Advanced Synthesis of Potent 23-Heteroatom OSW-1 Saponin Analogues for Oncology Applications
Advanced Synthesis of Potent 23-Heteroatom OSW-1 Saponin Analogues for Oncology Applications
The pharmaceutical industry is constantly seeking novel scaffolds with superior therapeutic indices, particularly in the realm of oncology. Patent CN1247610C introduces a groundbreaking class of 23-position heteroatom-substituted analogues of Tiger's Eye Rohdea Saponin OSW-1, a natural product renowned for its exceptional antitumor potency. Unlike traditional extraction methods which are plagued by low yields and supply inconsistencies, this patent discloses a robust, practical synthetic methodology that enables the scalable production of these high-value pharmaceutical intermediates. The core innovation lies in the strategic modification of the cholestane skeleton, specifically at the 23-position, allowing for oxygen, sulfur, or nitrogen substitution while maintaining the critical disaccharide moiety responsible for biological activity. This technological advancement provides a reliable pathway for developing next-generation anticancer agents with improved pharmacokinetic profiles.
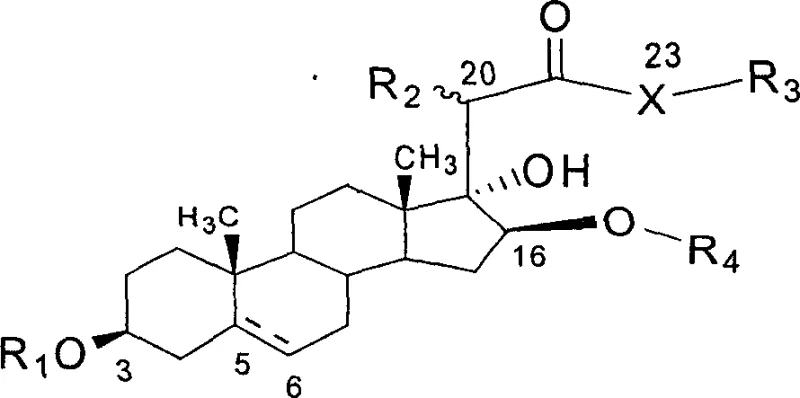
For research directors and procurement specialists, the significance of this patent extends beyond mere chemical novelty; it represents a viable solution to the supply chain bottlenecks associated with complex natural products. By establishing a defined synthetic route starting from commercially abundant steroid precursors, manufacturers can secure a consistent supply of high-purity intermediates essential for preclinical and clinical development. The ability to systematically vary the heteroatom at position 23 opens new avenues for optimizing drug efficacy and reducing toxicity, addressing a critical need in modern drug discovery. As a reliable pharmaceutical intermediate supplier, understanding these synthetic nuances is paramount for ensuring the continuity of pipeline projects dependent on these potent saponin derivatives.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the acquisition of OSW-1 and its related saponins has been heavily reliant on extraction from the underground corms of Ornithogalum saundersiae, a process fraught with significant logistical and economic challenges. The natural abundance of these compounds is exceedingly low, necessitating the processing of massive quantities of plant material to isolate minute amounts of the active ingredient, which drives up costs exponentially and raises sustainability concerns. Furthermore, natural extraction often results in a heterogeneous mixture of closely related analogues, making purification to the stringent standards required for pharmaceutical applications difficult and inefficient. The structural complexity of the OSW-1 molecule, particularly the specific stereochemistry of the disaccharide unit and the aglycone backbone, renders simple chemical modification of the natural product impractical for generating diverse libraries. Consequently, reliance on natural sources severely limits the ability of pharmaceutical companies to conduct extensive structure-activity relationship (SAR) studies or to secure the large-scale supply needed for commercial drug manufacturing.
The Novel Approach
The synthetic strategy outlined in Patent CN1247610C overcomes these historical barriers by employing a convergent total synthesis approach that builds the complex molecule from simpler, readily available building blocks. The method utilizes acetyl dehydroepiandrosterone or its dihydrogenated derivatives as the starting material, leveraging the inherent stereochemistry of the steroid nucleus to guide subsequent transformations. A key feature of this novel approach is the introduction of the side chain via an Aldol reaction, which simultaneously constructs the critical 17α-hydroxyl group with high stereocontrol. This eliminates the need for multiple protection and deprotection steps often seen in older synthetic routes, thereby streamlining the overall process. By enabling the systematic substitution of the heteroatom at the 23-position, this method allows for the creation of a diverse array of analogues that are inaccessible through natural extraction, providing a powerful tool for medicinal chemists to optimize therapeutic potential.
