Scalable Nickel-Catalyzed Asymmetric Hydroalkynylation for High-Purity AMG 837 Intermediates
Scalable Nickel-Catalyzed Asymmetric Hydroalkynylation for High-Purity AMG 837 Intermediates
The pharmaceutical industry is constantly seeking more efficient and cost-effective pathways to synthesize complex chiral intermediates, particularly for metabolic disease treatments like type 2 diabetes. Patent CN112679290B, published on March 15, 2022, introduces a groundbreaking nickel-catalyzed asymmetric hydroalkynylation method that addresses critical bottlenecks in producing benzylic alkynyl-substituted compounds. This technology leverages abundant and inexpensive nickel salts instead of precious metals like palladium or rhodium, operating under remarkably mild conditions to achieve exceptional regioselectivity and enantioselectivity. For R&D directors and procurement managers, this represents a significant opportunity to optimize the supply chain for key drug candidates such as AMG 837, ensuring both high purity and reduced manufacturing costs through a robust and scalable catalytic process.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of chiral alkynyl compounds, which are vital building blocks for bioactive molecules like the antineoplastic agent (-)-chamaecynone and the anti-HIV drug Efavirenz, has relied on multi-step sequences involving expensive transition metals. Conventional routes often utilize palladium or rhodium catalysts, which not only drive up the raw material costs significantly but also pose challenges regarding residual metal removal in the final active pharmaceutical ingredient (API). Furthermore, many existing methods require harsh reaction conditions, such as elevated temperatures or strong bases, which can lead to poor functional group tolerance and the formation of difficult-to-remove impurities. These limitations result in lower overall yields and extended production timelines, creating substantial friction for supply chain heads who need reliable, high-volume access to these critical intermediates without compromising on quality specifications.
The Novel Approach
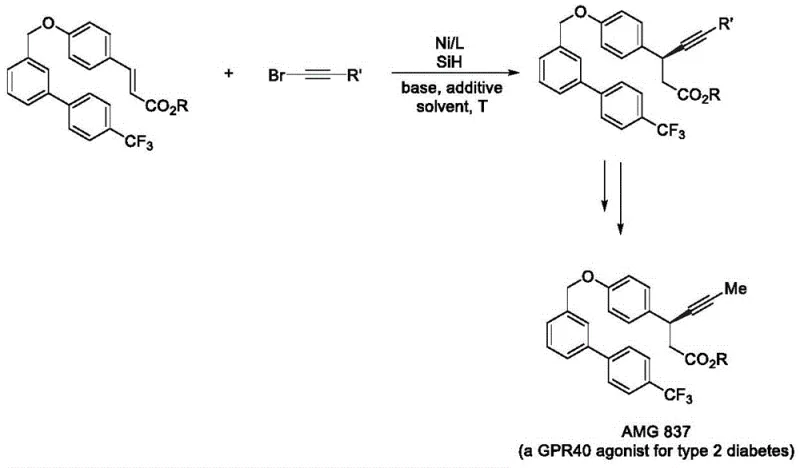
The novel approach disclosed in the patent utilizes a nickel-hydrogen catalytic strategy to couple readily available olefins with alkynyl bromides in a single step. This method employs cheap transition metal nickel salts, such as nickel iodide hydrate, in conjunction with specialized chiral ligands like the PyrOx series (specifically L*47). The reaction proceeds smoothly at 0°C in organic solvents like trifluorotoluene, utilizing silanes as a hydrogen source. This shift to nickel catalysis drastically simplifies the synthetic route, eliminating the need for pre-functionalized organometallic reagents often required in cross-coupling reactions. By achieving excellent enantioselectivity directly from simple olefins, this process reduces the number of purification steps and minimizes waste generation, offering a streamlined pathway that is highly attractive for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Nickel-Catalyzed Asymmetric Hydroalkynylation
The core of this technological advancement lies in the generation of an active nickel-hydride (Ni-H) species in situ, which serves as the key mediator for the asymmetric transformation. The mechanism involves the coordination of the chiral ligand L* to the metallic nickel center, creating a chiral environment that dictates the stereochemical outcome of the reaction. Upon activation by the hydrosilane hydrogen source, the nickel catalyst inserts into the olefin double bond with high regioselectivity, forming a benzylic nickel intermediate. This intermediate then undergoes transmetallation or oxidative addition with the alkynyl bromide compound, followed by reductive elimination to release the desired chiral benzyl alkynyl product. The use of additives like sodium iodide and inorganic bases such as potassium phosphate monohydrate further stabilizes the catalytic cycle and enhances the turnover number, ensuring consistent performance across diverse substrate scopes.
