Advanced Base-Catalyzed Nitrile Hydrolysis for Commercial Amide Production
The chemical manufacturing landscape is continuously evolving towards greener, more efficient synthetic pathways, and patent CN103232359A represents a significant breakthrough in the synthesis of primary amides. This intellectual property details an environmental-friendly nitrile hydrolysis method that fundamentally shifts the paradigm from harsh acidic conditions or expensive transition metal catalysis to a mild, base-catalyzed system operating in air. For R&D directors and process chemists, this technology offers a robust solution for converting diverse nitrile compounds into their corresponding primary amides with exceptional selectivity. The core innovation lies in the use of inexpensive, low-toxicity base catalysts, such as cesium hydroxide, which facilitate the reaction at moderate temperatures ranging from room temperature to 120°C. This approach not only simplifies the operational requirements by removing the need for inert gas protection but also significantly enhances the safety profile of the manufacturing process. By addressing the common issue of over-hydrolysis, which typically leads to unwanted carboxylic acid byproducts, this method ensures high recovery rates and simplifies downstream purification, making it an ideal candidate for the scalable production of high-purity pharmaceutical intermediates and fine chemicals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the industrial synthesis of amides from nitriles has been plagued by significant technical and economic hurdles that impact both product quality and operational costs. Conventional methods often rely on the use of excess strong acids, such as sulfuric acid, which necessitate harsh reaction conditions that limit the compatibility of sensitive functional groups on the substrate. Furthermore, the acidic medium generates substantial environmental pollution and creates complex post-processing challenges due to the need for neutralization and waste disposal. Alternatively, alkaline hydrolysis methods have been employed, but these frequently suffer from uncontrolled over-hydrolysis, where the desired amide intermediate is further converted into the corresponding carboxylic acid, drastically reducing the yield of the target product. While some strategies involve adding hydrogen peroxide to inhibit this over-hydrolysis, they introduce severe safety hazards that preclude their use in large-scale commercial manufacturing. Additionally, transition metal-catalyzed methods, though effective, often require costly noble metals, complex ligands, and strict inert atmosphere conditions, leading to high production costs and potential heavy metal contamination in the final active pharmaceutical ingredients.
The Novel Approach
In stark contrast to these legacy technologies, the novel approach described in patent CN103232359A utilizes a non-transition metal base catalyst to achieve efficient hydrolysis under mild and green conditions. This method operates successfully in an air atmosphere, eliminating the capital and operational expenses associated with inert gas systems and specialized pressure equipment. The reaction employs a mixed solvent system of DMSO and water, which optimizes the solubility of organic nitriles while providing the necessary medium for hydrolysis without promoting the formation of carboxylic acid byproducts. By carefully controlling the catalyst loading and reaction temperature, typically between 60°C and 100°C, the process achieves high conversion rates while maintaining excellent selectivity for the primary amide. This technological leap allows for a streamlined workflow where product separation and purification are significantly simplified, resulting in higher overall recovery rates. The elimination of toxic heavy metals and the use of commercially available, low-cost catalysts make this route particularly attractive for the cost reduction in fine chemical manufacturing, offering a sustainable pathway for producing complex amide structures.
