Advanced Synthesis of 3-Ethyl-4-Fluorobenzonitrile for High-Purity API Manufacturing
Advanced Synthesis of 3-Ethyl-4-Fluorobenzonitrile for High-Purity API Manufacturing
In the rapidly evolving landscape of oncology drug development, the efficient synthesis of key intermediates remains a critical bottleneck for pharmaceutical manufacturers. Patent CN108623496B introduces a groundbreaking preparation method for 3-ethyl-4-fluorobenzonitrile, a pivotal building block for the investigational gastrointestinal stromal tumor (GIST) therapeutic, TAS-116. This technical insight report analyzes the proprietary methodology disclosed in the patent, highlighting its potential to revolutionize supply chain stability and cost structures for global API producers. By shifting away from hazardous organotin reagents toward safer transition-metal catalyzed cross-coupling strategies, this innovation addresses long-standing safety and purification challenges inherent in previous synthetic routes.
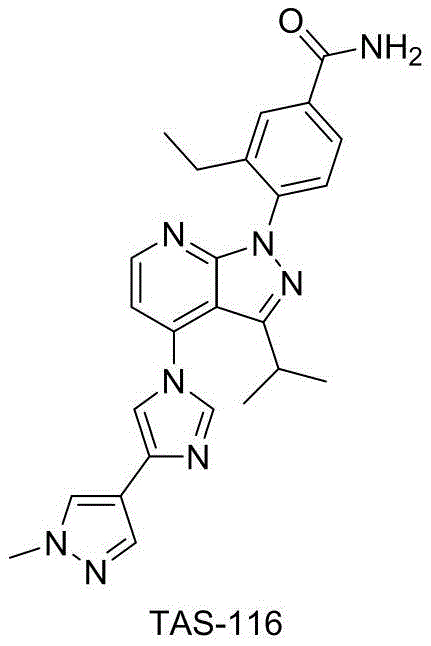
The strategic importance of this intermediate cannot be overstated, as it serves as the foundational scaffold for next-generation kinase inhibitors currently in clinical development. For R&D directors and procurement specialists, understanding the nuances of this new synthetic pathway is essential for securing a reliable pharmaceutical intermediate supplier capable of meeting stringent regulatory and quality demands. The following analysis dissects the chemical engineering advantages, mechanistic robustness, and commercial viability of this novel process, providing a comprehensive roadmap for its integration into commercial manufacturing workflows.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies, specifically those disclosed in Patent WO2005105760, have historically relied on the Stille coupling reaction using tetrakis(triphenylphosphine)palladium as a catalyst and tetraethyltin as the ethyl source. While chemically feasible, this approach presents catastrophic drawbacks for modern industrial application, primarily centered around the extreme toxicity of organotin compounds. The handling of tetraethyltin requires specialized containment infrastructure and rigorous waste management protocols to prevent operator exposure and environmental contamination, significantly inflating operational expenditures. Furthermore, the reaction typically yields the target 3-ethyl-4-fluorobenzonitrile as an oil, necessitating complex and labor-intensive purification via column chromatography to achieve acceptable purity levels.
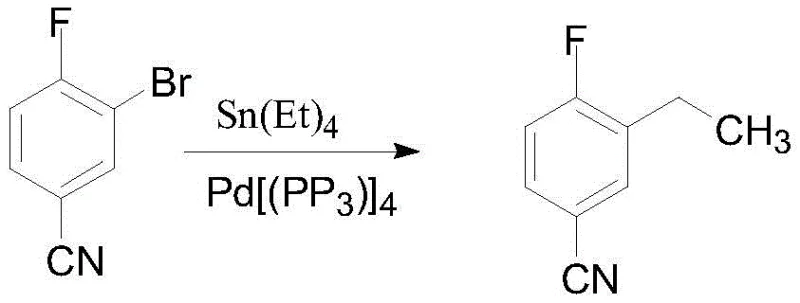
This reliance on chromatographic purification creates a severe bottleneck for scale-up, as column chromatography is notoriously difficult to translate from laboratory benchtop to multi-ton reactor scales without sacrificing throughput or yield. The oily nature of the product also complicates downstream processing, storage, and transportation, increasing the risk of degradation and handling errors. Consequently, the conventional route fails to meet the rigorous safety, environmental, and economic criteria required for the commercial manufacture of high-value pharmaceutical intermediates, driving the urgent need for a safer, more robust alternative that eliminates toxic reagents and simplifies isolation.