Impurity control is inherently managed through the high selectivity of the chiral ligand system. The patent data indicates that the L*47 ligand is superior in controlling chirality compared to other variants, effectively suppressing the formation of undesired enantiomers and regioisomers. The mild reaction temperature of 0°C plays a crucial role in kinetic control, preventing thermal degradation of sensitive functional groups and minimizing side reactions such as olefin isomerization or polymerization. This precise control over the reaction trajectory ensures that the resulting crude product has a clean impurity profile, significantly reducing the burden on downstream purification processes. For quality assurance teams, this means a more predictable and stable manufacturing process capable of consistently meeting stringent purity specifications required for clinical-grade materials.
How to Synthesize Chiral Benzyl Alkynyl Compounds Efficiently
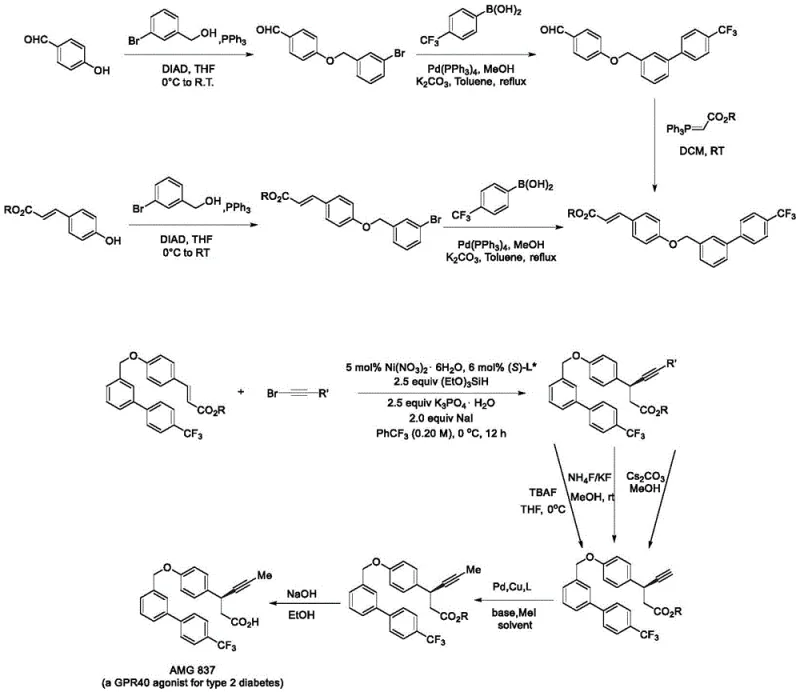
The synthesis protocol outlined in the patent provides a reproducible framework for generating high-value chiral intermediates. The process begins with the careful preparation of the catalytic mixture under an inert atmosphere to prevent oxidation of the nickel species. Key reagents including the nickel salt, chiral ligand, base, and additive are dissolved in a dried organic solvent before the introduction of the hydride source and substrates. Maintaining strict temperature control at 0°C throughout the 12-hour reaction period is essential for maximizing enantiomeric excess. Following the reaction, standard workup procedures involving solvent removal and column chromatography yield the target compound with high optical purity. Detailed standardized synthesis steps for implementing this protocol in a GMP environment are provided below.
