Advanced Enzymatic Resolution for Commercial (-)-Menthol Production and Supply Stability
The global demand for (-)-menthol, a cornerstone compound in the flavors, fragrances, and pharmaceutical industries, continues to outpace the volatile supply of natural peppermint oil. Patent CN1240646C presents a transformative technical breakthrough that addresses the critical instability of natural sourcing by offering a robust, fully synthetic route to high-purity (-)-menthol. Unlike traditional extraction methods which are susceptible to agricultural fluctuations and seasonal variations in major producing regions like India and China, this patented process leverages advanced biocatalysis to ensure consistent quality and supply continuity. The core innovation lies in a highly selective enzymatic resolution step that isolates the desired (-)-stereoisomer from a racemic mixture with exceptional precision. By integrating this biological specificity with efficient chemical recycling loops, the technology offers a compelling alternative for manufacturers seeking to decouple their supply chains from agricultural risks while maintaining the sensory profile that consumers expect.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of synthetic menthol has relied heavily on non-selective hydrogenation of thymol, which generates a complex mixture of eight stereoisomers, including the desired (-)-menthol alongside unwanted diastereomers like neomenthol and isomenthol. Conventional separation techniques, such as fractional distillation or crystallization, are inherently inefficient for this purpose because the physical properties (boiling points and melting points) of these stereoisomers are remarkably similar. This similarity necessitates energy-intensive multi-stage purification processes that often result in significant product loss and low overall yields. Furthermore, without a mechanism to recycle the unwanted isomers, nearly half of the synthesized material becomes low-value byproducts, creating substantial waste disposal challenges and inflating the cost of goods sold. These inefficiencies make traditional synthetic routes less competitive against natural extraction when oil prices are low, and they fail to provide the consistent purity profiles required by high-end pharmaceutical applications.
The Novel Approach
The methodology outlined in CN1240646C fundamentally shifts the paradigm from physical separation to chemical differentiation. Instead of attempting to physically separate the isomers, the process employs a stereoselective enzyme, specifically a lipase or esterase, to target only the hydroxyl group of the desired (-)-menthol stereoisomer. This enzyme catalyzes the esterification of the target molecule into a menthyl ester, while leaving the other seven stereoisomers chemically untouched. This chemical modification creates a significant difference in polarity and boiling point between the product and the impurities, allowing for easy separation via simple distillation or extraction. Crucially, the process includes a racemization step for the unconverted isomers, converting them back into a racemic mixture that can be reintroduced into the resolution reactor. This closed-loop system theoretically allows for 100% conversion of the starting racemic material into the desired product, eliminating the 50% yield ceiling typical of classical resolutions and drastically improving atom economy.
Mechanistic Insights into Enzymatic Kinetic Resolution
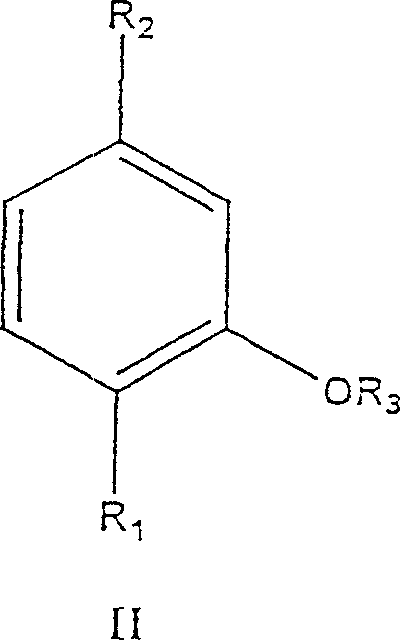
The heart of this technological advancement is the kinetic resolution driven by stereoselective biocatalysts. The process begins with the precursor chemistry, where m-cresol is alkylated to form thymol, which is subsequently hydrogenated to produce the racemic menthol mixture. 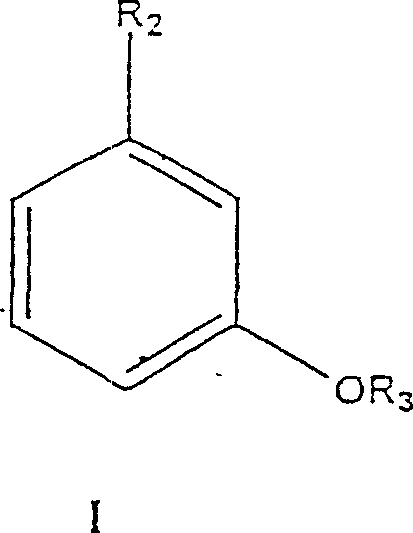
 Once the racemic mixture (Formula III) is obtained, it is dissolved in a suitable organic solvent, such as n-heptane, and treated with an acyl donor like vinyl acetate in the presence of the enzyme. The enzyme, such as the preferred Amano AK lipase from Pseudomonas fluorescens, possesses an active site geometry that strictly accommodates the (-)-enantiomer. Upon binding, the enzyme facilitates the nucleophilic attack of the alcohol oxygen on the ester carbonyl, forming the (-)-menthyl acetate. The specificity is so high that the reaction proceeds with optical yields approaching 98%, ensuring that the resulting ester fraction is virtually free of the unwanted (+)-isomers or diastereomers. This high selectivity is maintained under mild reaction conditions, typically around 50°C, which preserves the structural integrity of the terpene skeleton and prevents thermal degradation often seen in harsher chemical catalysis.
