Scalable Chiral Resolution of Dihydropyrimidine Intermediates for Antiviral Drug Manufacturing
Scalable Chiral Resolution of Dihydropyrimidine Intermediates for Antiviral Drug Manufacturing
The pharmaceutical industry's relentless pursuit of potent antiviral agents has placed significant emphasis on the efficient production of chiral intermediates, particularly those belonging to the dihydropyrimidine class. Patent CN101041658A introduces a groundbreaking resolution method for 2-heterocyclic substituted dihydropyrimidine racemic compounds, addressing the critical bottleneck of obtaining single enantiomers with high optical purity. This technology is particularly relevant for the synthesis of next-generation Hepatitis B virus (HBV) inhibitors, such as derivatives of the well-known HAP compound Bay 41-4109. By leveraging a specific binaphthol phosphate resolving agent, this process circumvents the limitations of traditional chiral chromatography, offering a robust pathway for reliable pharmaceutical intermediate supplier operations. The method ensures that the final active pharmaceutical ingredients meet the stringent stereochemical requirements necessary for biological activity, thereby securing the therapeutic efficacy of the end-product drug.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the isolation of single enantiomers for complex heterocyclic compounds like 2-heterocyclic substituted dihydropyrimidines has relied heavily on preparative chiral high-performance liquid chromatography (HPLC). While effective on a laboratory scale, this technique presents severe economic and logistical barriers when transitioning to commercial manufacturing. The loading capacity of chiral columns is inherently low, requiring massive amounts of expensive stationary phase and large volumes of mobile phase solvents to produce kilogram quantities of material. Furthermore, the throughput is limited, leading to extended production cycles that cannot meet the aggressive timelines demanded by global supply chains. Alternative chemical crystallization methods, such as those described in earlier patents like US024878A1, often struggle to balance yield and purity, frequently yielding products with optical purities below the 95% threshold required for clinical safety. These conventional approaches result in inflated cost structures and supply chain vulnerabilities that hinder the widespread availability of life-saving antiviral medications.
The Novel Approach
The innovative strategy outlined in the patent data utilizes a diastereomeric salt formation technique employing single optically active binaphthol phosphate derivatives, specifically (R)-(-)-1,1’-binaphthyl-2,2’-diylhydrogenphosphate or its (S)-(+) enantiomer. This approach fundamentally shifts the separation mechanism from adsorption-based chromatography to solubility-based crystallization, which is inherently more scalable. 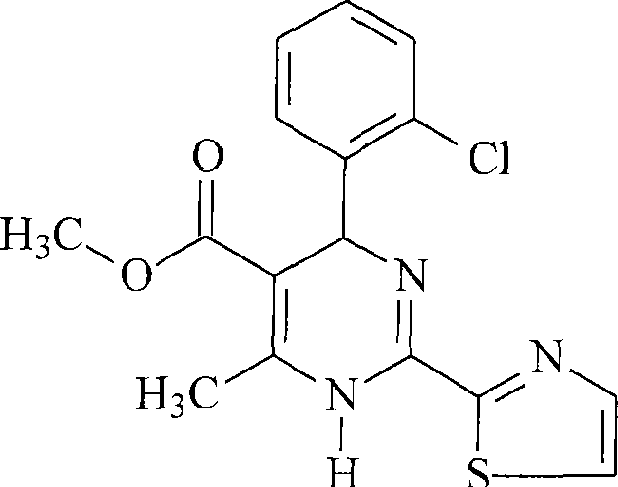 As illustrated by the specific embodiment of methyl 4-(2-chlorophenyl)-6-methyl-2-(thiazol-2-yl)-1,4-dihydropyrimidine-5-carboxylate, the process achieves optical purities exceeding 99% through a carefully controlled sequence of solvent mixing, stirring, and temperature-regulated crystallization. By optimizing the ratio of polar to non-polar solvents and precisely managing the freezing conditions between -20°C and 5°C, the method maximizes the precipitation of the desired diastereomeric salt while keeping the unwanted enantiomer in the mother liquor. This results in a stable, reproducible process that is exceptionally well-suited for industrial application, effectively solving the scalability issues associated with previous technologies.
As illustrated by the specific embodiment of methyl 4-(2-chlorophenyl)-6-methyl-2-(thiazol-2-yl)-1,4-dihydropyrimidine-5-carboxylate, the process achieves optical purities exceeding 99% through a carefully controlled sequence of solvent mixing, stirring, and temperature-regulated crystallization. By optimizing the ratio of polar to non-polar solvents and precisely managing the freezing conditions between -20°C and 5°C, the method maximizes the precipitation of the desired diastereomeric salt while keeping the unwanted enantiomer in the mother liquor. This results in a stable, reproducible process that is exceptionally well-suited for industrial application, effectively solving the scalability issues associated with previous technologies.
