Advanced Inclusion Resolution Technology for Scalable Optical Purity Thiosulfinate Manufacturing
Advanced Inclusion Resolution Technology for Scalable Optical Purity Thiosulfinate Manufacturing
The landscape of chiral intermediate manufacturing is undergoing a significant transformation, driven by the need for more robust and scalable synthetic routes that can meet the rigorous demands of modern pharmaceutical development. A pivotal advancement in this domain is documented in Chinese Patent CN1219758C, which discloses a novel method for preparing optical purity di-tert-butyl thioester-sulfinate, specifically 2-methyl-propane-2-thiosulfinic acid S-tert-butyl ester. This patent introduces a sophisticated inclusion resolution technique that utilizes optically pure resolving agents to simultaneously separate both configurations of the target molecule from a racemic mixture. Unlike traditional methods that rely on sensitive catalytic oxidations, this approach leverages the thermodynamic stability of host-guest inclusion complexes to achieve high stereoselectivity. For R&D directors and process chemists, this represents a critical shift towards methodologies that offer greater control over impurity profiles and stereochemical outcomes, ensuring that the resulting intermediates meet the exacting standards required for downstream asymmetric synthesis of bioactive compounds.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of optically pure sulfoxides, particularly tert-butyl sulfoxides, has relied heavily on asymmetric catalytic oxidation strategies, such as the method developed by J.A. Ellman involving the oxidation of di-tert-butyl disulfide. While scientifically elegant, these conventional catalytic routes suffer from severe practical limitations when translated to industrial scale. The reaction kinetics and stereoselectivity are notoriously sensitive to physical parameters such as solvent shape, stirring velocity, and even the specific geometry and size of the reaction flask. This sensitivity creates a significant barrier to scale-up, as conditions optimized in a laboratory glassware setting often fail to reproduce in large-scale stainless steel reactors due to differences in heat transfer and mixing efficiency. Furthermore, a single chiral catalyst typically yields only one specific enantiomer, meaning that producing the opposite configuration requires a completely different catalyst system or a cumbersome inversion step, effectively halving the theoretical yield from the starting racemic material and complicating supply chain logistics for manufacturers needing both isomers.
The Novel Approach
In stark contrast to the fragility of asymmetric catalysis, the methodology outlined in patent CN1219758C employs a classical yet highly refined inclusion resolution strategy that transforms these liabilities into assets. By utilizing readily available optically pure resolving agents—specifically binaphthol derivatives or tartaric acid-based dioxolanes—the process converts the racemic thiosulfinate into diastereomeric inclusion complexes that possess distinct physical properties, primarily solubility. This allows for separation via standard unit operations like crystallization and filtration, which are inherently more scalable and easier to control than sensitive catalytic oxidations. Crucially, this method enables the simultaneous recovery of both the (R) and (S) configurations from a single batch; while one isomer precipitates as a complex, the opposing isomer remains enriched in the mother liquor and can be recovered subsequently. This dual-recovery capability fundamentally alters the economics of production, effectively doubling the utility of the raw racemic starting material and providing a reliable source for both enantiomers without the need for separate synthetic campaigns.
Mechanistic Insights into Chiral Inclusion Resolution
The core of this technological breakthrough lies in the supramolecular interaction between the chiral host (the resolving agent) and the guest (the racemic thiosulfinate). The resolving agents function as molecular hosts that create a chiral environment capable of discriminating between the two enantiomers of the substrate based on steric fit and intermolecular forces such as hydrogen bonding and pi-stacking interactions. When the racemic mixture interacts with an optically pure agent like (R)-binaphthol, one enantiomer of the thiosulfinate fits more snugly into the chiral pocket of the host, forming a less soluble crystalline lattice that precipitates out of the solution. The other enantiomer, experiencing greater steric repulsion or weaker binding affinity, remains preferentially dissolved in the mother liquor. This thermodynamic differentiation is the driving force behind the separation, allowing for the isolation of high-purity isomers through successive recrystallization steps. The robustness of this mechanism ensures that minor fluctuations in process parameters do not catastrophically impact the optical purity, offering a safety margin that is absent in kinetic resolution or asymmetric catalysis.
