Revolutionizing Chiral Allenol Production: Enzymatic Kinetic Resolution for Scalable Pharma Intermediates
The landscape of asymmetric synthesis is continually evolving, driven by the demand for greener, safer, and more efficient pathways to chiral building blocks. Patent CN1304346C introduces a significant advancement in this domain by disclosing a novel, environmentally friendly method for the preparation of optically active 2,3-alkenol and allenol ester compounds. These molecules serve as pivotal intermediates in the construction of complex pharmacophores, including epoxy compounds, amino alcohols, and furan derivatives. Unlike conventional approaches that often rely on hazardous reagents, this invention leverages the power of biocatalysis, specifically utilizing lipases to perform kinetic resolution on racemic mixtures. This shift from chemocatalysis to biocatalysis represents a paradigm change in how fine chemical manufacturers approach the production of high-value chiral intermediates, offering a route that is not only chemically robust but also aligned with modern sustainability goals.
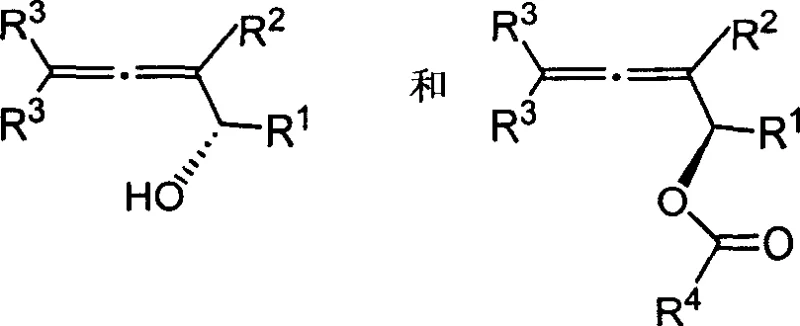
Historically, the synthesis of optically active 2,3-dien-1-ols has been fraught with significant technical and safety challenges that hindered their widespread adoption in large-scale manufacturing. Conventional methodologies frequently depended on the use of highly toxic organotin reagents, such as propargyl tin, which pose severe environmental risks and complicate waste disposal protocols. Furthermore, these traditional routes often necessitated the use of difficult-to-synthesize chiral ligands, like diphenylethylenediamine boron reagents, leading to elongated synthetic sequences and increased production costs. Perhaps most critically for industrial operations, these reactions typically required rigorous anhydrous and oxygen-free conditions, demanding specialized equipment and inert atmosphere handling that drastically reduced operational flexibility and throughput. The cumulative effect of these limitations was a manufacturing process that was expensive, hazardous, and difficult to scale, creating a bottleneck for the supply of these valuable chiral intermediates to the broader pharmaceutical and agrochemical industries.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
The reliance on stoichiometric chiral auxiliaries and toxic metal reagents in traditional synthesis creates a cascade of inefficiencies that extend far beyond the reaction vessel. The necessity for strictly anhydrous conditions means that raw materials must be meticulously dried, and reactors must be purged with inert gases, adding substantial time and energy costs to every batch. Moreover, the removal of tin residues from the final product is a notorious purification challenge, often requiring extensive chromatography or complex extraction procedures that lower overall yield and generate significant solvent waste. From a regulatory perspective, the presence of heavy metal impurities is a major red flag for pharmaceutical applications, necessitating additional testing and validation steps to ensure compliance with strict ICH guidelines. These factors combined make the conventional chemical synthesis of chiral allenols economically unviable for cost-sensitive applications and operationally risky for facilities aiming to minimize their environmental footprint and safety liabilities.
The Novel Approach
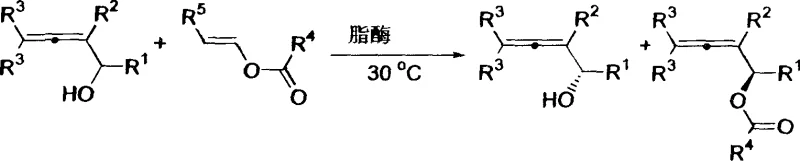
In stark contrast to these cumbersome legacy methods, the enzymatic kinetic resolution strategy outlined in the patent offers a streamlined and benign alternative that fundamentally simplifies the production workflow. By employing commercially available lipases, such as Candida antarctica lipase B (Novozym 435) or Pseudomonas fluorescens lipase, the process achieves high enantioselectivity under remarkably mild conditions. The reaction proceeds efficiently in common organic solvents like n-hexane or even in the acylating agent itself, vinyl acetate, at temperatures ranging from 25°C to 60°C. Crucially, this biocatalytic approach does not require the exclusion of air or moisture, allowing for standard reactor setups and significantly reducing infrastructure costs. The result is a robust process that delivers both the unreacted chiral alcohol and the acylated ester product with exceptional optical purity, often exceeding 98% ee, while eliminating the need for toxic heavy metals and complex chiral ligand synthesis.
