Advanced Manufacturing of Rivaroxaban Intermediate 4-(4-Aminophenyl)-3-Morpholinone
Advanced Manufacturing of Rivaroxaban Intermediate 4-(4-Aminophenyl)-3-Morpholinone
The pharmaceutical landscape for anticoagulants has been revolutionized by direct Factor Xa inhibitors, with Rivaroxaban standing as a cornerstone therapy for preventing deep venous thrombosis and pulmonary embolism. At the heart of this therapeutic success lies the critical intermediate, 4-(4-aminophenyl)-3-morpholinone, whose synthesis quality directly dictates the purity and safety profile of the final Active Pharmaceutical Ingredient (API). A pivotal advancement in this domain is detailed in patent CN103524447A, which discloses a robust, cost-effective methodology for synthesizing this key morpholinone derivative. Unlike traditional pathways that rely on precious metal catalysis or harsh nitration processes, this novel approach leverages a strategic sequence of ring-opening, cyclization, and iron-mediated reduction. For global procurement leaders and R&D directors, understanding this technological shift is essential for securing a reliable pharmaceutical intermediate supplier capable of delivering high-quality precursors while optimizing supply chain resilience.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4-(4-aminophenyl)-3-morpholinone has been plagued by significant economic and technical bottlenecks that hinder efficient cost reduction in API manufacturing. As illustrated in prior art such as patent WO0147919, conventional routes often commence with expensive substrates like 3-morpholinone and p-fluoronitrobenzene, necessitating a nucleophilic substitution followed by reduction using Palladium on Carbon (Pd/C). The reliance on Pd/C introduces severe downstream challenges, including the rigorous and costly removal of residual heavy metals to meet ppm-level specifications required by regulatory bodies. Furthermore, alternative pathways described in documents like WO20066063113 utilize Copper Iodide (CuI) coupling reactions, which not only involve costly transition metals but also require stringent reaction conditions that complicate process control.
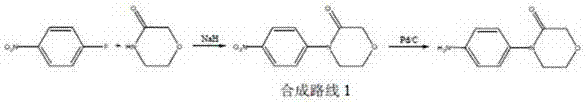
The dependency on these noble metal catalysts creates a volatile cost structure, as the prices of palladium and copper fluctuate wildly on the global market. Moreover, the starting materials for these conventional routes, particularly halogenated nitrobenzenes, are often subject to supply chain disruptions and environmental regulations regarding their production. The cumulative effect of expensive reagents, complex purification steps to remove metal residues, and lower overall yields renders these traditional methods suboptimal for large-scale commercial production. Consequently, manufacturers seeking a reliable pharmaceutical intermediate supplier often face inflated costs and extended lead times when relying on these legacy synthetic strategies.
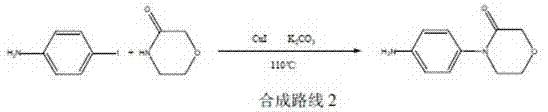
The Novel Approach
In stark contrast to the limitations of the past, the methodology outlined in patent CN103524447A presents a paradigm shift towards sustainable and economical synthesis. This innovative route bypasses the need for expensive halogenated aromatics and precious metal catalysts entirely. Instead, it initiates with p-Nitroaniline, a commodity chemical that is abundant, low-cost, and readily available in the global market. The process flows through a logical progression: first, a ring-opening reaction with ethylene oxide to form an amino-alcohol intermediate; second, a cyclization step using bromoacetyl bromide to construct the morpholinone core; and finally, a reduction step utilizing iron powder. This strategic redesign eliminates the most costly and environmentally burdensome aspects of the previous generations of synthesis.
Mechanistic Insights into Iron-Mediated Reduction and Cyclization
The chemical elegance of this new pathway lies in its mechanistic simplicity and robustness, offering R&D teams a clear view into the commercial scale-up of complex pharmaceutical intermediates. The initial step involves the nucleophilic attack of p-Nitroaniline on ethylene oxide within an ethanol/water solvent system at moderate temperatures ranging from 30-60°C. This ring-opening reaction is highly selective, generating nitro-N-β-anilino-ethanol (Compound II) with minimal byproduct formation. The subsequent cyclization is equally precise; by maintaining a strictly controlled alkaline environment with a pH value between 11-13 using inorganic bases like sodium hydroxide, the intermediate undergoes intramolecular closure with bromoacetyl bromide. This specific pH control is critical, as it ensures the deprotonation of the amine nitrogen to facilitate nucleophilic attack on the acyl bromide while preventing the hydrolysis of the reactive bromide reagent.
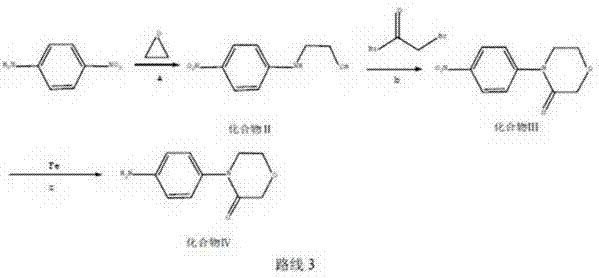
The final transformation, the reduction of the nitro group to the amine, represents the most significant departure from conventional wisdom. By employing iron powder in the presence of ammonium chloride at 70-80°C, the process achieves high conversion rates without introducing noble metal contaminants. This Bechamp-style reduction is historically proven yet often overlooked in modern fine chemistry due to the perception of lower efficiency; however, the patent data demonstrates that with optimized solvent systems (water) and precise stoichiometric control (iron to substrate ratio of 2.5-3.5:1), yields are exceptional. The mechanism proceeds through electron transfer from the iron surface to the nitro group, sequentially reducing it to the amine. This avoids the formation of azo or hydrazo impurities often seen in catalytic hydrogenation if not perfectly controlled, thereby simplifying the impurity profile and facilitating easier purification via recrystallization.