Once the racemic mixture (Formula III) is obtained, it is dissolved in a suitable organic solvent, such as n-heptane, and treated with an acyl donor like vinyl acetate in the presence of the enzyme. The enzyme, such as the preferred Amano AK lipase from Pseudomonas fluorescens, possesses an active site geometry that strictly accommodates the (-)-enantiomer. Upon binding, the enzyme facilitates the nucleophilic attack of the alcohol oxygen on the ester carbonyl, forming the (-)-menthyl acetate. The specificity is so high that the reaction proceeds with optical yields approaching 98%, ensuring that the resulting ester fraction is virtually free of the unwanted (+)-isomers or diastereomers. This high selectivity is maintained under mild reaction conditions, typically around 50°C, which preserves the structural integrity of the terpene skeleton and prevents thermal degradation often seen in harsher chemical catalysis.
Impurity control is intrinsic to this mechanism due to the binary nature of the enzymatic reaction: a molecule is either esterified or it is not. Unlike chemical catalysts which might promote side reactions like dehydration or rearrangement of the terpene backbone, the biocatalyst operates with surgical precision on the hydroxyl functionality. Following the reaction, the mixture contains the desired ester, the unreacted isomers, excess acyl donor, and the solvent. The enzyme itself, often immobilized on a support like diatomaceous earth, is removed via filtration or centrifugation. The patent highlights that this immobilized enzyme can be reused extensively; experimental data within the document cites successful recycling of the biocatalyst for over 150 cycles. This durability not only lowers the operational cost but also ensures batch-to-batch consistency, a critical parameter for regulatory compliance in pharmaceutical and food-grade manufacturing. The unreacted isomers are then subjected to a racemization catalyst, such as nickel or solid oxides, at elevated temperatures to scramble their stereochemistry, regenerating the feedstock for the next resolution cycle.
How to Synthesize (-)-Menthol Efficiently
The synthesis pathway described in the patent offers a scalable route for producing high-purity (-)-menthol suitable for industrial application. The process integrates classical petrochemical transformations with modern green chemistry principles, specifically utilizing biocatalysis to achieve chiral purity. For R&D teams looking to implement this technology, the procedure involves a sequential workflow starting from readily available phenolic precursors. The initial alkylation and hydrogenation steps establish the carbon skeleton, while the subsequent enzymatic step installs the critical stereochemistry. Detailed standardized synthesis steps follow below to guide the technical implementation of this route.
- Alkylate m-cresol (Formula I) with isopropanol or propylene using a zeolite or Lewis acid catalyst to form Thymol (Formula II).
- Hydrogenate Thymol over a nickel or platinum catalyst at elevated temperatures (160-200°C) to generate a racemic mixture of menthol stereoisomers (Formula III).
- Perform kinetic resolution using a stereoselective lipase (e.g., Amano AK) and vinyl acetate in n-heptane to selectively esterify the desired (-)-isomer.
- Separate the ester, hydrolyze it to recover pure (-)-menthol, and racemize the remaining unconverted isomers for recycling back into the resolution step.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the technology described in CN1240646C represents a strategic move towards supply security and cost optimization. The primary vulnerability in the menthol market has always been its reliance on agricultural output, which is prone to weather disruptions and geopolitical instability. By shifting to a fully synthetic process that mimics the quality of the natural product, companies can secure a reliable flavor and fragrance intermediate supplier status that is immune to crop failures. The enzymatic resolution process eliminates the need for expensive chiral pool starting materials, relying instead on cheap racemic mixtures that are easier and cheaper to manufacture in bulk. This fundamental shift in raw material strategy decouples production costs from the volatility of essential oil markets, providing a stable pricing structure for long-term contracts.
