Advanced Synthesis of 5-Fluoro-2-Nitrophenol: Technical Upgrades for Commercial Scale-Up
The global demand for high-performance agrochemicals continues to drive innovation in the synthesis of key intermediates, particularly those used in herbicide production. Patent CN107935858B introduces a transformative preparation method for 5-fluoro-2-nitrophenol, a vital precursor for the synthesis of flumioxazin and other fine chemicals. This technology addresses critical bottlenecks in existing manufacturing processes, specifically targeting the issues of low conversion rates, excessive byproduct formation, and prolonged reaction times that have historically plagued industrial operations. By shifting from traditional hydrolysis pathways to a selective ammonolysis strategy followed by diazotization, this method offers a robust solution for producing high-purity intermediates. For R&D directors and procurement managers alike, understanding the nuances of this patent is essential for optimizing supply chains and reducing the total cost of ownership in agrochemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 5-fluoro-2-nitrophenol has relied on two primary routes, both of which present significant drawbacks for large-scale industrial application. The first method involves the direct nitration of m-fluorophenol; however, this approach is economically unviable due to the high cost of the starting material and the generation of numerous isomeric byproducts during the nitration step, leading to poor yields and difficult purification processes. The second, more common method involves the direct hydrolysis of 2,4-difluoronitrobenzene using strong inorganic bases such as sodium hydroxide or potassium hydroxide. While operationally simple, this alkaline hydrolysis route suffers from inherent selectivity issues, inevitably producing approximately 15% of unwanted byproducts. This side reaction not only caps the maximum theoretical yield but also complicates downstream processing, requiring extensive resources for waste treatment and product isolation, thereby inflating production costs and extending lead times.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in the patent utilizes a two-step sequence that fundamentally alters the reaction pathway to maximize efficiency. The process begins with the selective nucleophilic substitution of 2,4-difluoronitrobenzene using ammonia, converting it into 5-fluoro-2-nitroaniline with exceptional specificity. This intermediate is then subjected to a diazotization and hydrolysis sequence using sulfuric acid and sodium nitrite to yield the final phenol product. This strategic detour through the aniline intermediate bypasses the competitive side reactions observed in direct hydrolysis, resulting in almost no side products and significantly shortened reaction cycles. The ability to crystallize the intermediate further enhances purity before the final step, ensuring that the final 5-fluoro-2-nitrophenol meets stringent quality specifications required for sensitive agrochemical applications.
Mechanistic Insights into Selective Ammonolysis and Diazotization
The core of this technological breakthrough lies in the precise control of nucleophilic aromatic substitution and subsequent diazonium chemistry. In the first step, ammonia acts as a nucleophile attacking the electron-deficient aromatic ring of 2,4-difluoronitrobenzene. The presence of the strongly electron-withdrawing nitro group activates the ring towards nucleophilic attack, particularly at positions ortho and para to itself. By carefully controlling the molar ratio of ammonia to substrate (optimized between 2:1 and 4:1) and maintaining a moderate temperature range of 35-40°C, the reaction achieves high regioselectivity, preferentially displacing one fluorine atom to form 5-fluoro-2-nitroaniline while leaving the other fluorine intact. This selectivity is crucial, as it prevents the formation of difluoro- or diamino- byproducts that would otherwise contaminate the stream.
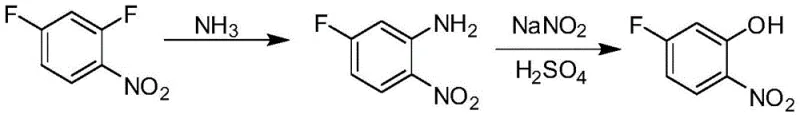
Following the isolation of the aniline intermediate via cooling crystallization, the second stage employs a classic diazotization mechanism. The amine group is converted into a diazonium salt using sodium nitrite in an acidic medium at low temperatures (0-10°C), a step that requires precise thermal management to prevent premature decomposition. Subsequent heating of the diazonium solution to 90-95°C facilitates the hydrolysis of the diazo group, replacing it with a hydroxyl group to form the phenol. This mechanistic pathway is inherently cleaner than direct nucleophilic substitution of fluorine by hydroxide because the diazonium group is an excellent leaving group, driving the reaction to completion with minimal resistance. The result is a process characterized by good selectivity and high yield, effectively solving the technical problems of low conversion and long reaction times associated with previous methods.
How to Synthesize 5-Fluoro-2-Nitrophenol Efficiently
Implementing this synthesis route requires careful attention to stoichiometry and thermal profiles to replicate the high yields reported in the patent data. The process is designed to be operationally simple, utilizing common inorganic reagents that are readily available in the global chemical market. The initial ammonolysis step is exothermic and must be managed to maintain the optimal 35-40°C window, ensuring complete conversion without degrading the sensitive nitro-fluoro substrate. Following the reaction, the mixture is cooled to induce crystallization of the 5-fluoro-2-nitroaniline, which serves as an in-process purification step. The subsequent diazotization requires strict temperature control during the addition of sodium nitrite to ensure stable diazonium salt formation before the final hydrolysis heating ramp. Detailed standardized synthetic steps for this high-efficiency route are provided in the guide below.
