Advanced Synthesis of Chiral Spiro Bisphosphine Ligands for Asymmetric Catalysis
Advanced Synthesis of Chiral Spiro Bisphosphine Ligands for Asymmetric Catalysis
The landscape of asymmetric catalysis is continually evolving, driven by the demand for higher enantioselectivity and operational efficiency in the production of chiral pharmaceutical intermediates. Patent CN1562926A introduces a groundbreaking class of novel chiral spirocyclic diphosphine ligands that address critical limitations in existing catalytic systems. This technology centers on a unique spiro[fluorene-9,9'-xanthene] backbone, which provides a rigid chiral environment essential for precise stereocontrol. The patent details not only the ligand structure but also a comprehensive synthetic methodology and its successful application in the asymmetric catalytic hydrogenation of α,β-unsaturated carboxylic acids. For R&D directors and procurement specialists, this represents a significant opportunity to enhance process robustness while securing a reliable supply of high-value chiral building blocks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional chiral diphosphine ligands, such as BINAP or DIPAMP, have served the industry well but often exhibit suboptimal performance with specific substrate classes, particularly sterically hindered α,β-unsaturated carboxylic acids. Conventional methods frequently suffer from moderate turnover numbers and inconsistent enantioselectivity, necessitating higher catalyst loadings and complex downstream purification to remove metal residues. Furthermore, the synthesis of many established ligands relies on expensive chiral pool starting materials or difficult resolution processes that limit scalability. These factors contribute to elevated manufacturing costs and extended lead times, creating bottlenecks in the supply chain for high-purity pharmaceutical intermediates. The inability to achieve high conversion rates efficiently often forces manufacturers to compromise on either yield or optical purity.
The Novel Approach
The methodology outlined in CN1562926A overcomes these hurdles through a modular and scalable synthetic strategy. The process begins with readily available commodity chemicals, specifically substituted dibromobenzenes and meta-bromoanisoles, which undergo palladium-catalyzed coupling to form biphenyl precursors. This is followed by a lithiation-carbonylation sequence and a critical ring-closure step using polyphosphoric acid to construct the rigid spiro fluorene skeleton. 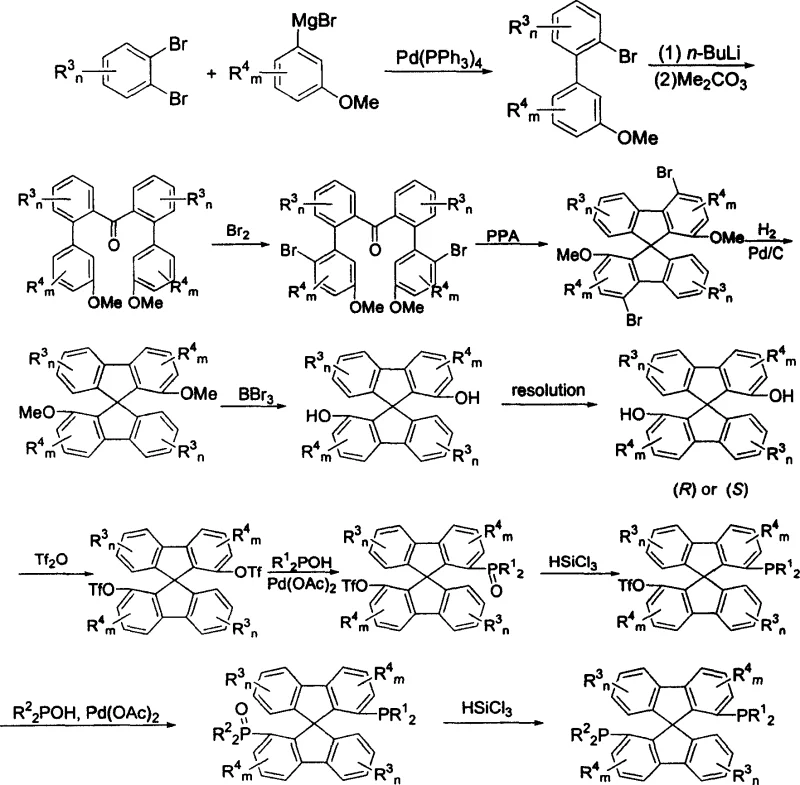 . This structural rigidity is key to the ligand's performance, locking the biaryl axes into a stable chiral conformation that minimizes flexibility-induced loss of selectivity. By utilizing a stepwise phosphination strategy involving triflate intermediates, the synthesis allows for the introduction of diverse phosphine groups, enabling fine-tuning of the electronic and steric properties for specific catalytic applications.