- Prepare the catalytic system by dissolving nickel iodide hydrate, chiral PyrOx ligand L*47, potassium phosphate, and sodium iodide in dry trifluorotoluene under inert atmosphere.
- Add trimethoxysilane as the hydrogen source, followed by the olefin substrate and alkynyl bromide compound, maintaining the reaction temperature at 0°C for 12 hours.
- Upon completion, remove the solvent under reduced pressure and purify the target chiral product via silica gel column chromatography to achieve high enantiomeric excess.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this nickel-catalyzed methodology offers transformative economic and operational benefits. The primary advantage stems from the substitution of precious metal catalysts with abundant nickel, which drastically reduces the raw material cost basis for the catalytic system. Unlike palladium or rhodium, which are subject to volatile market pricing and supply constraints, nickel is widely available and inexpensive, leading to substantial cost savings in API manufacturing. Additionally, the simplified one-step nature of the reaction reduces the consumption of solvents and reagents associated with multi-step sequences, further lowering the overall cost of goods sold (COGS) and minimizing the environmental footprint of the production facility.
- Cost Reduction in Manufacturing: The elimination of expensive precious metal catalysts and the reduction in synthetic steps directly translate to lower production costs. By avoiding the need for complex pre-functionalization of starting materials and utilizing commercially available olefins and alkynyl bromides, the process streamlines the supply chain. This efficiency gain allows for significant margin improvement without compromising the quality of the final pharmaceutical intermediate, making it a highly competitive option for large-scale production.
- Enhanced Supply Chain Reliability: The reliance on commercially available and stable starting materials ensures a robust supply chain that is less susceptible to disruptions. The mild reaction conditions and high functional group tolerance mean that the process is adaptable to various substrate derivatives, providing flexibility in sourcing raw materials. This reliability is critical for maintaining continuous production schedules and meeting the demanding delivery timelines of global pharmaceutical clients.
- Scalability and Environmental Compliance: The process is designed for scalability, with conditions that are easily transferable from laboratory to pilot and commercial scales. The use of milder reagents and the generation of less hazardous waste align with green chemistry principles, simplifying regulatory compliance and waste disposal management. This environmental compatibility not only reduces operational risks but also enhances the sustainability profile of the manufacturing operation, which is increasingly important for corporate social responsibility goals.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this nickel-catalyzed technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on reaction parameters and substrate compatibility. Understanding these details is essential for technical teams evaluating the feasibility of integrating this method into their existing manufacturing workflows.
Q: What is the optimal reaction temperature for this nickel-catalyzed hydroalkynylation?
A: According to patent CN112679290B, the reaction effect is optimal at 0°C. This mild temperature allows for excellent yield and enantioselectivity while minimizing side reactions often seen at higher temperatures.
Q: Which chiral ligand provides the best enantioselectivity in this process?
A: The patent identifies the L*47 PyrOx ligand as the dominant ligand. When combined with the metal catalyst, it effectively controls chirality, providing products with high yield and high enantioselectivity compared to other ligands like Box or Biox.
Q: Can this method be applied to the synthesis of AMG 837?
A: Yes, the patent explicitly details the application of this nickel-catalyzed asymmetric hydroalkynylation method in the preparation of AMG 837, a GPR40 agonist for type 2 diabetes, demonstrating simple steps and good functional group compatibility.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable AMG 837 Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced catalytic technologies like the nickel-catalyzed asymmetric hydroalkynylation described in CN112679290B. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including high enantiomeric excess, guaranteeing that every batch of AMG 837 intermediate meets the highest quality standards required for clinical and commercial applications.
We invite pharmaceutical companies and research institutions to collaborate with us to leverage this cost-effective synthesis route. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to accelerate your development timelines and secure a reliable supply of high-purity pharmaceutical intermediates for your next-generation therapies.