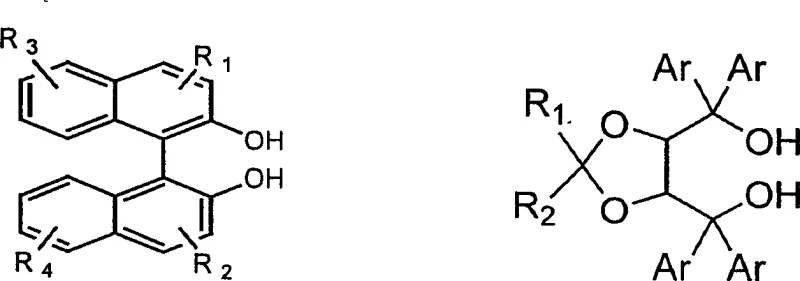
Beyond the primary separation event, the process incorporates sophisticated mechanisms for impurity control and agent recovery that are vital for commercial viability. The patent details specific protocols for decomposing the inclusion complexes, such as using hydrazine hydrate for substitution or employing vacuum distillation, which allows for the gentle release of the target thiosulfinate without degrading its sensitive sulfur-oxygen bond. Moreover, the resolving agents themselves are designed for recyclability; after the product is liberated, the binaphthol or dioxolane derivatives can be recovered through column chromatography or distillation and reused in subsequent batches. This closed-loop system minimizes the generation of chiral waste and significantly lowers the environmental footprint of the manufacturing process. For quality assurance teams, this means a consistent and well-defined impurity profile, as the crystallization steps inherently act as purification stages, excluding non-complexing impurities and ensuring that the final active pharmaceutical ingredient (API) precursors meet stringent regulatory specifications for chirality and chemical purity.
How to Synthesize 2-Methyl-Propane-2-Thiosulfinic Acid S-Tert-Butyl Ester Efficiently
Implementing this resolution technology requires a precise understanding of solvent systems and stoichiometric ratios to maximize the formation of the desired inclusion complex. The patent elucidates a versatile range of operating conditions, utilizing solvents ranging from polar alcohols like ethanol and isopropanol to non-polar hydrocarbons like n-hexane and aromatics like toluene. The choice of solvent dictates the solubility differential between the diastereomeric complexes, and optimization often involves mixed solvent systems to fine-tune the crystallization kinetics. Operators must carefully control the molar ratio of the resolving agent to the substrate, typically ranging from 0.3:1 to 1.2:1, to balance between yield and optical purity. Detailed standardized synthesis steps see the guide below.
- Mix racemic 2-methyl-propane-2-thiosulfinic acid S-tert-butyl ester with an optically pure resolving agent such as binaphthol in a suitable solvent like toluene or ethanol.
- Induce crystallization or suspension through reflux and cooling to form diastereomeric inclusion complexes, then filter to separate the solid complex from the mother liquor.
- Decompose the inclusion complex using post-separation techniques or hydrazine hydrate substitution to recover the resolving agent and isolate the high-purity optical isomer.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from asymmetric catalysis to inclusion resolution offers profound strategic advantages that extend far beyond simple yield metrics. The primary economic driver is the drastic simplification of the supply chain for chiral materials; instead of sourcing expensive, specialized transition metal catalysts that may have long lead times and supply volatility, this process relies on binaphthol and tartaric acid derivatives. These are commodity chiral pool chemicals produced on a massive global scale, ensuring a stable and continuous supply with minimal risk of disruption. Furthermore, the ability to recycle both the resolving agents and the solvents creates a circular economy within the manufacturing plant, significantly reducing the consumption of raw materials per kilogram of finished product. This reduction in material intensity directly translates to lower variable costs and a more predictable cost structure, shielding the organization from fluctuations in the prices of specialty reagents.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts and the implementation of agent recycling loops fundamentally reshape the cost basis of production. By avoiding the need for expensive重金属 removal steps and complex catalyst synthesis, the process streamlines the workflow and reduces utility consumption. The qualitative improvement in cost efficiency is driven by the high recovery rates of the resolving agents, which can be reused multiple times without significant loss of activity, thereby amortizing the initial cost of the chiral auxiliary over a much larger volume of product. Additionally, the use of common industrial solvents like ethanol and hexane, rather than exotic fluorinated or chlorinated solvents often required for sensitive catalysis, further drives down operational expenditures and waste disposal costs.