- React 2,4-difluoronitrobenzene with aqueous ammonia (molar ratio 2-4: 1) at 35-40°C to selectively produce 5-fluoro-2-nitroaniline.
- Cool the reaction mixture to 5-10°C to crystallize and isolate the 5-fluoro-2-nitroaniline intermediate with high purity.
- Dissolve the aniline derivative in sulfuric acid, diazotize with sodium nitrite at 0-10°C, and hydrolyze by heating to 90-95°C to obtain the final phenol product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers substantial strategic benefits beyond mere technical superiority. The shift away from expensive starting materials like m-fluorophenol and the elimination of the 15% byproduct loss seen in hydrolysis methods directly translates to improved material efficiency. By utilizing low-cost inorganic reagents such as ammonia and sodium nitrite in excess, the process ensures that the more expensive organic substrate is fully utilized, maximizing the return on raw material investment. This efficiency gain is critical in a volatile market where raw material price fluctuations can severely impact margins, making a high-yield, waste-minimized process a valuable asset for cost containment strategies.
- Cost Reduction in Manufacturing: The economic implications of this process are profound, primarily driven by the drastic improvement in yield and the simplification of the purification workflow. By avoiding the generation of significant byproduct loads, the need for complex separation techniques and extensive solvent usage is significantly reduced, leading to lower utility and waste disposal costs. Furthermore, the use of inexpensive inorganic reagents to drive the reaction eliminates the reliance on costly catalysts or specialized organic bases, streamlining the bill of materials. This structural cost advantage allows manufacturers to offer more competitive pricing for high-purity agrochemical intermediates while maintaining healthy profit margins.
- Enhanced Supply Chain Reliability: From a supply chain perspective, the robustness of this synthesis route ensures greater continuity of supply. The reaction conditions are mild and do not require extreme pressures or temperatures that often lead to equipment downtime or safety incidents in less optimized processes. The ability to crystallize the intermediate provides a buffer stock of high-purity material that can be stored and processed as needed, decoupling the two reaction steps and allowing for flexible production scheduling. This flexibility is essential for meeting the fluctuating demands of the agrochemical sector, reducing lead times for high-purity intermediates and ensuring that downstream herbicide production lines remain operational without interruption.
- Scalability and Environmental Compliance: The environmental footprint of this process is markedly lower than conventional methods, aligning with increasingly stringent global regulations on chemical manufacturing. The reduction in byproduct formation means less hazardous waste is generated per kilogram of product, simplifying compliance with environmental discharge standards. Additionally, the simplicity of the operation—using water-based systems and common acids—makes the process highly scalable from pilot plants to multi-ton commercial reactors. This ease of scale-up reduces the risk associated with technology transfer, allowing suppliers to rapidly increase capacity to meet market surges without compromising on quality or safety protocols.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 5-fluoro-2-nitrophenol based on the insights from patent CN107935858B. These answers are derived directly from the experimental data and beneficial effects described in the intellectual property documentation, providing clarity on the feasibility and advantages of this specific synthetic route for potential partners and stakeholders.
Q: What are the primary advantages of the ammonolysis route over direct hydrolysis for 5-fluoro-2-nitrophenol?
A: The ammonolysis route described in patent CN107935858B avoids the formation of approximately 15% byproducts common in direct alkaline hydrolysis methods. By converting the difluoro precursor to an aniline intermediate first, the process achieves superior regioselectivity and higher overall yields, significantly reducing raw material waste.
Q: How does the new synthesis method impact industrial scalability?
A: The method utilizes inexpensive inorganic reagents like ammonia and sodium nitrite in excess to drive reactions to completion, ensuring robust conversion rates. The operational conditions (35-40°C for ammonolysis and standard diazotization temperatures) are mild and easily manageable in large-scale reactors, facilitating smooth commercial scale-up without complex pressure or temperature controls.
Q: Is this intermediate suitable for flumioxazin production?
A: Yes, 5-fluoro-2-nitrophenol is a critical building block for the synthesis of flumioxazin, a widely used herbicide. The high purity achieved through the crystallization steps in this patent ensures the quality required for downstream agrochemical synthesis, minimizing impurities that could affect the efficacy of the final herbicide.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Fluoro-2-Nitrophenol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient synthesis routes play in the competitiveness of the agrochemical industry. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative technologies like the one described in CN107935858B are translated into reliable supply solutions. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 5-fluoro-2-nitrophenol meets the exacting standards required for herbicide synthesis. We are committed to leveraging our technical expertise to optimize these processes further, delivering consistent quality and performance to our global clientele.
We invite you to collaborate with us to explore how this advanced manufacturing technology can benefit your specific supply chain requirements. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume needs, demonstrating the tangible economic benefits of switching to this high-yield route. Please contact us today to request specific COA data and route feasibility assessments, and let us help you secure a stable, cost-effective source of this essential agrochemical intermediate.