- Cost Reduction in Manufacturing: The economic model of this process is driven by the high reusability of the biocatalyst and the recycling of byproducts. In traditional resolutions, the unwanted 50% of the material is a sunk cost, but here, the racemization loop converts waste back into value. Qualitatively, this implies a drastic reduction in raw material consumption per kilogram of final product. Furthermore, the ability to reuse the immobilized enzyme for hundreds of cycles amortizes the cost of the biocatalyst over a massive production volume, significantly lowering the variable cost component. The mild reaction conditions also translate to lower energy consumption compared to high-pressure or cryogenic separation methods, contributing to overall operational expenditure savings.
- Enhanced Supply Chain Reliability: Synthetic production facilities can operate continuously, 24 hours a day, 365 days a year, unlike agricultural harvesting which is seasonal. This continuous manufacturing capability ensures a steady flow of inventory, reducing the need for safety stock and minimizing the risk of stockouts during peak demand periods. The use of robust, immobilized enzymes simplifies the logistics of catalyst management, as the biocatalyst does not require cold chain storage or frequent replenishment. This stability allows supply chain planners to forecast production with high accuracy, ensuring that downstream customers in the confectionery, oral care, and pharmaceutical sectors receive their shipments on time, every time.
- Scalability and Environmental Compliance: The process is designed for scalability, utilizing standard unit operations like stirred tank reactors and distillation columns that are common in fine chemical plants. The environmental footprint is significantly reduced due to the atom-economic nature of the recycling loop and the absence of heavy metal waste streams often associated with asymmetric chemical hydrogenation. The use of organic solvents like n-heptane allows for efficient recovery and reuse, minimizing volatile organic compound (VOC) emissions. This alignment with green chemistry principles facilitates easier regulatory approval and enhances the corporate sustainability profile, which is increasingly important for multinational corporations aiming to meet ESG (Environmental, Social, and Governance) targets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this enzymatic resolution technology. These answers are derived directly from the experimental data and claims found within the patent documentation, providing clarity on the feasibility and advantages of the method for potential partners and technical stakeholders.
Q: How does the enzymatic resolution process improve purity compared to traditional distillation?
A: Traditional fractional distillation struggles to separate menthol stereoisomers due to similar boiling points, often requiring multiple passes. The enzymatic method described in CN1240646C utilizes the high stereospecificity of lipases (like Amano AK) to chemically differentiate the (-)-isomer by converting it into an ester, allowing for physical separation from unreacted isomers with significantly higher optical purity.
Q: What are the economic benefits of recycling unconverted isomers in this process?
A: In a standard resolution, 50% of the material (the unwanted isomers) is often discarded or sold at a lower value. This patent introduces a racemization step where unconverted isomers (like (+)-menthol or neomenthol) are chemically reverted to a racemic mixture and fed back into the reactor. This theoretical 100% yield potential drastically reduces raw material costs and waste generation.
Q: Is the enzyme catalyst reusable for large-scale manufacturing?
A: Yes, the patent data indicates high reusability. In specific embodiments, the lipase catalyst was successfully recycled up to 150 times without significant loss of activity. This longevity minimizes the cost per kilogram of product attributed to biocatalysts, making the process economically viable for industrial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (-)-Menthol Supplier
The technological potential of enzymatic resolution for (-)-menthol production is immense, offering a pathway to high-purity products that meet the stringent requirements of the global flavor and pharmaceutical markets. NINGBO INNO PHARMCHEM stands ready to leverage this expertise as your trusted partner. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of synthetic menthol matches the sensory and chemical profile of the finest natural extracts. We understand the critical nature of supply continuity and are committed to deploying robust manufacturing protocols that guarantee consistency.
We invite you to engage with our technical procurement team to discuss how this innovative process can be tailored to your specific volume requirements. By partnering with us, you gain access to a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this synthetic route for your specific application. We encourage you to request specific COA data and route feasibility assessments to validate the compatibility of our high-purity (-)-menthol with your existing formulations. Let us collaborate to build a resilient, cost-effective, and sustainable supply chain for your menthol needs.