- Enhanced Supply Chain Reliability: Reliance on widely available chiral building blocks such as binaphthol mitigates the risk of single-source dependency that often plagues proprietary catalyst systems. Since the resolving agents are derived from established chemical feedstocks, procurement teams can leverage multiple suppliers to negotiate better terms and ensure backup options are always available. The robustness of the crystallization process also means that production schedules are less susceptible to delays caused by batch failures; unlike catalytic reactions that might stall due to trace impurities or oxygen ingress, inclusion resolution is a thermodynamic equilibrium process that is far more forgiving of minor variations in feedstock quality, ensuring consistent on-time delivery to downstream customers.
- Scalability and Environmental Compliance: From an EHS (Environment, Health, and Safety) perspective, this methodology aligns perfectly with green chemistry principles by minimizing waste generation and avoiding toxic heavy metals. The scalability is inherent to the unit operations involved; crystallization and filtration are standard processes in any multipurpose chemical plant, requiring no specialized high-pressure or cryogenic equipment. This ease of scale-up allows manufacturers to rapidly respond to market demand surges, moving from pilot kilogram quantities to multi-ton commercial production with minimal capital investment. The reduced solvent usage and the ability to recycle aqueous and organic streams significantly lower the facility's environmental burden, simplifying regulatory compliance and enhancing the corporate sustainability profile.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this inclusion resolution technology. These answers are derived directly from the experimental data and technical specifications provided in the patent literature, offering clarity on process capabilities and limitations. Understanding these nuances is essential for project managers evaluating the feasibility of adopting this route for their specific pipeline candidates.
Q: What are the primary advantages of inclusion resolution over asymmetric oxidation for thiosulfinate production?
A: Inclusion resolution offers superior scalability and operational robustness compared to asymmetric oxidation, which is highly sensitive to reactor geometry and stirring conditions. Furthermore, resolution allows for the simultaneous recovery of both enantiomers from a single batch, maximizing raw material utilization.
Q: Can the chiral resolving agents be recycled in this process?
A: Yes, the patent explicitly details methods for recovering the resolving agents, such as binaphthol derivatives, through column separation or vacuum distillation after the product is isolated. This recyclability is a key factor in reducing the overall cost of goods sold (COGS).
Q: What level of optical purity (e.e. value) can be achieved with this method?
A: Through optimized crystallization and recrystallization steps, the process can achieve optical purity values exceeding 98% e.e., making the resulting intermediates suitable for stringent pharmaceutical applications requiring high stereochemical integrity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Methyl-Propane-2-Thiosulfinic Acid S-Tert-Butyl Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of chiral intermediates requires more than just a patent; it demands deep process engineering expertise and a commitment to quality that spans the entire production lifecycle. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We understand the critical nature of stereochemical purity in pharmaceutical applications, which is why our facilities are equipped with rigorous QC labs and advanced analytical instrumentation to verify stringent purity specifications for every batch. By leveraging the inclusion resolution technology described in CN1219758C, we can offer a supply solution that balances high optical purity with cost-effectiveness, providing our partners with a competitive edge in their own drug development timelines.
We invite you to engage with our technical procurement team to discuss how this advanced manufacturing route can be tailored to your specific project needs. Whether you require a Customized Cost-Saving Analysis to evaluate the economic benefits of switching to this resolution method or need specific COA data to validate the quality of our intermediates, we are prepared to provide comprehensive support. Contact us today to request route feasibility assessments and discover how our scalable production capabilities can secure your supply chain for high-purity thiosulfinate derivatives.