Mechanistic Insights into Lipase-Catalyzed Kinetic Resolution
The core of this technological breakthrough lies in the stereoselective nature of the lipase enzyme, which acts as a chiral filter within the reaction mixture. In a kinetic resolution scenario, the lipase actively distinguishes between the two enantiomers of the racemic 2,3-dien-1-ol substrate based on their spatial arrangement within the enzyme's active site. One enantiomer fits perfectly, allowing the enzyme to catalyze the transesterification with vinyl acetate rapidly, converting it into the corresponding allenol ester. The other enantiomer, due to steric clashes or improper orientation, reacts extremely slowly or not at all, remaining largely as the free alcohol. This differential reaction rate allows for the effective separation of the two enantiomers simply by stopping the reaction at approximately 50% conversion. The specificity of enzymes like Novozym 435 ensures that the resulting products—the free alcohol and the ester—are obtained in high enantiomeric excess, providing two distinct chiral building blocks from a single racemic starting material.
From an impurity control perspective, this enzymatic mechanism offers inherent advantages that are difficult to replicate with small-molecule catalysts. Because the reaction is highly specific, side reactions such as polymerization of the allene system or rearrangement of the double bonds are minimized. The mild reaction conditions further suppress thermal degradation pathways that might otherwise compromise the integrity of the sensitive allene moiety. Furthermore, the use of vinyl acetate as the acyl donor drives the equilibrium forward irreversibly by generating acetaldehyde as a byproduct, which prevents the reverse hydrolysis reaction and ensures high conversion rates for the reactive enantiomer. This thermodynamic drive, combined with the kinetic selectivity of the enzyme, results in a clean reaction profile that simplifies downstream purification, reducing the burden on quality control laboratories and ensuring a consistent, high-purity product stream suitable for sensitive pharmaceutical applications.
How to Synthesize Optically Active 2,3-Allenols Efficiently
Implementing this synthesis route requires careful attention to enzyme selection and reaction monitoring to maximize the yield and optical purity of the target intermediates. The process begins with the dissolution of the racemic allenol substrate in a suitable organic solvent, followed by the addition of the lipase catalyst and the acylating agent. The mixture is then agitated at a controlled temperature, typically around 30°C, for a period extending from 10 to 96 hours depending on the specific substrate reactivity. Regular sampling and analysis via chiral HPLC are recommended to determine the optimal endpoint for the resolution, ensuring that the balance between conversion and enantiomeric excess is maintained. Once the desired conversion is reached, the immobilized enzyme is easily removed by filtration, allowing for potential reuse, and the products are isolated through standard workup procedures such as solvent evaporation and silica gel chromatography.
- Prepare the reaction mixture by combining racemic 2,3-dien-1-ol, vinyl acetate as the acylating agent, and a selected lipase catalyst (e.g., Candida antarctica lipase B) in an organic solvent such as n-hexane or vinyl acetate itself.
- Maintain the reaction temperature between 25°C and 60°C, ideally around 30°C, and stir the mixture for a duration ranging from 10 to 96 hours to ensure sufficient conversion and enantioselectivity.
- Upon completion, filter the mixture to remove the immobilized enzyme, wash the enzyme cake with ether, concentrate the filtrate, and purify the resulting optically active allenol and allenol ester via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this enzymatic synthesis route offers tangible strategic benefits that directly impact the bottom line and operational resilience. The elimination of toxic organotin reagents removes a significant regulatory hurdle and reduces the costs associated with hazardous waste disposal and environmental compliance. By avoiding the need for specialized anhydrous and oxygen-free infrastructure, manufacturing facilities can utilize existing general-purpose reactors, thereby lowering capital expenditure requirements and increasing production flexibility. The simplicity of the workup procedure, which involves basic filtration and chromatography rather than complex metal scavenging steps, translates into shorter cycle times and higher throughput. These efficiencies collectively contribute to a more reliable supply chain capable of meeting fluctuating market demands without the bottlenecks associated with hazardous chemical handling.
