Advanced Synthesis of Branched Glycidyl Esters for High-Performance Industrial Coatings
Advanced Synthesis of Branched Glycidyl Esters for High-Performance Industrial Coatings
The chemical industry continuously seeks methodologies that balance high purity with economic viability, particularly in the realm of reactive diluents and resin modifiers. Patent CN1325392A introduces a transformative approach for the preparation of glycidyl esters of branched carboxylic acids, specifically targeting alpha-branched monocarboxylic acids containing 5 to 20 carbon atoms. This technology addresses the longstanding challenge of color instability and heavy by-product formation that has plagued conventional synthesis routes. By optimizing the reaction environment through the strategic use of water-miscible solvents and precise catalyst dosing, this process achieves glycidyl esters with exceptional purity levels, often exceeding 96 percent, while maintaining a Pt-Co color value significantly lower than prior art standards. For R&D directors and procurement specialists alike, this represents a critical advancement in securing reliable supply chains for high-performance coating additives.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
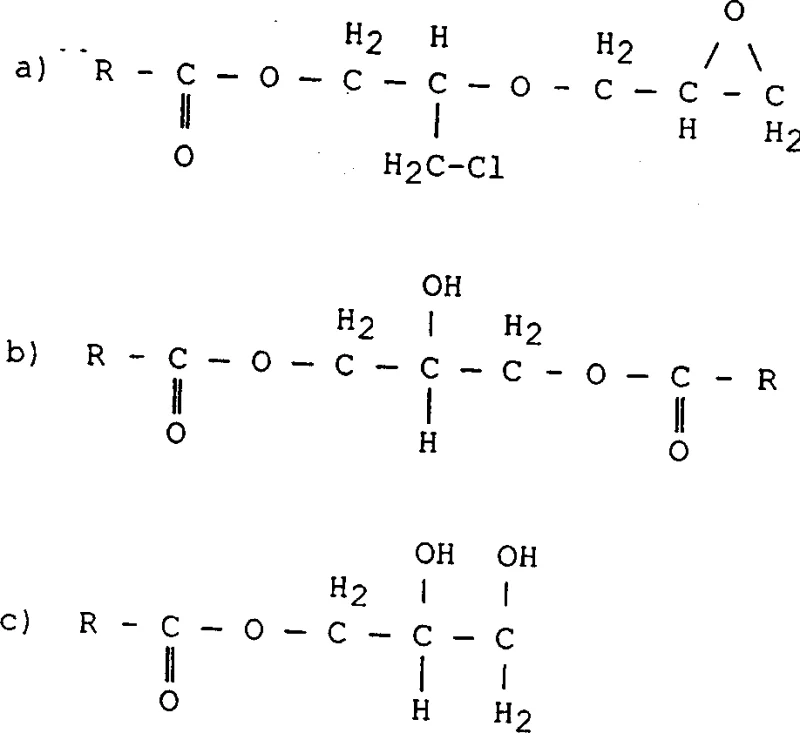
Historically, the production of glycidyl esters involved reacting carboxylic acid salts with excess epichlorohydrin, followed by heating and subsequent purification. However, these traditional pathways suffered from inherent thermodynamic and kinetic limitations that resulted in the formation of substantial quantities of heavier by-products, typically ranging from 8 percent to 12 percent by weight. These impurities, often formed through the reaction of the glycidyl ester product with unreacted epichlorohydrin or free acid, contained functional groups that were prone to thermal degradation. Consequently, attempting to purify the product via conventional atmospheric or even vacuum distillation often exacerbated the problem, leading to darker colors and further decomposition. To achieve acceptable purity levels of 99 percent, manufacturers were frequently forced to sacrifice up to 30 percent of their yield during the distillation of tailings, creating a significant economic burden and generating large volumes of chlorine-containing waste that posed environmental compliance challenges.