. This structural rigidity is key to the ligand's performance, locking the biaryl axes into a stable chiral conformation that minimizes flexibility-induced loss of selectivity. By utilizing a stepwise phosphination strategy involving triflate intermediates, the synthesis allows for the introduction of diverse phosphine groups, enabling fine-tuning of the electronic and steric properties for specific catalytic applications.
Mechanistic Insights into Spiro Backbone Rigidity and Catalytic Activity
The exceptional performance of this ligand class stems from its unique three-dimensional architecture. The spiro linkage at the 9,9'-position of the fluorene units creates a C2-symmetric environment that effectively transfers chiral information to the metal center. When complexed with divalent ruthenium compounds, such as ruthenium acetate or ruthenium chloride dimers, the ligand forms a highly active catalytic species. 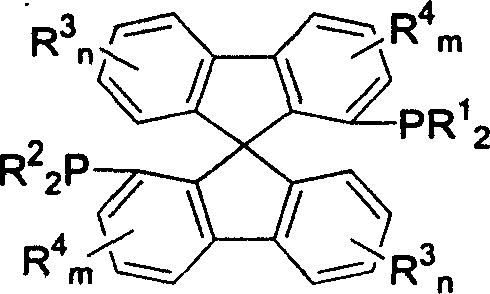 . The mechanism involves the coordination of the α,β-unsaturated carboxylic acid substrate to the ruthenium center, where the bulky spiro framework directs the approach of hydrogen to the prochiral double bond. This steric guidance ensures that hydride transfer occurs exclusively from one face of the molecule, resulting in the observed high enantiomeric excess (ee) values of up to 98%. The rigid backbone prevents unfavorable conformational changes during the catalytic cycle, maintaining high stereoselectivity even under varying reaction conditions.
. The mechanism involves the coordination of the α,β-unsaturated carboxylic acid substrate to the ruthenium center, where the bulky spiro framework directs the approach of hydrogen to the prochiral double bond. This steric guidance ensures that hydride transfer occurs exclusively from one face of the molecule, resulting in the observed high enantiomeric excess (ee) values of up to 98%. The rigid backbone prevents unfavorable conformational changes during the catalytic cycle, maintaining high stereoselectivity even under varying reaction conditions.
Impurity control is another critical aspect addressed by this mechanistic design. The synthetic route includes a dedicated resolution step for the spiro diphenol intermediate using chiral resolving agents like tartaric acid derivatives. This ensures that only the desired enantiomer ((R) or (S)) proceeds to the final phosphination stages, preventing the formation of diastereomeric impurities that could complicate downstream processing. The subsequent conversion of hydroxyl groups to triflates and their displacement by phosphine oxides proceeds with inversion or retention depending on the specific conditions, allowing for precise control over the final ligand configuration. The final reduction of phosphine oxides to phosphines using trichlorosilane completes the synthesis, yielding a ligand with minimal metal contamination potential prior to complexation. 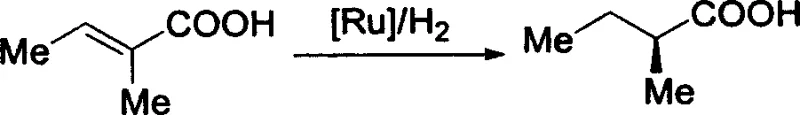 .
.