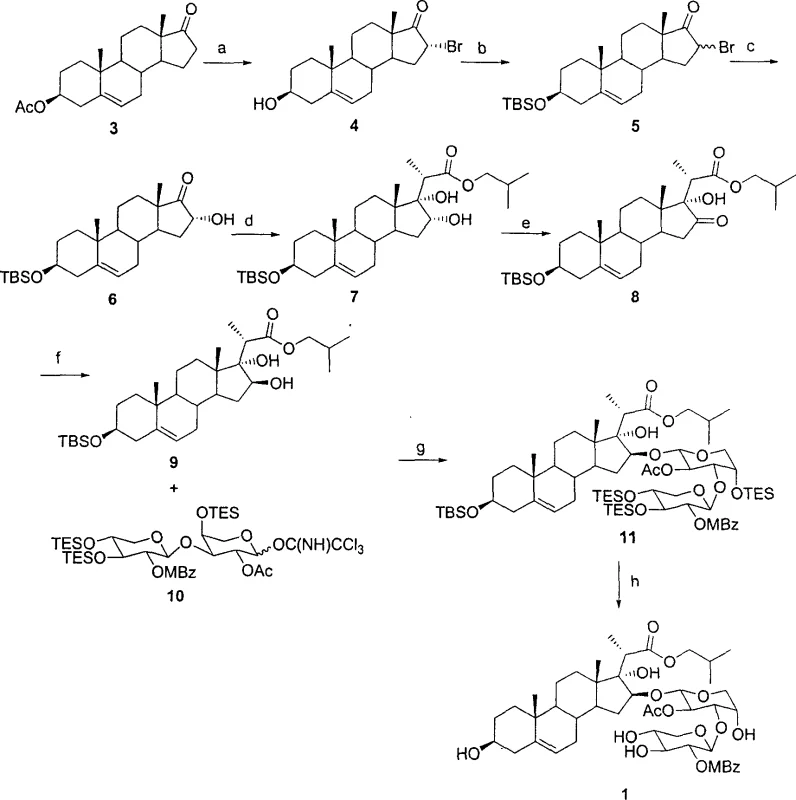
Mechanistic Insights into Stereoselective Aldol Condensation and Glycosylation
The chemical elegance of this synthesis is anchored in the precise control of stereochemistry during the construction of the D-ring side chain. The process begins with the introduction of a 16α-hydroxyl group, typically achieved through bromination followed by hydrolysis, which sets the stage for the pivotal Aldol reaction. In this step, a lithium enolate generated from an ester or thioester attacks the 17-ketone of the steroid intermediate. The steric environment created by the 16-hydroxyl group induces a high degree of diastereoselectivity, favoring the formation of the desired 17α-hydroxyl configuration. This stereochemical outcome is crucial, as the spatial arrangement of the side chain directly influences the molecule's interaction with biological targets. Following the Aldol condensation, the 16-hydroxyl is oxidized to a ketone and then selectively reduced using reagents like sodium borohydride in the presence of cerium chloride. This Luche reduction protocol ensures the inversion of the 16-hydroxyl configuration from alpha to beta, aligning the stereochemistry with that of the natural OSW-1 scaffold.
Subsequent glycosylation at the 16-position represents another critical mechanistic challenge addressed by this patent. The method employs trichloroacetimidate-activated glycosyl donors, specifically designed to mimic the unique disaccharide unit found in natural OSW-1, comprising D-xylose and L-arabinose. The use of catalytic Lewis acids, such as trimethylsilyl trifluoromethanesulfonate (TMSOTf), facilitates the formation of the glycosidic bond under mild conditions, preserving the integrity of the sensitive steroid backbone. The choice of protecting groups on the sugar moieties, such as acetyl and p-methoxybenzoyl groups, is strategically selected to ensure solubility and reactivity during the coupling phase while allowing for orthogonal removal in the final steps. This meticulous attention to mechanistic detail ensures that the final product possesses the exact structural features required for potent antitumor activity, minimizing the formation of inactive isomers that could complicate downstream purification and regulatory approval processes.
How to Synthesize 23-Heteroatom OSW-1 Analogues Efficiently
The synthesis of these complex saponin analogues requires a disciplined approach to reaction conditions and reagent selection to maximize yield and purity. The patented procedure outlines a logical sequence of transformations that convert inexpensive steroid precursors into high-value oncology intermediates. Key to the success of this route is the maintenance of anhydrous conditions during the organometallic steps, particularly the generation of enolates for the Aldol reaction, to prevent side reactions that could compromise the stereochemical integrity of the molecule. Additionally, the careful control of temperature during the glycosylation step is essential to achieve the desired alpha-linkage at the anomeric center. For detailed operational parameters, stoichiometry, and workup procedures, refer to the standardized synthesis guide below.
- Introduction of 16α-hydroxyl group using copper bromide reflux followed by protection and hydrolysis.
- Aldol reaction to introduce the side chain and construct the 17α-hydroxyl group simultaneously.
