Advanced Aminocyclopentadienyl Ruthenium Catalysts for Efficient Chiral Resolution and Commercial Scale-Up
Advanced Aminocyclopentadienyl Ruthenium Catalysts for Efficient Chiral Resolution and Commercial Scale-Up
The pharmaceutical and fine chemical industries constantly demand more efficient methods for obtaining enantiomerically pure compounds, particularly chiral secondary alcohols which serve as critical building blocks for active pharmaceutical ingredients. Patent CN1622932A introduces a groundbreaking advancement in this field by disclosing a novel class of aminocyclopentadienyl ruthenium complexes designed specifically for the dynamic kinetic resolution of chiral compounds. This technology addresses long-standing inefficiencies in traditional resolution methods, offering a pathway to high-yield production under remarkably mild conditions. By integrating these specialized metal catalysts with enzyme catalysts and alkenyl acetates, manufacturers can achieve superior stereochemical control while bypassing the harsh thermal requirements and complex purification steps associated with legacy technologies. This report analyzes the technical merits and commercial implications of this innovation for global supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional dynamic kinetic resolution processes have historically relied on ruthenium complexes such as the Shvo catalyst or [(p-cymene)RuCl2]2, which present significant operational drawbacks for large-scale manufacturing. The Shvo catalyst, typically existing as a dimer, necessitates high-temperature activation and often requires the addition of hydrogen transfer reagents like ketones to facilitate the racemization of the unwanted enantiomer. Furthermore, conventional methods frequently employ aryl acetates as acylating agents, which generate aromatic alcohols as stoichiometric by-products. These phenolic by-products possess physical properties similar to the desired product, making their removal via standard chromatography or crystallization extremely difficult and costly. Additionally, previous attempts to substitute aryl acetates with alkenyl acetates in the presence of standard metal catalysts often resulted in the undesirable oxidation of the target chiral alcohol, compromising both yield and purity.
The Novel Approach
The methodology described in patent CN1622932A fundamentally overcomes these barriers by introducing a structurally distinct aminocyclopentadienyl ruthenium complex that functions synergistically with alkenyl acetates. This new catalytic system effectively racemizes the undesired enantiomer at moderate temperatures without the need for external hydrogen transfer reagents, thereby streamlining the reaction setup. Crucially, the specific ligand environment of this novel ruthenium complex prevents the oxidation side reactions that typically plague the use of alkenyl acetates with other metal catalysts. By utilizing alkenyl acetates such as isopropenyl acetate or vinyl acetate, the acylation by-product becomes a volatile ketone or aldehyde rather than a stubborn aromatic alcohol. This shift dramatically simplifies the work-up procedure, allowing for easier isolation of the high-purity chiral ester and significantly reducing the burden on downstream processing units.
Mechanistic Insights into Aminocyclopentadienyl Ruthenium-Catalyzed Racemization
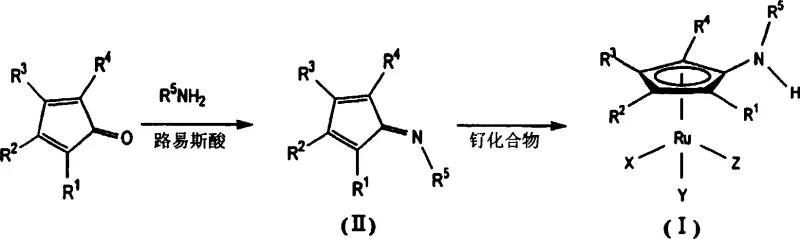
The core of this technological breakthrough lies in the unique structure of the Formula (I) ruthenium complex, which features an aminocyclopentadienyl ligand coordinated to the metal center. In the dynamic kinetic resolution process, this complex acts as a highly efficient racemization catalyst, rapidly interconverting the R and S enantiomers of the starting chiral alcohol in the presence of a base. Simultaneously, a lipase enzyme selectively acylates one specific enantiomer using the alkenyl acetate, pulling the equilibrium towards the desired product. The aminocyclopentadienyl ligand provides a specific electronic and steric environment that stabilizes the ruthenium hydride species necessary for hydrogen transfer during racemization, yet remains inert enough to prevent the oxidative degradation of the substrate. This delicate balance allows the reaction to proceed at temperatures ranging from 0°C to 120°C, offering flexibility for temperature-sensitive substrates that would decompose under the harsher conditions required by older catalyst systems.
Furthermore, the impurity profile of the reaction is meticulously controlled through the choice of acylating agent and the stability of the catalyst. Unlike aryl acetates which leave behind phenolic residues that can poison enzymes or contaminate the final API, the cleavage of alkenyl acetates yields volatile fragments that can be easily removed by evaporation. The patent data indicates that this system maintains high enantioselectivity, often exceeding 99% ee, even when using robust industrial lipases like Candida antarctica lipase B. The compatibility of the ruthenium complex with various bases, including inorganic carbonates and organic amines, further enhances the process robustness, ensuring that the racemization rate matches the enzymatic acylation rate to prevent the accumulation of the unwanted enantiomer. This mechanistic harmony is essential for achieving theoretical yields of up to 100% in kinetic resolutions, a significant improvement over the 50% limit of classical resolution.
