Advanced Manufacturing of Fosfomycin Calcium Using Novel ZSM-5 Supported Catalysts
The pharmaceutical industry is constantly seeking more efficient and environmentally benign pathways for the synthesis of critical antibiotics, and the production of Fosfomycin Calcium is no exception. Patent CN113336791A introduces a groundbreaking production method for Fosfomycin Calcium bulk drug that addresses long-standing challenges in yield optimization and catalyst recovery. This innovation centers on the strategic use of a ZSM-5 molecular sieve-supported catalyst system, comprising sodium tungstate and sodium molybdate, which facilitates the epoxidation of cis-propenyl phosphonic acid under remarkably mild conditions. By shifting from traditional homogeneous catalysis to this advanced heterogeneous approach, the process not only enhances the stereochemical integrity of the final product but also drastically simplifies the downstream purification steps. For R&D directors and process chemists, this patent represents a significant leap forward in antibiotic manufacturing, offering a robust route that minimizes heavy metal contamination while maximizing overall process efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Fosfomycin has relied heavily on the Glamkowski synthesis method or variations involving homogeneous catalysts like free sodium tungstate or sodium molybdate. These traditional approaches suffer from inherent inefficiencies, particularly during the chiral resolution and epoxidation stages. The original research processes often yielded Fosfomycin (R)-1-phenethylamine salt at rates as low as 32.5%, with subsequent catalytic epoxidation steps struggling to exceed 28% to 30% yield. Beyond the poor atom economy, the use of soluble heavy metal catalysts creates a severe bottleneck in purification; removing trace tungsten or molybdenum residues to meet stringent pharmaceutical safety standards requires complex and costly additional processing steps. Furthermore, the difficulty in recovering these homogeneous catalysts leads to substantial chemical waste and increased operational expenditures, making the conventional routes less sustainable for large-scale commercial production.
The Novel Approach
The methodology disclosed in CN113336791A fundamentally reengineers the catalytic landscape by immobilizing the active catalytic species onto a ZSM-5 molecular sieve framework. This novel approach allows the epoxidation of cis-propenyl phosphonic acid to proceed effectively at lower temperatures, typically ranging between 35°C and 60°C, which preserves the stability of the sensitive epoxy ring. The ZSM-5 support ensures that the sodium tungstate and sodium molybdate are well-dispersed within the microporous channels, maintaining a highly active crystal structure throughout the reaction lifecycle. This structural innovation solves the critical issue of catalyst-product separation; because the catalyst remains solid and distinct from the liquid reaction phase, it can be easily filtered off and recycled. Consequently, this method eliminates the risk of heavy metal residues in the final API, ensuring a safer product profile while simultaneously boosting the yield of the Fosfomycin Calcium raw material to an impressive 49.6% to 51.2%.
Mechanistic Insights into ZSM-5 Supported Heterogeneous Epoxidation
The core of this technological advancement lies in the synergistic interaction between the transition metal oxides and the zeolite support. In the initial stage of the synthesis, cis-propenyl phosphonic acid undergoes a chiral resolution reaction with (R)-(+)-alpha-phenylethylamine. This step is crucial for establishing the correct stereochemistry required for the biological activity of Fosfomycin. The reaction proceeds in an ethanol solution where the acid and the resolving agent form a stable intermediate salt. This intermediate serves as the substrate for the subsequent oxidation, and its purity is paramount for the success of the epoxidation step. The precise control of temperature during this salt formation, maintained between 35°C and 40°C, ensures optimal crystallization and minimizes the formation of diastereomeric impurities that could complicate downstream processing.
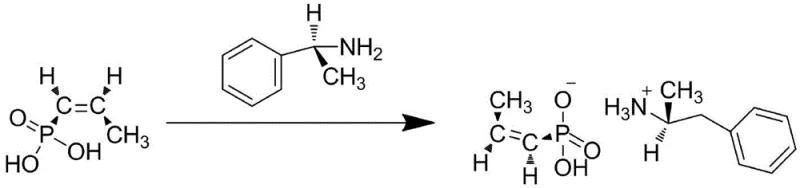
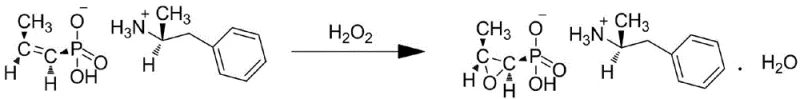
Following the formation of the intermediate, the system transitions to the critical epoxidation phase driven by the novel catalyst. The ZSM-5 molecular sieve, pre-treated to enhance its surface properties, acts as a nanoreactor. When hydrogen peroxide is introduced, the active sites within the zeolite pores facilitate the transfer of oxygen to the double bond of the propenyl group. The confinement effect of the ZSM-5 pores not only accelerates the reaction kinetics but also imposes steric constraints that favor the formation of the desired cis-epoxide configuration. This mechanism effectively suppresses side reactions such as over-oxidation or ring-opening hydrolysis, which are common pitfalls in homogeneous systems. The result is a clean conversion to the epoxypropenephosphonic acid derivative, which is then isolated as the final Fosfomycin Calcium bulk drug after workup.