How to Synthesize Spiro Bisphosphine Ligands Efficiently
The synthesis of these advanced ligands requires careful attention to reaction conditions, particularly during the lithiation and cyclization phases. The process is designed to be robust, utilizing standard organic solvents like THF and toluene, which facilitates technology transfer from laboratory to pilot plant. The key to success lies in the efficient construction of the spiro core and the high-yielding phosphination steps. Detailed standardized operating procedures for each stage, including temperature controls and stoichiometry, are essential for reproducibility.
- Perform Pd-catalyzed coupling of substituted dibromobenzenes with Grignard reagents to form biphenyl precursors.
- Execute lithiation and reaction with dimethyl carbonate followed by polyphosphoric acid cyclization to form the spiro fluorene core.
- Conduct demethylation, chiral resolution of the diphenol intermediate, triflation, and sequential palladium-catalyzed phosphination to yield the final ligand.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology offers tangible benefits beyond mere technical performance. The reliance on commodity starting materials significantly de-risks the supply chain, reducing dependency on scarce natural products or highly specialized precursors. This accessibility translates directly into cost stability and improved availability for long-term production contracts. Furthermore, the high turnover number (S/C = 10,000) reported in the patent implies that significantly less catalyst is required per kilogram of product, drastically reducing the cost of goods sold (COGS) associated with precious metal usage.
- Cost Reduction in Manufacturing: The synthetic route eliminates the need for expensive chiral pool starting materials by constructing chirality through resolution of a synthetic intermediate. This shift from extraction-based to synthesis-based chirality allows for substantial cost savings and scalability. Additionally, the high catalytic activity reduces the loading of expensive ruthenium metal, lowering the overall material cost per batch. The streamlined purification steps further reduce solvent consumption and waste disposal costs, contributing to a leaner manufacturing profile.
- Enhanced Supply Chain Reliability: By utilizing a fully synthetic route based on petrochemical derivatives, the supply of these ligands is decoupled from agricultural or biological variability. This ensures consistent quality and uninterrupted supply, which is critical for Just-In-Time manufacturing environments in the pharmaceutical sector. The robustness of the Pd-coupling and cyclization steps means that production can be scaled rapidly to meet surging demand without compromising on the stringent purity specifications required for API intermediates.
- Scalability and Environmental Compliance: The process avoids the use of highly toxic reagents where possible and employs standard workup procedures compatible with existing industrial infrastructure. The high efficiency of the catalyst reduces the environmental footprint associated with metal waste. Scaling from 100 kgs to 100 MT is feasible due to the use of common unit operations like crystallization and filtration, ensuring that the technology remains viable and compliant with increasingly strict environmental regulations regarding heavy metal discharge.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this spiro ligand technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making.
Q: What is the enantioselectivity performance of this spiro ligand in hydrogenation?
A: According to patent CN1562926A, the ruthenium acetate complex of this ligand achieves up to 98% ee in the asymmetric hydrogenation of alpha,beta-unsaturated carboxylic acids.
Q: How does the catalyst loading compare to traditional systems?
A: The system demonstrates a high turnover number (S/C) of up to 10,000, allowing for significantly reduced catalyst loading compared to many conventional phosphine ligands.
Q: Is the synthesis scalable for industrial production?
A: Yes, the route utilizes standard industrial reactions such as Grignard coupling and Pd-catalyzed cross-coupling, which are amenable to large-scale manufacturing from 100 kgs to multi-ton quantities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Spiro Bisphosphine Ligand Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this novel spirocyclic diphosphine ligand technology for the fine chemical and pharmaceutical industries. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to full-scale manufacturing is seamless. Our facility is equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of ligand or catalyst complex delivered meets the highest standards of enantiomeric excess and chemical purity required for sensitive asymmetric hydrogenation processes.
We invite you to collaborate with our technical team to evaluate the feasibility of integrating this high-performance ligand into your existing process workflows. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to this superior catalytic system. Please contact our technical procurement team today to索取 specific COA data and route feasibility assessments tailored to your specific α,β-unsaturated carboxylic acid substrates.