How to Synthesize Aminocyclopentadienyl Ruthenium Complexes Efficiently
The preparation of the active catalyst involves a straightforward two-step synthetic route that utilizes readily available starting materials, making it accessible for custom synthesis partners. The process begins with the condensation of a cyclopentadienone derivative with a primary amine in the presence of a Lewis acid to form an imine intermediate, followed by complexation with a ruthenium source. This modular approach allows for the tuning of the catalyst's steric properties by varying the amine substituent, enabling optimization for specific substrate classes. For detailed operational parameters, stoichiometry, and purification protocols required to reproduce these high-performance catalysts in a GMP environment, please refer to the standardized synthesis guide below.
- React a cyclopentadienone derivative with a primary amine in the presence of a Lewis acid in an aprotic solvent to form the imine intermediate.
- Combine the resulting imine compound with a ruthenium source such as Ru3(CO)12 in a haloform solvent under heating to yield the final ruthenium complex.
- Utilize the synthesized catalyst in a kinetic resolution setup with a lipase enzyme and alkenyl acetate to resolve chiral secondary alcohols.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this aminocyclopentadienyl ruthenium catalytic system offers tangible benefits in terms of cost structure and operational reliability. The elimination of difficult-to-remove aromatic by-products translates directly into reduced solvent consumption and shorter processing times during the purification phase. By avoiding complex chromatographic separations or multiple recrystallizations needed to remove phenolic impurities, manufacturing facilities can significantly increase their throughput capacity without additional capital investment. The ability to use volatile acylating by-products also simplifies waste management protocols, reducing the environmental footprint and associated disposal costs. These factors collectively contribute to a more lean and agile manufacturing process that is better equipped to handle fluctuating market demands for chiral intermediates.
- Cost Reduction in Manufacturing: The shift from aryl acetates to alkenyl acetates fundamentally alters the economics of the purification stage. Since the by-products are volatile and easily separated, the reliance on expensive chromatography resins and large volumes of elution solvents is drastically minimized. This reduction in material usage leads to substantial cost savings per kilogram of produced intermediate. Additionally, the mild reaction conditions reduce energy consumption for heating and cooling, further lowering the overall utility costs associated with the production batch. The high catalytic efficiency also means that lower loadings of the precious metal catalyst may be sufficient, optimizing the utilization of expensive ruthenium resources.
- Enhanced Supply Chain Reliability: The robustness of this catalytic system under mild conditions enhances the reliability of the supply chain by reducing the risk of batch failures due to thermal degradation. Traditional methods requiring high temperatures are more prone to side reactions and safety incidents, which can disrupt production schedules. By operating at lower temperatures, the process becomes safer and more predictable, ensuring consistent delivery timelines for downstream customers. Furthermore, the reagents involved, such as simple primary amines and cyclopentadienones, are commercially available from multiple global suppliers, mitigating the risk of raw material shortages that could otherwise halt production lines.
- Scalability and Environmental Compliance: Scaling this technology from laboratory to commercial production is facilitated by the simplicity of the work-up procedure. The absence of persistent organic pollutants like phenolic by-products simplifies compliance with increasingly stringent environmental regulations regarding wastewater and solid waste. The process generates less hazardous waste, aligning with green chemistry principles and reducing the regulatory burden on manufacturing sites. This environmental compatibility makes the technology attractive for production in regions with strict ecological standards, ensuring long-term viability and uninterrupted supply for global pharmaceutical partners seeking sustainable sourcing options.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this resolution technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the process capabilities. Understanding these details is crucial for R&D teams evaluating the feasibility of integrating this catalytic system into their existing process workflows for chiral alcohol production.
Q: What is the primary advantage of using alkenyl acetates over aryl acetates in this resolution process?
A: Using alkenyl acetates avoids the formation of aromatic alcohol by-products, which are notoriously difficult to separate from the reaction mixture, thereby significantly simplifying downstream purification.
Q: How does the novel aminocyclopentadienyl ruthenium catalyst improve upon traditional Shvo catalysts?
A: Unlike Shvo catalysts which require high temperatures for activation and hydrogen transfer reagents, this novel catalyst operates effectively under mild conditions and prevents the oxidation of chiral alcohols often seen with other metal catalysts.
Q: Can this technology be scaled for industrial production of chiral intermediates?
A: Yes, the process utilizes robust reaction conditions and commercially available reagents, making it highly suitable for commercial scale-up of complex pharmaceutical intermediates with high optical purity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aminocyclopentadienyl Ruthenium Complex Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating advanced academic research into practical industrial solutions, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is well-versed in the nuances of organometallic catalysis and enzymatic resolution, ensuring that the transition from patent literature to commercial reality is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of chiral intermediate meets the exacting standards required by the global pharmaceutical industry. Our commitment to quality ensures that the high optical purity and yield demonstrated in the patent are consistently replicated at scale.
We invite procurement leaders and R&D directors to engage with our technical procurement team to discuss how this innovative resolution technology can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to this cleaner, more efficient process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that will enhance your supply chain resilience and product competitiveness in the marketplace.