Mechanistic Insights into Binaphthol Phosphate-Mediated Chiral Resolution
The core of this technology lies in the stereoselective interaction between the racemic dihydropyrimidine substrate and the chiral binaphthol phosphate resolving agent. The dihydropyrimidine scaffold, characterized by its nitrogen-containing heterocyclic ring, possesses basic sites capable of forming ionic or strong hydrogen-bonding interactions with the acidic phosphate group of the resolving agent. 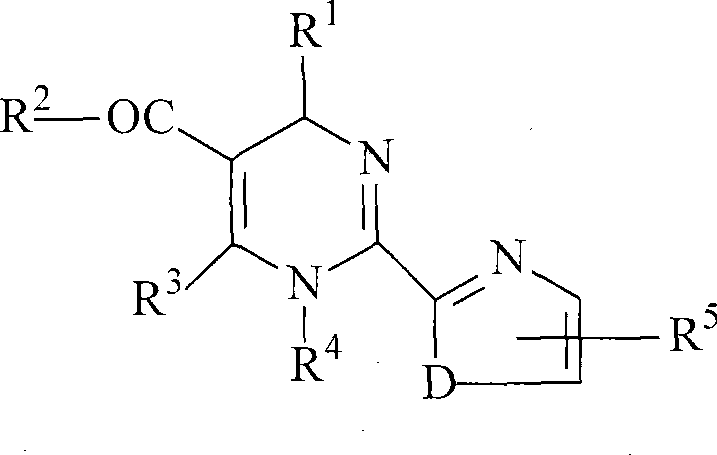 As depicted in the general structural formula, the variability at positions R1 through R5 allows for a broad scope of substrates, yet the steric bulk of the binaphthyl backbone provides a rigid chiral environment that differentiates between the enantiomers. When the racemate and the resolving agent are mixed in a binary solvent system, they form two distinct diastereomeric salts. Due to their different spatial arrangements, these salts exhibit significantly different solubility profiles in the chosen solvent mixture. The less soluble diastereomer precipitates out of the solution upon concentration and cooling, while the more soluble counterpart remains dissolved. This thermodynamic differentiation is the driving force behind the high efficiency of the separation, allowing for the isolation of the target enantiomer with minimal contamination from its mirror image.
As depicted in the general structural formula, the variability at positions R1 through R5 allows for a broad scope of substrates, yet the steric bulk of the binaphthyl backbone provides a rigid chiral environment that differentiates between the enantiomers. When the racemate and the resolving agent are mixed in a binary solvent system, they form two distinct diastereomeric salts. Due to their different spatial arrangements, these salts exhibit significantly different solubility profiles in the chosen solvent mixture. The less soluble diastereomer precipitates out of the solution upon concentration and cooling, while the more soluble counterpart remains dissolved. This thermodynamic differentiation is the driving force behind the high efficiency of the separation, allowing for the isolation of the target enantiomer with minimal contamination from its mirror image.
Impurity control is rigorously maintained through the multi-step workup procedure described in the patent. After the initial crystallization and filtration, the mother liquor is concentrated and subjected to a secondary freezing step, which recovers additional product and enhances the overall mass balance. The final liberation of the free base involves treatment with a weak base salt solution, such as 10% to 30% sodium carbonate or potassium bicarbonate. This neutralization step is critical; it breaks the ionic bond between the dihydropyrimidine and the phosphate resolver without degrading the sensitive heterocyclic core. Subsequent extraction and washing with saturated brine remove residual inorganic salts and traces of the resolving agent, ensuring that the final product meets the rigorous purity specifications required for downstream coupling reactions. This meticulous attention to downstream processing guarantees that the intermediate is not only optically pure but also chemically clean, reducing the burden on subsequent purification steps in the API synthesis.
How to Synthesize High-Purity Dihydropyrimidine Enantiomers Efficiently
Implementing this resolution protocol requires precise adherence to the solvent ratios and temperature controls specified in the intellectual property. The process begins with the dissolution of the racemic starting material and the chiral resolving agent in a mixture of polar solvents like dichloromethane or acetone and non-polar anti-solvents like petroleum ether or cyclohexane. The detailed standardized synthesis steps see the guide below for exact operational parameters regarding molar ratios and stirring times. Following the reaction, the strategic manipulation of solubility through concentration and freezing is the key to success. The recovery of the free base from the diastereomeric salt must be performed under mild alkaline conditions to prevent racemization or decomposition. This streamlined workflow eliminates the need for specialized chromatographic equipment, allowing standard chemical manufacturing facilities to produce high-value chiral intermediates with confidence and consistency.