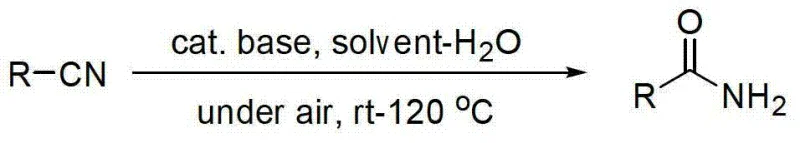
Mechanistic Insights into Base-Catalyzed Nitrile Hydrolysis
The mechanistic foundation of this green hydrolysis method relies on the nucleophilic attack of water on the nitrile carbon, activated by the base catalyst without the involvement of transition metal centers. The base, preferably cesium hydroxide, facilitates the hydration of the nitrile group to form an intermediate imidic acid tautomer, which subsequently rearranges to the stable primary amide. The unique solvent environment created by the DMSO and water mixture plays a critical role in stabilizing the transition states and preventing the further hydrolysis of the amide to the carboxylic acid. This selectivity is paramount for R&D teams focused on impurity control, as it ensures that the final product profile is clean and free from acidic degradation products that are difficult to separate. The reaction kinetics are optimized such that the conversion proceeds efficiently within 12 to 24 hours, balancing reaction speed with energy consumption. Furthermore, the absence of transition metals means there is no risk of metal-ligand complex formation that could complicate the reaction mechanism or lead to catalyst deactivation over time. This straightforward mechanistic pathway allows for predictable scale-up and consistent batch-to-batch quality, which is essential for maintaining stringent purity specifications in pharmaceutical applications.
Impurity control is a critical aspect of this synthesis, particularly given the regulatory requirements for pharmaceutical intermediates. The use of a non-transition metal catalyst inherently eliminates the risk of heavy metal residues, which are a major concern in drug substance manufacturing and require costly removal steps. The mild reaction conditions also preserve sensitive functional groups on the aromatic or heteroaromatic rings, preventing side reactions such as halogen displacement or ring degradation that can occur under harsh acidic or high-temperature conditions. The patent data demonstrates high conversion rates across a broad substrate scope, including substituted benzonitriles, heterocyclic nitriles, and aliphatic nitriles, indicating a robust tolerance for various electronic and steric environments. This broad compatibility ensures that the impurity profile remains manageable even for complex molecules, reducing the burden on analytical teams during method development. By minimizing the formation of byproducts like carboxylic acids or unreacted starting materials, the process simplifies the crystallization or chromatography steps required for isolation, thereby enhancing the overall efficiency of the production line and ensuring the delivery of high-purity pharmaceutical intermediates to the market.
How to Synthesize Primary Amides Efficiently
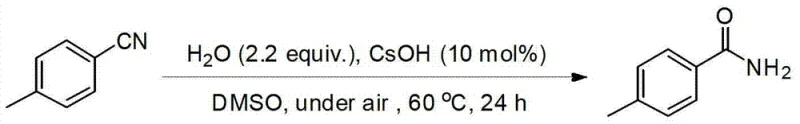
The implementation of this synthesis route involves a straightforward procedure that begins with the preparation of a reaction mixture containing the specific nitrile substrate, a catalytic amount of base such as CsOH, and a defined ratio of DMSO to water. The reaction is then heated to the optimal temperature range under ambient air pressure, allowing the hydrolysis to proceed to completion without the need for specialized atmospheric controls. Detailed standardized synthesis steps see the guide below.
- Prepare a reaction mixture containing the nitrile substrate, a base catalyst such as CsOH (5-30 mol%), and a mixed solvent system of DMSO and water.
- Heat the reaction mixture to a temperature between 60°C and 100°C under an air atmosphere, maintaining the condition for 12 to 24 hours.
- Upon completion, isolate the primary amide product through standard separation and purification techniques such as column chromatography to ensure high purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this base-catalyzed hydrolysis technology translates into tangible strategic advantages that directly impact the bottom line and operational resilience. The primary benefit lies in the drastic simplification of the manufacturing infrastructure, as the elimination of inert gas protection systems reduces both capital expenditure on equipment and ongoing operational costs associated with gas consumption and monitoring. Furthermore, the use of inexpensive, commercially available base catalysts instead of precious transition metals significantly lowers the raw material costs, contributing to substantial cost savings in the overall production budget. The mild reaction conditions also enhance safety protocols, reducing the risk of accidents and lowering insurance and compliance costs related to hazardous chemical handling. From a supply chain perspective, the robustness of the reaction under air conditions ensures greater flexibility in production scheduling and facility utilization, as it does not compete for limited inert gas resources or specialized reactor lines. This reliability is crucial for maintaining continuous supply streams to downstream customers, minimizing the risk of production delays caused by equipment failure or resource constraints.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the replacement of expensive noble metal catalysts with low-cost base catalysts like cesium hydroxide or potassium carbonate, which are readily available in the global chemical market. This substitution eliminates the need for complex catalyst recovery systems and expensive ligand synthesis, further driving down the variable costs per kilogram of product. Additionally, the simplified workup procedure reduces the consumption of solvents and purification media, leading to lower waste disposal costs and improved atom economy. The energy efficiency of running reactions at moderate temperatures compared to high-pressure or high-temperature alternatives also contributes to reduced utility bills, making the process economically viable for large-scale commercial production. These cumulative factors result in a highly competitive cost structure that allows for better pricing strategies in the global market.