- Cost Reduction in Manufacturing: The economic implications of switching to this lipase-catalyzed method are profound, primarily driven by the simplification of the raw material portfolio and process operations. Traditional methods require the procurement of expensive and often custom-synthesized chiral ligands and toxic organometallic reagents, which carry high price tags and volatile supply risks. In contrast, lipases are robust, commercially available biocatalysts that can often be recovered and reused multiple times, drastically reducing the cost per kilogram of catalyst consumed. Additionally, the ability to run reactions under ambient atmospheric conditions eliminates the energy costs associated with maintaining inert gas blankets and drying solvents. The reduction in purification complexity further lowers solvent consumption and labor costs, resulting in a significantly leaner cost structure for the production of high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Supply chain stability is greatly enhanced by the use of widely available and stable reagents inherent to this biological process. Unlike specialized organotin compounds that may be subject to strict shipping regulations and limited supplier bases, lipases and vinyl acetate are commodity chemicals with robust global supply networks. This diversification of the supply base mitigates the risk of production stoppages due to raw material shortages. Furthermore, the operational simplicity of the process means that it can be easily transferred between different manufacturing sites or scaled up without the need for highly specialized technical expertise or unique equipment configurations. This flexibility ensures continuity of supply even in the face of logistical disruptions, providing a secure source of critical chiral intermediates for downstream drug synthesis.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety and environmental issues, but this enzymatic route inherently mitigates those risks, making it ideal for commercial expansion. The absence of pyrophoric or highly toxic reagents means that safety protocols are less burdensome, allowing for larger batch sizes without proportional increases in risk management overhead. From an environmental standpoint, the process aligns with Green Chemistry principles by reducing waste generation and avoiding persistent toxic pollutants. This compliance facilitates easier permitting for new production lines and enhances the corporate sustainability profile, which is increasingly important for partnerships with major multinational pharmaceutical companies that prioritize eco-friendly supply chains. The combination of safety, scalability, and sustainability makes this technology a future-proof investment for long-term manufacturing strategies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this lipase-catalyzed synthesis technology. These insights are derived directly from the experimental data and technical specifications detailed in the patent literature, providing a factual basis for evaluating the feasibility of adopting this method for your specific production needs. Understanding these nuances is essential for R&D teams planning process validation and procurement officers assessing supplier capabilities.
Q: What are the primary advantages of using lipase catalysis over traditional organotin methods for synthesizing chiral allenols?
A: Traditional methods often rely on toxic organotin reagents and require stringent anhydrous and oxygen-free conditions, posing significant safety and environmental hazards. The lipase-catalyzed method described in patent CN1304346C eliminates the need for toxic heavy metals, operates under mild conditions without strict exclusion of air or moisture, and simplifies post-reaction processing, making it far more suitable for industrial scale-up and regulatory compliance.
Q: What level of enantiomeric excess (ee) can be achieved with this enzymatic resolution process?
A: Experimental data within the patent demonstrates that this kinetic resolution process can achieve exceptionally high enantiomeric purity. For various substrates, including those with alkyl, alkenyl, and benzyl substituents, the resulting optically active 2,3-alkenols and their corresponding esters consistently exhibit ee values exceeding 98%, with many examples reaching 99% or higher, ensuring high-quality intermediates for asymmetric synthesis.
Q: What are the downstream applications of these optically active 2,3-alkenol compounds?
A: These chiral building blocks are highly versatile precursors in organic synthesis. They can be readily transformed into a variety of valuable structures, including optically active epoxy compounds, amino alcohols, and furan derivatives through reactions such as Diels-Alder cycloadditions, aldol condensations, and carbonyl allylations, making them critical intermediates for the development of complex pharmaceutical agents and agrochemicals.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Optically Active 2,3-Allenols Supplier
As the demand for chiral intermediates continues to grow, partnering with a manufacturer that possesses deep technical expertise in advanced synthesis technologies is crucial for maintaining a competitive edge. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging cutting-edge biocatalytic methods like the one described in patent CN1304346C to deliver superior quality intermediates. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements without compromising on quality. We operate stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of optically active 2,3-allenols meets the highest standards of enantiomeric excess and chemical purity required for pharmaceutical applications.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs through the adoption of these innovative synthetic routes. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific project requirements, demonstrating exactly how switching to our enzymatically produced intermediates can improve your margins. Please contact us today to request specific COA data for our current inventory or to discuss route feasibility assessments for your custom synthesis projects. Let us be your partner in transforming complex chemical challenges into commercial successes.