- Mix the racemic dihydropyrimidine compound with a single optically active binaphthol phosphate resolving agent in a reactor containing a mixture of polar and non-polar solvents.
- Stir the reaction mixture at room temperature for 0.5 to 3 hours to allow complete formation of diastereomeric salts.
- Filter the mixture, concentrate the mother liquor to 40-60% of its original volume, and freeze the concentrate at temperatures between -20°C and 5°C to induce crystallization.
- Filter the frozen mother liquor to isolate the intermediate product with single optical activity, then concentrate the remaining liquor to dryness.
- Dissolve the intermediate in an organic solvent, treat with a 10-30% weak base salt solution (e.g., sodium carbonate) to liberate the free base, then extract and purify.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this binaphthol phosphate resolution method represents a significant strategic advantage in the sourcing of antiviral intermediates. By moving away from dependency on preparative HPLC services, manufacturers can drastically reduce the cost of goods sold (COGS) associated with chiral separation. The elimination of expensive chiral columns and the reduction in solvent consumption per kilogram of product directly translate to substantial cost savings in pharmaceutical intermediate manufacturing. Furthermore, the use of commodity solvents such as dichloromethane, ethyl acetate, and petroleum ether ensures that raw material availability remains high and price volatility is minimized. This stability is crucial for long-term supply agreements and budget forecasting, allowing partners to secure reliable pricing models without the risk of sudden spikes in processing costs.
- Cost Reduction in Manufacturing: The transition from chromatographic separation to crystallization-based resolution removes the capital expenditure associated with maintaining high-pressure liquid chromatography systems. Additionally, the resolving agent, binaphthol phosphate, can potentially be recovered and recycled from the mother liquors, further enhancing the economic viability of the process. The simplified operational workflow reduces labor hours and energy consumption, contributing to a leaner manufacturing profile that supports competitive pricing strategies for the final API.
- Enhanced Supply Chain Reliability: Scalability is the primary driver of supply chain security. Unlike chiral columns which have fixed loading limits, crystallization tanks can be scaled up linearly from liters to cubic meters with minimal process re-engineering. This inherent scalability ensures that production volumes can be rapidly increased to meet surges in demand for hepatitis B treatments without the lead time delays typically associated with procuring additional chromatographic capacity. The robustness of the method also means fewer batch failures, ensuring a consistent flow of material to downstream synthesis units.
- Scalability and Environmental Compliance: The process utilizes standard organic solvents that are well-understood in terms of waste management and recovery. The ability to concentrate mother liquors and recover solvents aligns with green chemistry principles by minimizing waste generation. Moreover, the avoidance of silica gel waste from chromatography columns significantly reduces the solid waste burden on the facility. This environmental efficiency simplifies regulatory compliance and supports the sustainability goals of modern pharmaceutical enterprises, making the supply chain more resilient to environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this resolution technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on the method's capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing pipelines.
Q: What is the optical purity achievable with this binaphthol phosphate resolution method?
A: According to patent CN101041658A, this resolution method consistently achieves an optical purity of over 99%, with specific embodiments demonstrating up to 99.5% ee, which is critical for antiviral drug efficacy.
Q: How does this method compare to chiral column chromatography for industrial scale-up?
A: While chiral column chromatography offers high purity, it is prohibitively expensive and difficult to scale for tonnage production. This chemical resolution method utilizes standard crystallization and filtration equipment, making it significantly more viable for commercial manufacturing.
Q: What types of solvents are required for this resolution process?
A: The process utilizes a binary solvent system comprising a polar solvent (such as dichloromethane, acetone, or ethyl acetate) and a non-polar solvent (such as petroleum ether, cyclohexane, or heptane) to optimize the solubility differences between diastereomeric salts.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Heterocyclic Substituted Dihydropyrimidine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity chiral intermediates in the development of next-generation antiviral therapies. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering materials that meet stringent purity specifications, utilizing our rigorous QC labs to verify optical purity and chemical identity at every stage of production. Our infrastructure is designed to handle complex chiral resolutions safely and effectively, guaranteeing the supply continuity that global pharmaceutical partners demand.
We invite you to collaborate with us to leverage this advanced resolution technology for your specific project needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can optimize your supply chain and accelerate your drug development timeline. Let us be your partner in bringing high-quality antiviral medications to the market faster and more economically.