The Novel Approach
The invention detailed in CN108623496B offers a transformative solution by replacing the hazardous tin-based coupling with a versatile palladium or nickel-catalyzed cross-coupling reaction using safer ethylating agents such as diethyl zinc, ethyl magnesium bromide, or ethyl boronic acid. This strategic shift not only mitigates the severe toxicity risks associated with organotin chemistry but also fundamentally alters the physical properties of the final product, yielding 3-ethyl-4-fluorobenzonitrile as a stable off-white solid rather than an oil. The ability to isolate the product as a solid enables the use of simple, scalable crystallization techniques using common solvents like n-heptane or n-hexane, completely bypassing the need for inefficient column chromatography.
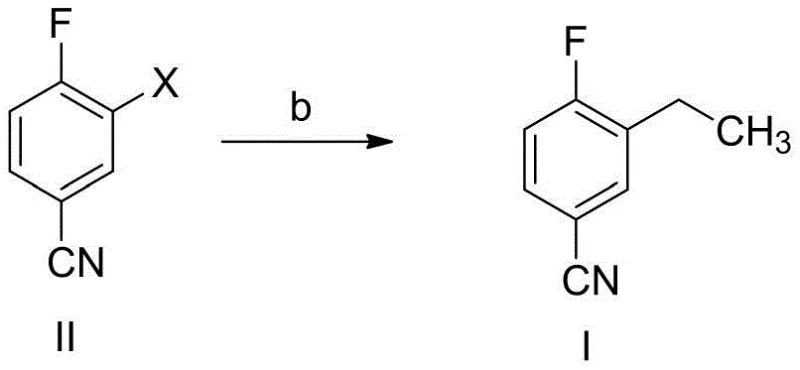
Moreover, the new method utilizes readily available and cost-effective starting materials, such as 3-bromo-4-fluorobenzaldehyde, which can be efficiently converted to the necessary nitrile precursor through mild oximation or iodination protocols. The reaction conditions are remarkably flexible, operating effectively across a broad temperature range of 0-100°C and accommodating various solvent systems including THF, toluene, and acetonitrile. This adaptability ensures that the process can be easily optimized for different reactor configurations and scale requirements, providing a robust platform for cost reduction in API manufacturing while maintaining exceptional product quality and consistency.
Mechanistic Insights into Palladium-Catalyzed Ethylation
The core of this innovative synthesis lies in the transition-metal catalyzed cross-coupling mechanism, likely proceeding through a Negishi-type or Kumada-type pathway depending on the specific ethylating agent employed. When utilizing diethyl zinc, the catalytic cycle initiates with the oxidative addition of the aryl halide (Compound II) to the low-valent palladium(0) species, forming an aryl-palladium(II) intermediate. Subsequent transmetallation with the diethyl zinc reagent transfers the ethyl group to the metal center, followed by reductive elimination to forge the new carbon-carbon bond, regenerating the active catalyst and releasing the desired 3-ethyl-4-fluorobenzonitrile. This mechanism is highly efficient and tolerant of various functional groups, ensuring high conversion rates even at low catalyst loadings of 0.0001 to 0.5 molar equivalents.
Impurity control is meticulously managed through the selection of specific crystallization solvents and temperatures, which exploit the differential solubility of the target molecule versus side products such as homocoupling byproducts or unreacted starting materials. The patent specifies crystallization temperatures ranging from -120°C to 200°C, with a preferred window of -80°C to -20°C, allowing for the precise exclusion of trace impurities that could compromise the biological activity of the final drug substance. By avoiding the use of silica gel or other adsorbents used in chromatography, the process minimizes the risk of introducing particulate matter or metal leaching, thereby enhancing the overall purity profile and ensuring compliance with strict residual solvent and heavy metal guidelines mandated by global health authorities.