The Novel Approach
The methodology disclosed in CN1325392A fundamentally alters the reaction landscape by introducing a multi-step protocol that prioritizes impurity suppression over post-reaction correction. Instead of relying solely on distillation to remove contaminants, this novel approach utilizes a water-miscible solvent, preferably an alkanol such as isopropanol, during the initial esterification phase. This solvent choice facilitates the dissolution of the alkaline catalyst into the organic phase, ensuring a more homogeneous reaction environment that minimizes localized hot spots and side reactions. Furthermore, the process employs a staged addition of alkali metal hydroxides, first to form the intermediate and subsequently to drive the dehydrohalogenation to completion under controlled temperatures between 0 and 80 degrees Celsius. This precise control prevents the thermal stress that typically generates colored bodies, allowing for the production of glycidyl esters with heavy by-product content reduced to less than 6 percent, and ideally below 4 percent, without the need for yield-sacrificing purification steps.
Mechanistic Insights into Solvent-Mediated Catalytic Esterification
The core innovation lies in the mechanistic interplay between the solvent system and the catalytic species during the formation of the chlorohydrin ester intermediate. In conventional systems, the heterogeneity between the aqueous base and the organic acid/epoxide mixture often leads to incomplete conversion and the accumulation of hydrolysable chlorine. By incorporating a solvent like isopropanol, the patent demonstrates that the solubility of the base is enhanced, allowing for a more efficient nucleophilic attack on the epoxide ring. This results in a cleaner conversion to the chlorohydrin ester, which is the precursor to the final glycidyl ring closure. The reaction is conducted at moderate temperatures, typically between 50 and 110 degrees Celsius in the first stage, which is sufficient to drive kinetics without triggering the polymerization or degradation of the sensitive epoxide functionality. The subsequent addition of base in the second stage is carefully calibrated to a molar ratio of 0.9:1 to 1.2:1 relative to the acid, ensuring that enough hydroxide ions are available to close the epoxide ring without leaving excessive residual base that could hydrolyze the newly formed glycidyl ester.
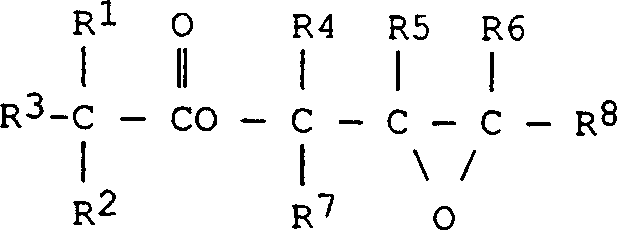
Impurity control is further achieved through the management of water content throughout the synthesis. While water is present in the initial reaction mixture to facilitate salt formation, the process includes a distillation step to remove excess epichlorohydrin, solvent, and formed water before the final dehydrohalogenation. This is critical because high water content in the final basic treatment step can lead to the hydrolysis of the glycidyl ester back to the diol, reducing the epoxy equivalent weight (EGC) and compromising the reactivity of the final product. The patent highlights that maintaining hydrolysable chlorine levels below 2500 mg/kg after the intermediate stage is essential for achieving the final specification of less than 300 mg/kg in the purified product. This rigorous control over the reaction microenvironment ensures that the final glycidyl ester retains its structural integrity, as represented by the general formula where R groups denote branched alkyl chains, providing the steric hindrance necessary for hydrolytic stability in end-use applications.
How to Synthesize Glycidyl Esters Efficiently
The synthesis of these high-value intermediates requires strict adherence to the sequential addition of reagents and temperature profiles outlined in the patent data. The process begins with the charging of the alpha-branched acid and excess epichlorohydrin into a reactor equipped with efficient agitation and temperature control. The detailed standardized synthesis steps involve precise molar ratios of catalysts and solvents to ensure reproducibility at scale. Operators must monitor the reaction progress via titration or chromatographic methods to confirm the consumption of the acid before proceeding to the base addition phase. The following guide summarizes the critical operational parameters required to replicate the high yields and low color values demonstrated in the experimental examples.
- React alpha-branched monocarboxylic acid with excess epichlorohydrin in the presence of a water-miscible solvent and catalyst at 30-110°C.
- Add alkali metal hydroxide or alkoxide to reach an equimolar ratio and react at 0-80°C to form the chlorohydrin ester intermediate.