- Enhanced Supply Chain Reliability: The operational simplicity of running reactions under air atmosphere significantly de-risks the supply chain by removing dependencies on specialized infrastructure that can be prone to bottlenecks. Since the catalysts are stable and easily sourced, there is minimal risk of supply disruption due to raw material scarcity, ensuring consistent production capabilities. The high tolerance of the method to various substrates means that the same production line can be adapted for multiple products with minimal changeover time, increasing asset utilization and responsiveness to market demand. This flexibility allows supply chain managers to optimize inventory levels and reduce lead times for high-purity pharmaceutical intermediates, ensuring that customer orders are fulfilled promptly and reliably. The robustness of the process also means fewer batch failures, leading to a more predictable and stable supply of critical chemical building blocks.
- Scalability and Environmental Compliance: Scaling this technology from laboratory to commercial production is facilitated by the absence of hazardous reagents and the use of standard chemical engineering unit operations. The green nature of the process, characterized by low toxicity and minimal waste generation, aligns perfectly with increasingly stringent environmental regulations and corporate sustainability goals. The elimination of heavy metals simplifies the environmental compliance process, reducing the regulatory burden and potential fines associated with toxic effluent discharge. Furthermore, the high selectivity of the reaction minimizes the formation of byproducts that require complex treatment, making the waste stream easier to manage and treat. This environmental stewardship not only protects the company from regulatory risks but also enhances its reputation as a responsible manufacturer, which is increasingly valued by global partners and stakeholders in the chemical industry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this nitrile hydrolysis technology, based on the specific advantages and data outlined in the patent documentation. These answers are designed to provide clarity on the operational feasibility and quality benefits of the method for potential partners and technical teams. Understanding these details is crucial for evaluating the fit of this technology within existing manufacturing portfolios and supply chain strategies.
Q: Does this hydrolysis method require inert gas protection?
A: No, the patented process operates effectively under standard air conditions, eliminating the need for expensive nitrogen or argon protection systems.
Q: How does this method prevent over-hydrolysis to carboxylic acids?
A: The specific base catalyst and solvent system control the reaction kinetics, stopping the hydrolysis at the primary amide stage with high selectivity and yield.
Q: Are there heavy metal residues in the final amide product?
A: No, the method utilizes non-transition metal base catalysts, ensuring the final product is free from toxic heavy metal contamination common in traditional catalytic methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Primary Amides Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this green hydrolysis technology and have integrated similar advanced synthetic pathways into our CDMO capabilities to serve the global pharmaceutical and fine chemical industries. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of primary amides meets the highest quality standards required for drug development and manufacturing. Our infrastructure is designed to handle the specific requirements of base-catalyzed reactions, providing a safe and compliant environment for the production of complex intermediates.
We invite you to collaborate with us to optimize your supply chain and leverage these cost-effective synthetic routes for your next project. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific molecule and volume requirements. We encourage you to reach out for specific COA data and route feasibility assessments to understand how our capabilities can enhance your production efficiency and reduce time-to-market. Let us partner with you to drive innovation and reliability in your chemical supply chain.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
