaceutical and nutraceutical applications. Our commitment to technical excellence ensures that our clients receive a product that is not only chemically pure but also free from the residual solvents and impurities that plague lower-grade alternatives.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis route can be tailored to your specific volume and quality requirements. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic advantages of switching to this purification-enhanced supply chain. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that will strengthen your supply chain resilience and product quality in the competitive anti-aging market.