How to Synthesize 4-(4-Aminophenyl)-3-Morpholinone Efficiently
Implementing this synthesis route requires adherence to specific operational parameters to maximize yield and purity, making it an ideal candidate for technology transfer to manufacturing sites. The process is divided into three distinct unit operations that can be easily managed in standard glass-lined or stainless steel reactors. The initial alkylation requires careful temperature management to prevent polymerization of the epoxide, while the cyclization step demands precise pH monitoring to ensure complete ring closure. The final reduction is exothermic and requires controlled addition of the nitro-compound to the iron slurry to maintain thermal safety. For a detailed breakdown of the specific reagent quantities, reaction times, and workup procedures, please refer to the standardized protocol below.
- React p-Nitroaniline with ethylene oxide in an ethanol/water system at 30-60°C to form nitro-N-β-anilino-ethanol.
- Perform cyclization using bromoacetyl bromide and inorganic alkali at pH 11-13 to obtain 4-(4-nitrophenyl)-3-morpholinone.
- Reduce the nitro compound using iron powder and ammonium chloride in water at 70-80°C to yield the final amine product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers profound strategic advantages that extend far beyond simple unit price reductions. The shift away from precious metal catalysts fundamentally alters the cost structure of the intermediate, removing exposure to the volatile pricing of palladium and copper markets. Furthermore, the use of commodity chemicals like p-Nitroaniline and ethylene oxide ensures a stable and diversified supply base, significantly mitigating the risk of raw material shortages that frequently plague the pharmaceutical sector. This stability allows for more accurate long-term forecasting and inventory planning, essential for maintaining continuous API production lines.
- Cost Reduction in Manufacturing: The elimination of expensive noble metal catalysts such as Pd/C and CuI removes a major cost driver from the bill of materials. Additionally, the avoidance of complex metal scavenging steps—often required to reduce metal residues to single-digit ppm levels—drastically simplifies the downstream processing workflow. This simplification translates to reduced consumption of specialized filtration media and lower waste disposal costs associated with heavy metal containment. The overall process efficiency is enhanced by the use of inexpensive solvents like water and ethanol, further driving down the operational expenditure per kilogram of product.
- Enhanced Supply Chain Reliability: By utilizing p-Nitroaniline as the starting scaffold, the process leverages a chemical that is produced on a massive industrial scale for the dye and agrochemical industries, ensuring consistent availability. This contrasts sharply with specialized fluorinated or iodinated intermediates which may have limited suppliers and longer lead times. The robustness of the iron reduction step also means that the process is less sensitive to minor fluctuations in reaction conditions, leading to higher batch-to-batch consistency and fewer failed batches, which is critical for maintaining supply continuity for downstream API manufacturers.
- Scalability and Environmental Compliance: The process is inherently greener, avoiding the use of toxic organic solvents in the reduction step and eliminating the need for industrial nitration, a high-risk operation often restricted in many jurisdictions. The aqueous nature of the final reduction step simplifies effluent treatment, as iron salts are easier to manage and dispose of compared to complex organic-metallic waste streams. This environmental compatibility facilitates easier permitting for new production facilities and aligns with the increasing regulatory pressure for sustainable manufacturing practices in the fine chemical industry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of this Rivaroxaban intermediate. These insights are derived directly from the experimental data and comparative analysis provided in the patent literature, offering clarity on purity profiles, scalability, and regulatory compliance. Understanding these nuances is vital for technical teams evaluating potential manufacturing partners.
Q: How does this synthesis route improve cost efficiency compared to traditional methods?
A: This method replaces expensive noble metal catalysts like Palladium on Carbon (Pd/C) and Copper Iodide (CuI) with inexpensive iron powder. Additionally, it utilizes low-cost starting materials like p-Nitroaniline and ethylene oxide, significantly reducing raw material expenditure.
Q: What are the purity specifications achievable with this iron reduction method?
A: The patent data indicates that through careful control of reaction temperatures (70-80°C) and recrystallization using isopropanol/water systems, the final product can achieve purity levels exceeding 98.6%, meeting stringent pharmaceutical intermediate standards.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process avoids hazardous industrial nitration steps and uses common solvents like water, ethanol, and isopropanol. The operational simplicity and high total recovery rate (>55%) make it highly adaptable for commercial scale-up from kilograms to metric tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-(4-Aminophenyl)-3-Morpholinone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the integrity of your final drug product begins with the quality of its precursors. Our technical team has extensively analyzed the pathway described in CN103524447A and possesses the expertise to optimize this iron-reduction route for maximum efficiency. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale discovery to full-scale manufacturing is seamless. Our facilities are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications, consistently delivering material that meets or exceeds the 98% purity benchmark required for next-generation anticoagulant synthesis.
We invite you to collaborate with us to leverage this cost-effective technology for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data from recent pilot batches and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can enhance your project's bottom line while ensuring uninterrupted supply.