- Distill off excess reagents and solvents, then treat with concentrated alkali to complete dehydrohalogenation and wash to remove salts.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented process translates directly into tangible operational efficiencies and risk mitigation. Traditional manufacturing routes for glycidyl esters often incurred hidden costs associated with yield loss during purification and the disposal of hazardous waste streams. By shifting to this solvent-mediated, low-temperature cyclization method, manufacturers can significantly reduce the unit cost of production. The elimination of aggressive thin-film distillation steps not only preserves yield but also reduces energy consumption and equipment wear, leading to a more sustainable and cost-effective manufacturing footprint. This efficiency allows suppliers to offer more competitive pricing structures while maintaining healthy margins, a crucial factor in the volatile raw material market of the coatings industry.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the drastic reduction in yield loss. Conventional methods often required sacrificing substantial portions of the product to achieve high purity, effectively increasing the cost per kilogram of the saleable good. By minimizing heavy by-product formation to below 6 percent through superior reaction engineering, this method maximizes the conversion of expensive raw materials like Versatic acid and epichlorohydrin into the final product. Furthermore, the ability to operate at lower temperatures during the critical cyclization phase reduces energy demands for heating and cooling, contributing to overall lower utility costs. The qualitative improvement in process efficiency means that capital expenditure on complex distillation columns can be minimized or deferred, freeing up resources for other strategic investments.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the complexity of purification steps that are prone to fouling or operational upsets. The simplified workflow of this novel approach, which relies on robust liquid-liquid separations and standard distillation rather than specialized short-path equipment, enhances the reliability of production schedules. Raw materials such as isopropanol and sodium hydroxide are commodity chemicals with stable global supply chains, reducing the risk of bottlenecks associated with specialty catalysts or reagents. This robustness ensures that a reliable glycidyl ester supplier can maintain consistent delivery timelines even during periods of market fluctuation, providing downstream formulators with the confidence needed for their own production planning.
- Scalability and Environmental Compliance: From an environmental perspective, the reduction in chlorine-containing waste is a significant advantage. Older processes generated large volumes of saline waste and chlorinated organics that required costly treatment. The improved selectivity of this reaction minimizes the generation of such by-products, aligning with increasingly stringent global environmental regulations. Additionally, the patent confirms that the method is suitable for both batch and continuous operation, offering flexibility for commercial scale-up of complex polymer additives. Whether producing 100 kilograms for pilot testing or 100 metric tons for annual commercial production, the chemistry remains consistent, ensuring that quality specifications are met regardless of batch size.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this advanced synthesis technology. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on how this method outperforms legacy technologies in terms of purity, color, and reactivity. Understanding these nuances is vital for technical teams evaluating the feasibility of integrating these intermediates into new resin formulations.
Q: How does this process improve color stability compared to conventional methods?
A: By minimizing the formation of heavy by-products (less than 6% by weight) through controlled solvent use and specific base addition sequences, the process avoids the thermal degradation associated with aggressive distillation, resulting in significantly lighter initial color and better aging stability.
Q: What are the key raw materials required for this synthesis?
A: The primary inputs include alpha-branched monocarboxylic acids (such as Versatic acid), a halogen-substituted monoepoxide like epichlorohydrin, a water-miscible solvent such as isopropanol, and alkali metal hydroxides or alkoxides.
Q: Can this method be scaled for industrial production?
A: Yes, the patent explicitly states the method can be carried out in both batch and continuous modes, making it highly suitable for commercial scale-up of complex polymer additives without the yield losses typical of older purification techniques.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Glycidyl Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to industrial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN1325392A are fully realized in every drum we ship. We maintain stringent purity specifications and operate rigorous QC labs to verify that every batch of glycidyl ester meets the exacting standards required for high-performance epoxy and acrylic resin applications. Our commitment to quality assurance means that you receive a product with consistent epoxy equivalent weight and minimal color, ready for immediate integration into your formulation lines.
We invite you to collaborate with us to explore how this advanced chemistry can optimize your specific application needs. Our technical procurement team is prepared to provide a Customized Cost-Saving Analysis tailored to your current volume requirements, demonstrating exactly how switching to our optimized grade can impact your bottom line. Please contact us today to request specific COA data and route feasibility assessments for your next project, and let us demonstrate why we are the preferred partner for cost reduction in coating additive manufacturing.