How to Synthesize Fosfomycin Calcium Efficiently
The synthesis protocol outlined in the patent provides a clear, scalable pathway for producing high-purity Fosfomycin Calcium. The process begins with the preparation of the chiral intermediate, followed by the catalytic epoxidation using the specialized ZSM-5 system. Operators must adhere to strict temperature controls and addition rates, particularly during the dropwise addition of hydrogen peroxide, to manage the exothermic nature of the oxidation safely. The detailed标准化 synthesis steps见下方的指南 ensure that the catalyst maintains its structural integrity and that the final product meets all pharmacopeial standards for heavy metals and assay.
- Formation of Chiral Intermediate: React cis-propenyl phosphonic acid with (R)-(+)-alpha-phenylethylamine in ethanol at 35-40°C to form the resolved salt intermediate.
- Catalyst Preparation: Impregnate modified ZSM-5 molecular sieves with sodium tungstate and sodium molybdate to create a stable, reusable heterogeneous catalyst system.
- Epoxidation Reaction: Treat the intermediate with hydrogen peroxide and the ZSM-5 catalyst at 45-60°C to achieve epoxidation, followed by filtration and drying to isolate the final bulk drug.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this ZSM-5 catalyzed process offers tangible economic and logistical benefits that extend far beyond simple yield metrics. The primary value driver is the drastic simplification of the purification workflow. By eliminating the need for complex heavy metal scavenging steps, manufacturers can significantly reduce the consumption of auxiliary chemicals and shorten the overall batch cycle time. This streamlining translates directly into lower operational costs and a more agile production schedule, allowing facilities to respond more rapidly to market demand fluctuations without compromising on quality or compliance.
- Cost Reduction in Manufacturing: The implementation of a reusable heterogeneous catalyst system fundamentally alters the cost structure of Fosfomycin production. Unlike traditional homogeneous catalysts that are consumed or lost in the aqueous waste stream, the ZSM-5 supported catalyst can be recovered via simple filtration and regenerated for multiple cycles. This recyclability drastically reduces the recurring cost of expensive tungsten and molybdenum salts. Furthermore, the higher reaction yield means that less raw material is required to produce the same amount of finished API, optimizing the utilization of starting materials like cis-propenyl phosphonic acid and reducing the overall cost of goods sold.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route enhances supply chain security by reducing dependency on specialized purification resins or complex waste treatment protocols. The mild reaction conditions, operating effectively at temperatures below 60°C, reduce the energy load on manufacturing plants and minimize the risk of thermal runaways or equipment failure. This operational stability ensures consistent batch-to-batch quality and reliable delivery timelines, which is critical for maintaining uninterrupted supply of essential antibiotics to global healthcare markets.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this method aligns perfectly with modern green chemistry principles. The reduction in heavy metal waste simplifies effluent treatment and lowers the environmental footprint of the manufacturing site. The ability to scale this process from laboratory to commercial production is facilitated by the solid nature of the catalyst, which behaves predictably in large-scale reactors. This scalability ensures that manufacturers can meet increasing global demand for Fosfomycin while adhering to increasingly stringent environmental regulations regarding heavy metal discharge.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel Fosfomycin Calcium production technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear understanding of the process advantages.
Q: How does the ZSM-5 supported catalyst improve product purity compared to traditional methods?
A: The ZSM-5 molecular sieve acts as a rigid carrier that disperses sodium tungstate and sodium molybdate within its pore channels. This structural confinement prevents the catalyst components from mixing freely with the product, thereby eliminating heavy metal residues and toxic side effects often associated with homogeneous catalysts.
Q: What yield improvements can be expected with this novel production method?
A: According to patent data, this method achieves a yield range of 49.6% to 51.2% for the Fosfomycin Calcium bulk drug. This represents a significant increase over conventional literature methods which typically report yields between 28% and 33.5%.
Q: Is the catalyst system reusable for industrial scale-up?
A: Yes, a key advantage of this technology is the effective recovery and recycling of the catalyst. The heterogeneous nature of the ZSM-5 supported system allows for simple filtration and reuse, reducing raw material costs and environmental waste.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fosfomycin Calcium Supplier
The technological advancements detailed in CN113336791A underscore the potential for high-efficiency, low-impact antibiotic manufacturing. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial realities. Our commitment to quality is backed by rigorous QC labs and stringent purity specifications, guaranteeing that every batch of Fosfomycin Calcium we supply meets the highest international standards for safety and efficacy.
We invite pharmaceutical partners to leverage our technical expertise to optimize their supply chains. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our advanced manufacturing capabilities can drive value and reliability for your organization.