- Oxidation of the 16-hydroxyl to a ketone followed by stereoselective reduction to invert configuration.
- Glycosidation at the 16-position using activated trichloroacetimidate donors.
- Final deprotection of protecting groups to yield the target 23-heteroatom OSW-1 compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic methodology offers substantial advantages over traditional sourcing models, directly impacting the bottom line and operational resilience of pharmaceutical manufacturing. By shifting from a dependence on agricultural extraction to a chemical synthesis model, companies can decouple their supply chains from seasonal variations, geopolitical instability, and environmental regulations that often plague botanical sourcing. The use of commodity chemicals like acetyl dehydroepiandrosterone as starting materials ensures a stable and predictable cost base, shielding projects from the volatile price fluctuations associated with rare natural extracts. Furthermore, the modular nature of the synthesis allows for the parallel production of multiple analogues, enabling rapid iteration in drug discovery programs without the need to establish entirely new supply lines for each variant. This flexibility is invaluable for procurement managers tasked with securing materials for fast-moving clinical trials.
- Cost Reduction in Manufacturing: The synthetic route eliminates the need for expensive and inefficient natural extraction processes, which typically suffer from low recovery rates and high solvent consumption. By utilizing high-yielding chemical transformations such as the Aldol condensation and catalytic glycosylation, the overall material throughput is significantly increased, leading to a drastic reduction in the cost of goods sold (COGS). The avoidance of transition metal catalysts in certain steps further reduces the burden on downstream purification, removing the need for costly heavy metal scavenging resins and extensive chromatography. This streamlined process translates directly into lower production costs, making the development of OSW-1 based therapeutics economically viable for a broader range of indications.
- Enhanced Supply Chain Reliability: Relying on a synthetic route grounded in established organic chemistry principles ensures a consistent and reliable supply of critical intermediates. Unlike natural products, where batch-to-batch variability can be significant due to differences in plant growth conditions, synthetic intermediates offer reproducible quality and purity profiles. This consistency simplifies quality control protocols and reduces the risk of batch failures during manufacturing. For supply chain heads, this means shorter lead times and the ability to forecast material requirements with greater accuracy, ensuring that clinical and commercial production schedules are met without interruption. The scalability of the process from gram to kilogram scales further reinforces supply security.
- Scalability and Environmental Compliance: The synthetic method is designed with scalability in mind, utilizing reagents and solvents that are compatible with large-scale industrial reactors. The elimination of hazardous extraction solvents and the reduction of waste streams associated with plant processing contribute to a more environmentally sustainable manufacturing footprint. This alignment with green chemistry principles not only reduces disposal costs but also ensures compliance with increasingly stringent environmental regulations globally. The ability to scale up production without compromising yield or purity makes this technology an attractive option for contract development and manufacturing organizations (CDMOs) looking to expand their portfolio of oncology intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of 23-heteroatom OSW-1 analogues. These insights are derived directly from the technical specifications and experimental data provided in the patent documentation, offering clarity for stakeholders evaluating this technology for integration into their development pipelines. Understanding these details is crucial for making informed decisions regarding resource allocation and strategic partnerships.
Q: What is the biological activity of 23-heteroatom OSW-1 analogues?
A: These compounds exhibit extremely strong broad-spectrum antitumor activity, with IC50 values ranging from 0.1 to 0.7 nM, significantly outperforming standard chemotherapeutics like cisplatin and taxol in vitro.
Q: How does this synthesis method improve upon natural extraction?
A: Natural extraction yields are negligible and environmentally taxing. This synthetic route utilizes readily available steroid precursors like acetyl dehydroepiandrosterone, ensuring consistent quality and scalable production volumes.
Q: Can the heteroatom at position 23 be modified?
A: Yes, the patented method allows for flexibility at the 23-position, supporting oxygen, sulfur, or nitrogen substitutions, enabling the creation of diverse libraries for structure-activity relationship studies.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 23-Heteroatom OSW-1 Analogue Supplier
The synthesis of complex saponin analogues like those described in Patent CN1247610C requires a partner with deep expertise in steroid chemistry and glycosylation technologies. NINGBO INNO PHARMCHEM stands at the forefront of this field, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the rigorous demands of synthesizing high-purity pharmaceutical intermediates, ensuring that every batch meets stringent purity specifications through our rigorous QC labs. We understand the critical nature of oncology drug development and are committed to providing the reliability and quality necessary to advance your projects from bench to bedside.
We invite you to engage with our technical team to explore how our manufacturing capabilities can support your specific requirements for 23-heteroatom OSW-1 analogues. Whether you need custom synthesis for SAR studies or bulk supply for clinical trials, we are prepared to deliver a Customized Cost-Saving Analysis tailored to your project's unique constraints. Contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you accelerate your path to market with confidence.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