How to Synthesize 3-Ethyl-4-Fluorobenzonitrile Efficiently
The synthesis protocol outlined in the patent provides a clear, step-by-step framework for producing high-purity 3-ethyl-4-fluorobenzonitrile suitable for GMP manufacturing. The process begins with the preparation of the nitrile precursor from the corresponding aldehyde, followed by the critical coupling step and final purification. Detailed standardized synthesis steps are provided below to guide process engineers in replicating these results.
- Convert 3-bromo-4-fluorobenzaldehyde to 3-bromo-4-fluorobenzonitrile using hydroxylamine sulfonic acid or iodine/ammonia.
- Perform coupling reaction with diethyl zinc or ethyl magnesium halides using a Pd or Ni catalyst.
- Purify the resulting solid product via crystallization in non-polar solvents like n-heptane or n-hexane.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route offers profound strategic advantages that extend far beyond simple chemical efficiency. By eliminating the dependency on tetraethyltin, manufacturers can drastically reduce the costs associated with hazardous waste disposal, specialized safety training, and regulatory compliance monitoring. The transition to a solid-state product further streamlines logistics, as solids are inherently easier to package, store, and transport than oils, reducing the risk of leakage and degradation during transit. These operational improvements collectively contribute to significant cost savings in pharmaceutical intermediate manufacturing, allowing companies to allocate resources more effectively toward R&D and market expansion.
- Cost Reduction in Manufacturing: The replacement of expensive and toxic tin reagents with commercially available organozinc or Grignard reagents results in a substantial decrease in raw material costs. Furthermore, the elimination of column chromatography in favor of crystallization significantly reduces solvent consumption and labor hours, leading to a leaner, more cost-effective production process. The ability to recover and recycle solvents like n-heptane and THF further enhances the economic viability of the route, ensuring long-term profitability and competitive pricing for downstream API customers.
- Enhanced Supply Chain Reliability: The use of cheap and easily obtained raw materials, such as 3-bromo-4-fluorobenzaldehyde, ensures a stable supply base that is less susceptible to market fluctuations or geopolitical disruptions. The robustness of the reaction conditions, which tolerate a wide range of temperatures and solvents, provides manufacturing flexibility that allows for rapid scale-up in response to surging demand. This reliability is crucial for maintaining continuous production schedules and meeting the tight delivery timelines required by pharmaceutical partners developing time-sensitive oncology therapies.
- Scalability and Environmental Compliance: The process is explicitly designed for industrial large-scale production, with safety profiles that align with modern green chemistry principles. By avoiding highly toxic reagents and minimizing waste generation through efficient crystallization, the method facilitates easier regulatory approval and environmental permitting. The scalability is further supported by the high yields reported in the examples, demonstrating that the chemistry translates effectively from gram-scale laboratory experiments to kilogram and ton-scale commercial reactors without loss of efficiency or purity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for industry stakeholders.
Q: Why is the new method superior to the traditional tetraethyltin route?
A: The traditional method utilizes highly toxic tetraethyltin, posing severe safety and environmental risks, and yields an oily product requiring complex column chromatography. The new method employs safer ethylating agents like diethyl zinc and yields a solid product purifiable by simple crystallization.
Q: What purity levels can be achieved with this synthesis route?
A: The patented process achieves product purity exceeding 99.5% through optimized reaction conditions and effective crystallization protocols, eliminating the need for preparative HPLC or silica gel chromatography.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the use of commercially available raw materials, mild reaction temperatures (20-80°C), and the formation of a solid final product make this route highly scalable and compliant with industrial safety standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Ethyl-4-Fluorobenzonitrile Supplier
As a premier CDMO and fine chemical manufacturer, NINGBO INNO PHARMCHEM is uniquely positioned to leverage this advanced synthetic technology to deliver high-purity 3-ethyl-4-fluorobenzonitrile to the global market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volumetric demands of major pharmaceutical clients. We operate state-of-the-art rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of intermediate meets the exacting standards required for clinical and commercial API synthesis.
We invite you to collaborate with us to optimize your supply chain for TAS-116 and related oncology programs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our implementation of this patented technology can drive value and security for your organization.
